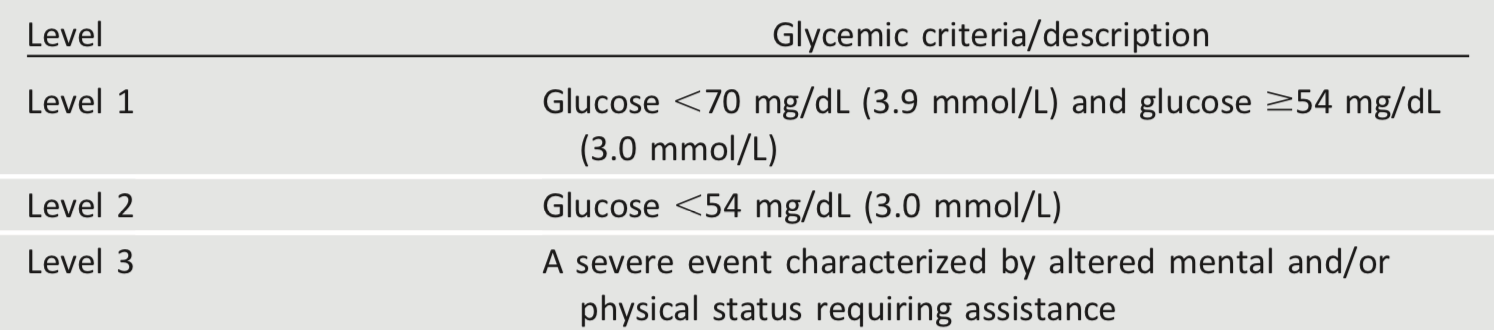
Hypoglycemia is the major limiting factor in the glycemic management of type 1 and type 2 diabetes. Recommendations regarding the classification of hypoglycemia are outlined in Table 6.3 (44). Level 1 hypoglycemia is defined as a measurable glucose concentration <70 mg/dL (3.9 mmol/L) but ≥54 mg/dL (3.0 mmol/L). A blood glucose concentration of 70 mg/dL (3.9 mmol/L) has been recognized as a threshold for neuroendocrine responses to falling glucose in people without diabetes. Because many people with diabetes demonstrate impaired counterregulatory responses to hypoglycemia and/or experience hypoglycemia unawareness, a measured glucose level <70 mg/dL (3.9 mmol/L) is considered clinically important, independent of the severity of acute hypoglycemic symptoms. Level 2 hypoglycemia (defined as a blood glucose concentration <54 mg/dL [3.0 mmol/L]) is the threshold at which neuroglycopenic symptoms begin to occur and requires immediate action to resolve the hypoglycemic event. Lastly, level 3 hypoglycemia is defined as a severe event characterized by altered mental and/or physical functioning that requires assistance from another person for recovery.
Studies of rates of level 3 hypoglycemia that rely on claims data for hospitalization, emergency department visits, and ambulance use substantially underestimate rates of level 3 hypoglycemia (45), yet find high burden of hypoglycemia in adults over 60 years of age in the community (46). African Americans are at substantially increased risk of level 3 hypoglycemia (46,47). In addition to age and race, other important risk factors found in a community-based epidemiologic cohort of older black and white adults with type 2 diabetes include insulin use, poor or moderate versus good glycemic control, albuminuria, and poor cognitive function (46).
Symptoms of hypoglycemia include, but are not limited to, shakiness, irritability, confusion, tachycardia, and hunger. Hypoglycemia may be inconvenient or frightening to patients with diabetes. Level 3 hypoglycemia may be recognized or unrecognized and can progress to loss of consciousness, seizure, coma, or death. It is reversed by administration of rapid-acting glucose or glucagon. Hypoglycemia can cause acute harm to the person with diabetes or others, especially if it causes falls, motor vehicle accidents, or other injury. A large cohort study suggested that among older adults with type 2 diabetes, a history of level 3 hypoglycemia was associated with greater risk of dementia (48). Conversely, in a substudy of the ACCORD trial, cognitive impairment at baseline or decline in cognitive function during the trial was significantly associated with subsequent episodes of level 3 hypoglycemia (49). Evidence from DCCT/EDIC, which involved adolescents and younger adults with type 1 diabetes, found no association between frequency of level 3 hypoglycemia and cognitive decline (50), as discussed in Section 13 “Children and Adolescents.”
Level 3 hypoglycemia was associated with mortality in participants in both the standard and the intensive glycemia arms of the ACCORD trial, but the relationships between hypoglycemia, achieved A1C, and treatment intensity were not straight-forward. An association of level 3 hypoglycemia with mortality was also found in the ADVANCE trial (51). An association between self-reported level 3 hypoglycemia and 5-year mortality has also been reported in clinical practice (52).
Young children with type 1 diabetes and the elderly, including those with type 1 and type 2 diabetes (48,53), are noted as particularly vulnerable to hypoglycemia because of their reduced ability to recognize hypoglycemic symptoms and effectively communicate their needs. Individualized glucose targets, patient education, dietary intervention (e.g., bedtime snack to prevent overnight hypoglycemia when specifically needed to treat low blood glucose), exercise management, medication adjustment, glucose monitoring, and routine clinical surveillance may improve patient outcomes (54). CGM with automated low glucose suspend has been shown to be effective in reducing hypoglycemia in type 1 diabetes (55). For patients with type 1 diabetes with level 3 hypoglycemia and hypoglycemia unawareness that persists despite medical treatment, human islet transplantation may be an option, but the approach remains experimental (56,57).
In 2015, the ADA changed its preprandial glycemic target from 70–130 mg/dL (3.9–7.2 mmol/L) to 80–130 mg/dL (4.4–7.2 mmol/L). This change reflects the results of the ADAG study, which demonstrated that higher glycemic targets corresponded to A1C goals (7). An additional goal of raising the lower range of the glycemic target was to limit overtreatment and provide a safety margin in patients titrating glucose-lowering drugs such as insulin to glycemic targets.