
1.0.0.0 Type 1 Diabetes
1.1.0.0 Introduction
The American Diabetes Association (ADA) “Standards of Medical Care in Diabetes” includes ADA’s current clinical practice recommendations and is intended to provide the components of diabetes care, general treatment goals and guidelines, and tools to evaluate quality of care. Members of the ADA Professional Practice Committee, a multidisciplinary expert committee, are responsible for updating the Standards of Care annually, or more frequently as warranted. For a detailed description of ADA standards, statements, and reports, as well as the evidence-grading system for ADA’s clinical practice recommendations, please refer to the Standards of Care Introduction. Readers who wish to comment on the Standards of Care are invited to do so at professional.diabetes.org/SOC.
The management of diabetes in children and adolescents cannot simply be derived from care routinely provided to adults with diabetes. The epidemiology, pathophysiology, developmental considerations, andresponse totherapy inpediatric-onset diabetes are different from adult diabetes. There are also differences in recommended care for children and adolescents with type 1 as opposed to type 2 diabetes. This section first addresses care for children and adolescents with type 1 diabetes and next addresses care for children and adolescents with type 2 diabetes. Figure 13.1 provides guidance on managing new-onset diabetes in overweight youth before type 1 or type 2 diabetes is diagnosed and so applies to all overweight youth. Lastly, guidance is provided in this section on transition of care from pediatric to adult providers to ensure that the continuum of care is appropriate as the child with diabetes develops into adulthood. Due to the nature of clinical research in children, the recommendations for children and adolescents with diabetes are less likely to be based on clinical trial evidence. However, expert opinion and a review of available and relevant experimental data are summarized in the American Diabetes Association (ADA) position statements “Type 1 Diabetes in Children and Adolescents” (1) and “Evaluation and Management of Youth-Onset Type 2 Diabetes” (2). The ADA consensus report “Youth-Onset Type 2 Diabetes Consensus Report: Current Status, Challenges, and Priorities” (3) characterizes type 2 diabetes in children and evaluates treatment options as well, but also discusses knowledge gaps and recruitment challenges in clinical and translational research in youth-onset type 2 diabetes.
TYPE 1 DIABETES
Type 1 diabetes is the mostcommon form of diabetes in youth (4), although recent data suggestthat itmay accountfor a large proportionofcases diagnosed in adultlife (5). The provider must consider the unique aspects of care and management of children and adolescents with type 1 diabetes, such as changes in insulin sensitivity related to physical growth and sexual maturation, ability to provide self-care, supervision in the child care and school environment, neurological vulnerability to hypoglycemia and hyperglycemia in young children, as well as possible adverse neurocognitive effects of diabetic ketoacidosis (DKA) (6,7). Attention to family dynamics, developmental stages, and physiologic differences related to sexual maturity is essential in developing and implementing an optimal diabetes treatment plan (8).
A multidisciplinary team of specialists trained in pediatric diabetes management and sensitive to the challenges of children and adolescents with type 1 diabetes and their families should provide care for this population. It is essential that diabetes self-management education and support, medical nutrition therapy, and psychosocial support be provided at diagnosis and regularly thereafter in a developmentally appropriate format that builds on prior knowledge by individuals experienced with the educational, nutritional, behavioral, and emotional needs of the growing child and family. The appropriate balance between adult supervision and independent selfcare should be defined at the first interaction and reevaluated at subsequent visits, with the expectation that it will evolve as the adolescent gradually becomes an emerging young adult.
Suggested citation: American Diabetes Association. 13. Children and adolescents: Standards of Medical Care in Diabetesd2019. Diabetes Care 2019;42(Suppl. 1):S148–S164 © 2018 by the American Diabetes Association. Readers may use this article as long as the work is properly cited, the use is educational and not for profit, and the work is not altered. More information is available at http://www.diabetesjournals .org/content/license.
1.2.0.0 Diabetes Self-management Education and Support
Recommendation
13.1 Youth with type 1 diabetes and parents/caregivers (for patients aged <18 years) should receive culturally sensitive and developmentally appropriate individualized diabetes self-management education and support according to national standards at diagnosis and routinely thereafter. B
No matter how sound the medical regimen, it can only be effective if the family and/or affected individuals are able to implement it. Family involvement is a vital component of optimal diabetes management throughout childhood and adolescence. Health care providers in the diabetes care team who care for children and adolescents must be capable of evaluating the educational, behavioral, emotional, and psychosocial factors that impact implementation of a treatment plan and must work with the individual and family to overcome barriers or redefine goals as appropriate. Diabetes self-management education and support requires periodic reassessment, especially as the youth grows, develops, and acquires the need for greater independent self-care skills. In addition, it is necessary to assess the educational needs and skills of day care providers, school nurses, or other school personnel who participate in the care of the young child with diabetes (9).
1.3.0.0 Nutrition Therapy
Recommendations
13.2 Individualized medical nutrition therapy is recommended for children and adolescents with type 1 diabetes as an essential component of the overall treatment plan. A
13.3 Monitoring carbohydrate intake, whether by carbohydrate counting or experience-based estimation, is key to achieving optimal glycemic control. B
13.4 Comprehensive nutrition education at diagnosis, with annual updates, by an experienced registered dietitian is recommended to assess caloric and nutrition intake in relation to weight status and cardiovascular disease risk factors and to inform macronutrient choices. E
Dietary management should be individualized: family habits, food preferences, religious or cultural needs, schedules, physical activity, and the patient’s and family’s abilities in numeracy, literacy, and self-management should be considered. Dietitian visits should include assessment for changes in food preferences over time, access to food, growth and development, weight status, cardiovascular risk, and potential for eating disorders. Dietary adherence is associated with better glycemic control in youth with type 1 diabetes (10).
1.4.0.0 Physical Activity and Exercise
Recommendations
13.5 Exercise is recommended for all youth with type 1 diabetes with the goal of 60 min of moderate-to vigorous-intensity aerobic activity daily, with vigorous muscle-strengthening and bone-strengthening activities at least 3 days per week. C
13.6 Education about frequent patterns of glycemia during and after exercise, which may include initial transient hyperglycemia followed by hypoglycemia, is essential. Families should also receive education on prevention and management of hypoglycemia during and after exercise, including ensuring patients have a preexercise glucose level of 90–250 mg/dL (5–13 mmol/L) and accessible carbohydrates before engaging in activity, individualized according to the type/intensity of the planned physical activity. E
13.7 Patients should be educated on strategies to prevent hypoglycemia during exercise, after exercise, and overnight following exercise, which may include reducing prandial insulin dosing for the meal/snack preceding (and, if needed, following) exercise, increasing carbohydrate intake, eating bedtime snacks, using continuous glucose monitoring, and/or reducing basal insulin doses. C
13.8 Frequent glucose monitoring before, during, and after exercise, with or without use of continuous glucose monitoring, is important to prevent, detect, and treat hypoglycemia and hyperglycemia with exercise. C
Exercise positively affects insulin sensitivity, physical fitness, strength building, weight management, social interaction, mood, self-esteem building, and creation of healthful habits for adulthood, but it also has the potential to cause both hypoglycemia and hyperglycemia.
See below for strategies to mitigate hypoglycemia risk and minimize hyperglycemia with exercise. For an in-depth discussion, see recently published reviews and guidelines (11-13).
Overall, it is recommended that youth with type 1 diabetes participate in 60 min of moderate- (e.g., brisk walking, dancing) to vigorous- (e.g., running, jumping rope intensity aerobic activity daily, including resistance and flexibility training (14). Although uncommon in the pediatric population, patients should be medically evaluated for comorbid conditions or diabetes complications thatmay restrictparticipation in an exercise program. As hyperglycemia can occur before, during, and after physical activity, it is important to ensure that the elevated glucose level is not related to insulin deficiency that would lead to worsening hyperglycemia with exercise and ketosis risk. Intense activity should be postponed with marked hyperglycemia (glucose ≥350 mg/dL [19.4 mmol/L]), moderate to large urine ketones, and/or b-hydroxybutyrate (B-OHB) >1.5 mmol/L. Caution may be needed when B-OHB levels are ≥0.6 mmol/L (10,11).
The prevention and treatment of hypoglycemia associated with physical activity include decreasing the prandial insulin for the meal/snack before exercise and/or increasing food intake. Patients on insulin pumps can lower basal rates by ˜10–50% or more or suspend for 1–2 h during exercise (15). Decreasing basal rates or long acting insulin doses by ˜20% after exercise may reduce delayed exercise-induced hypoglycemia (16). Accessible rapid-acting carbohydrates and frequent blood glucose monitoring before, during, and after exercise, with or without continuous glucose monitoring, maximize safety with exercise.
Blood glucose targets prior to exercise should be 90–250 mg/dL (5.0–13.9 mmol/L). Consider additional carbohydrate intake during and/or after exercise, depending on the duration and intensity of physical activity, to prevent hypoglycemia. For low- to moderate-intensity aerobic activities (30260 min), and if the patient is fasting, 10215 g of carbohydrate may prevent hypoglycemia (17). After insulin boluses (relative hyperinsulinemia), consider 0.5–1.0 g of carbohydrates/kg per hour of exercise (˜30–60 g), which is similar to carbohydrate requirements to optimize performance in athletes without type 1 diabetes (18-20).
In addition, obesity is as common in children and adolescents with type 1 diabetes as in those without diabetes. It is associated with higher frequency of cardiovascular risk factors, and it disproportionately affects racial/ethnic minorities in the U.S. (21-25). Therefore, diabetes care providers should monitor weight status and encourage a healthy diet, exercise, and healthy weight as key components of pediatric type 1 diabetes care.
1.5.0.0 School and Child Care
As a large portion of a child’s day is spent in school, close communication with and the cooperation of school or day care personnel are essential for optimal diabetes management, safety, and maximal academic opportunities. Refer to the ADA position statements “Diabetes Care in the School Setting” (26) and “Care of Young Children With Diabetes in the Child Care Setting” (27) for additional details.
1.6.0.0 Psychosocial Issues
Recommendations
13.9 At diagnosis and during routine follow-up care, assess psychosocial issues and family stresses that could impactdiabetes management and provide appropriate referrals to trained mental health professionals, preferably experienced in childhood diabetes. E
13.10 Mental health professionals should be considered integral members of the pediatric diabetes multidisciplinary team. E
13.11 Encourage developmentally appropriate family involvement in diabetes management tasks for children and adolescents, recognizing that premature transfer of diabetes care to the child can result in diabetes burn-out nonadherence and deterioration in glycemic control. A
13.12 Providers should consider asking youth and their parents about social adjustment (peer relationships) and school performance to determine whether further intervention is needed. B
13.13 Assess youth with diabetes for psychosocial and diabetes-related distress, generally starting at 7–8 years of age. B
13.14 Offer adolescents time by themselves with their care provider(s) starting at age 12 years, or when develop-mentally appropriate. E
13.15 Starting at puberty, preconception counseling should be incorporated into routine diabetes care for all girls of childbearing potential. A
13.16 Begin screening youth with type 1 diabetes for eating disorders between 10 and 12 years of age. The Diabetes Eating Problems Survey-Revised (DEPS-R) is a reliable, valid, and brief screening tool for identifying disturbed eating behavior. B
Rapid and dynamic cognitive, developmental, and emotional changes occur during childhood, adolescence, and emerging adulthood. Diabetes management during childhood and adolescence places substantial burdens on the youth and family, necessitating ongoing assessment of psychosocial status and diabetes distress in the patient and the caregiver during routine diabetes visits (28-34). Early detection of depression, anxiety, eating disorders, and learning disabilities can facilitate effective treatment options and help minimize adverse effects on diabetes management and disease outcomes (33,35). There are validated tools, such as the Problem Areas in Diabetes- Teen (PAID-T) and Parent (P-PAID-Teen) (34), that can be used in assessing diabetes-specific distress in youth starting at age 12 years and in their parent caregivers. Furthermore, the complexities of diabetes management require ongoing parental involvement in care throughout childhood with developmentally appropriate family teamwork between the growing child/teen and parent in order to maintain adherence and to prevent deterioration in glycemic control (36,37). As diabetes-specific family conflict is related to poorer adherence and glycemic control, it is appropriate to inquire about such conflict during visits and to either help to negotiate a plan for resolution or refer to an appropriate mental health specialist (38). Monitoring of social adjustment (peer relationships) and school performance can facilitate both well-being and academic achievement (39). Suboptimal glycemic control is a risk factor for underperformance at school and increased absenteeism (40). Shared decision making with youth regarding the adoption of regimen components and self-management behaviors can improve diabetes self-efficacy, adherence, and metabolic outcomes (22,41).
Although cognitive abilities vary, the ethical position often adopted is the “mature minor rule,” whereby children after age 12 or 13 years who appear to be “mature” have the right to consent or withhold consent to general medical treatment, except in cases in which refusal would significantly endanger health (42).
Beginning at the onset of puberty or at diagnosis of diabetes, all adolescent girls and women with childbearing potential should receive education about the risks of malformations associated with poor metabolic control and the use of effective contraception to prevent unplanned pregnancy. Preconception counseling using developmentally appropriate educational tools enables adolescent girls to make well-informed decisions (43). Pre-conception counseling resources tailored for adolescents are available at no cost through the ADA (44). Refer to the ADA position statement “Psychosocial Care for People With Diabetes” for further details (35).
Youth with type 1 diabetes have an increased risk of disordered eating behavior as well as clinical eating disorders with serious short-term and long-term negative effects on diabetes outcomes and health in general. Therefore, it is important to screen for eating disorders in youth with type 1 diabetes using tools such as the Diabetes Eating Problems Survey-Revised (DEPS-R) to allow for early diagnosis and intervention (45-48).
Screening
Screening for psychosocial distress and mental health problems is an important component of ongoing care. It is important to consider the impact of diabetes on quality of life as well as the development of mental health problems related to diabetes distress, fear of hypoglycemia (and hyperglycemia), symptoms of anxiety, disordered eating behaviors as well as eating disorders, and symptoms of depression (49). Consider assessing youth for diabetes distress, generally starting at 7 or 8 years of age (35). Consider screening for depression and disordered eating behaviors using available screening tools (28,45). With respect to disordered eating, it is important to recognize the unique and dangerous disordered eating behavior of insulin omission for weight control in type 1 diabetes (50). The presence of a mental health professional on pediatric multidisciplinary teams highlights the importance of attending to the psychosocial issues of diabetes. These psychosocial factors are significantly related to self-management difficulties, suboptimal glycemic control, reduced quality of life, and higher rates of acute and chronic diabetes complications.
1.7.0.0 Glycemic Control
Table 13.1—Blood glucose and A1C targets for children and adolescents with type 1 diabetes

Recommendations
13.17 The majority of children and adolescents with type 1 diabetes should be treated with intensive insulin regimens, either via multiple daily injections or continuous subcutaneous insulin infusion. A
13.18 All children and adolescents with type 1 diabetes should self-monitor glucose levels multiple times daily (up to 6–10 times/day), including premeal, prebedtime, and as needed for safety in specific situations such as exercise, driving, or the presence of symptoms of hypoglycemia. B
13.19 Continuous glucose monitoring should be considered in all children and adolescents with type 1 diabetes, whether using injections or continuous subcutaneous insulin infusion, as an additional tool to help improve glucose control. Benefits of continuous glucose monitoring correlate with adherence to ongoing use of the device. B
13.20 Automated insulin delivery systems appear to improve glycemic control and reduce hypoglycemia in children and should be considered in children with type 1 diabetes. B
13.21 An A1C target of <7.5% (58 mmol/mol) should be considered in children and adolescents with type 1 diabetes but should be individualized based on the needs and situation of the patient and family. E
Please refer to Section 7 “Diabetes Technology” for more information on the use of blood glucose meters, continuous glucose monitors, and insulin pumps. More information on insulin injection technique can be found in Section 9 “Pharmacologic Approaches to Glycemic Treatment,” p. S90.
Current standards for diabetes management reflect the need to lower glucose as safely as possible. This should be done with stepwise goals. When establishing individualized glycemic targets, special consideration should be given to the risk of hypoglycemia in young children (aged <6 years) who are often unable to recognize, articulate, and/or manage hypoglycemia. However, registry data indicate that lower A1C can be achieved in children, including those <6 years, without increased risk of severe hypoglycemia (51,52).
Type 1 diabetes can be associated with adverse effects on cognition during childhood and adolescence. Factors that contribute to adverse effects on brain development and function include young age or DKA at onset of type 1 diabetes, severe hypoglycemia at <6 years of age, and chronic hyperglycemia (53,54). However, meticulous use of new therapeutic modalities such as rapidand long-acting insulin analogs, technological advances (e.g., continuous glucose monitors, low-glucose suspend insulin pumps, and automated insulin delivery systems), and intensive self-management education now make it more feasible to achieve excellent glycemic control while reducing the incidence of severe hypoglycemia (55-64). Intermittently scanned continuous glucose monitors (sometimes referred to as “flash” continuous glucose monitors) are not currently approved for use in children and adolescents. A strong relationship exists between frequency of blood glucose monitoring and glycemic control (57-66).
The Diabetes Control and Complications Trial (DCCT), which did not enroll children <13 years of age, demonstrated that near normalization of blood glucose levels was more difficult to achieve in adolescents than in adults. Nevertheless, the increased use of basal-bolus regimens, insulin pumps, frequent blood glucose monitoring, goal setting, and improved patient education in youth from infancy through adolescence has been associated with more children reaching the blood glucose targets recommended by ADA (67-70), particularly in those families in which both the parents and the child with diabetes participate jointly to perform the required diabetes-related tasks. Furthermore, studies documenting neurocognitive imaging differences related to hyperglycemia in children provide another motivation for lowering glycemic targets (6).
In selecting glycemic targets, the long-term health benefits of achieving a lower A1C should be balanced against the risks of hypoglycemia and the developmental burdens of intensive regimens in children and youth. Inaddition, achieving lower A1C levels is likely facilitated by setting lower A1C targets (51,71). A1C and blood glucose targets are presented in Table 13.1. Lower goals may be possible during the “honeymoon” phase of type 1 diabetes.
Key Concepts in Setting Glycemic Targets
Targets should be individualized, and lower targets may be reasonable based on a benefit-risk assessment.
Blood glucose targets should be modi- fied in children with frequent hypoglycemia or hypoglycemia unawareness.
Postprandial blood glucose values should be measured when there is a discrepancy between preprandial blood glucose values and A1C levels and to assess preprandial insulin doses in those on basal-bolus or pump regimens.
1.8.0.0 Autoimmune Conditions
1.8.1.0 Recommendations
Recommendation
13.22 Assess for additional autoimmune conditions soon after the diagnosis of type 1 diabetes and if symptoms develop. E
Because of the increased frequency of other autoimmune diseases in type 1 diabetes, screening for thyroid dysfunction and celiac disease should be considered (72,73). Periodic screening in asymptomatic individuals has been recommended, but the optimal frequency of screening is unclear.
Although much less common than thyroid dysfunction and celiac disease, other autoimmune conditions, such as Addison disease (primary adrenal insufficiency), autoimmune hepatitis, autoimmune gastritis, dermatomyositis, and myasthenia gravis, occur more commonly in the population with type 1 diabetes than in the general pediatric population and should be assessed and monitored as clinically indicated. In addition, relatives of patients should be offered testing for islet autoantibodies through research studies (e.g., TrialNet) for early diagnosis of preclinical type 1 diabetes (stages 1 and 2).
1.8.2.0 Thyroid Disease
Recommendations
13.23 Consider testing children with type 1 diabetes for antithyroid peroxidase and antithyroglobulin antibodies soon after the diagnosis. B
13.24 Measure thyroid-stimulating hormone concentrations at diagnosis when clinically stable or soon after glycemic control has been established. If normal, suggest rechecking every 1–2 years or sooner if the patient develops symptoms or signs suggestive of thyroid dysfunction, thyromegaly, an abnormal growth rate, or unexplained glycemic variability. E
1.8.3.0 Celiac Disease
Recommendations
13.25 Screen children with type 1 diabetes for celiac disease by measuring IgA tissue transglutaminase (tTG) antibodies, with documentation of normal total serum IgA levels, soon after the diagnosis of diabetes, or IgG to tTG and deamidated gliadin antibodies if IgA deficient. E
13.26 Repeat screening within 2 years of diabetes diagnosis and then again after 5 years and consider more frequent screening in children who have symptoms or a first-degree relative with celiac disease. B
13.27 Individuals with biopsy-confirmed celiac disease should be placed on a gluten-free diet and have a consultation with a dietitian experienced in managing both diabetes and celiac disease. B
Celiac disease is an immune-mediated disorder that occurs with increased frequency in patients with type 1 diabetes (1.6–16.4% of individuals compared with 0.3–1% in the general population) (72,73,80–83).
Screening for celiac disease includes measuring serum levels of IgA and tissue transglutaminase antibodies, or, with IgA deficiency, screening can include measuring IgG tissue transglutaminase antibodies or IgG deamidated gliadin peptide antibodies. Because most cases of celiac disease are diagnosed within the first 5 years after the diagnosis of type 1 diabetes, screening should be considered at the time of diagnosis and repeated at 2 and then 5 years (82) or if clinical symptoms indicate, such as poor growth or increased hypoglycemia (83,84).
Although celiac disease can be diagnosed more than 10 years after diabetes diagnosis, there are insufficient data after 5 years to determine the optimal screening frequency. Measurement of tissue transglutaminase antibody should be considered at other times in patients with symptoms suggestive of celiac disease (82). Monitoring for symptoms should include assessment of linear growth and weight gain (83,84). A small-bowel biopsy in antibody-positive children is recommended to confirm the diagnosis (85). European guidelines on screening for celiac disease in children (not specific to children with type 1 diabetes) suggest that biopsy may not be necessary in symptomatic children with high antibody titers (i.e., greater than 10 times the upper limit of normal) provided that further testing is performed (verification of endomysial antibody positivity on a separate blood sample). Whether this approach may be appropriate for asymptomatic children in high-risk groups remains an open question, though evidence is emerging (86). It is also advisable to check for celiac disease–associated HLA types in patients who are diagnosed without a small intestinal biopsy. In symptomatic children with type 1 diabetes and confirmed celiac disease, gluten-free diets reduce symptoms and rates of hypoglycemia (87). The challenging dietary restrictions associated with having both type 1 diabetes and celiac disease place a significant burden on individuals. Therefore, a biopsy to confirm the diagnosis of celiac disease is recommended, especially in asymptomatic children, before establishing a diagnosis of celiac disease (88) and endorsing significant dietary changes. A gluten-free diet was beneficial in asymptomatic adults with positive antibodies confirmed by biopsy (89).
1.9.0.0 Cardiovascular Risk Factors Mgmt.
1.9.1.0 Hypertension
Recommendations
Screening
13.28 Blood pressure should be measured at each routine visit. Children found to have highnormal blood pressure (systolic blood pressure or diastolic blood pressure$90th percentile for age, sex, and height) or hypertension (systolic blood pressure or diastolic blood pressure $95th percentile for age, sex, and height) should have elevated blood pressure confirmed on 3 separate days. B
Treatment
13.29 Initial treatment of high-normal blood pressure (systolic blood pressure or diastolic blood pressure consistently ≥90th percentile for age, sex, and height) includes dietary modification and increased exercise, if appropriate, aimed at weight control. If target blood pressure is not reached within 3–6 months of initiating lifestyle intervention, pharmacologic treatment should be considered. E
13.30 In addition to lifestyle modifi- cation, pharmacologic treatment of hypertension (systolic blood pressure or diastolic blood pressure consistently ≥95th percentile for age, sex, and height) should be considered as soon as hypertension is confirmed. E
13.31 ACE inhibitors or angiotensin receptor blockers should be considered for the initial pharmacologic treatment of hypertension E in children and adolescents, following reproductive counseling due to the potential teratogenic effects of both drug classes. E
13.32 The goal of treatment is blood pressure consistently <90th percentile for age, sex, and height. E
Blood pressure measurements should be performed using the appropriate size cuff with the child seated and relaxed. Hypertension should be confirmed on at least 3 separate days. Evaluation should proceed as clinically indicated (90). Treatment is generally initiated with an ACE inhibitor, but an angiotensin receptor blocker can be used if the ACE inhibitor is not tolerated (e.g., due to cough) (91).
Normal blood pressure levels for age, sex, and height and appropriate methods for measurement are available online at nhlbi.nih.gov/files/docs/resources/heart/hbp_ped.pdf.
1.9.2.0 Dyslipidemia
Recommendations
Testing13.33 Obtain a fasting lipid profile in children ≥10 years of age soon after the diagnosis of diabetes (after glucose control has been established). E
13.34 If LDL cholesterol values are within the accepted risk level (<100 mg/dL [2.6 mmol/L]), a lipid profile repeated every 3–5 years is reasonable. E
Treatment
13.35 If lipids are abnormal, initial therapy should consist of optimizing glucose control and medical nutrition therapy using a Step 2 American Heart Association diet to decrease the amount of saturated fat to 7% of total calories and dietary cholesterol to 200 mg/day, which is safe and does not interfere with normal growth and development. B
13.36 After the age of 10 years, addition of a statin is suggested in patients who, despite medical nutrition therapy and lifestyle changes, continue to have LDL cholesterol >160 mg/dL (4.1 mmol/L) or LDL cholesterol >130 mg/dL (3.4 mmol/L) and one or more cardiovascular disease risk factor, following reproductive counseling because of the potential teratogenic effects of statins. E
13.37 The goal of therapy is an LDL cholesterol value <100 mg/dL (2.6 mmol/L). E
Population-based studies estimate that 14–45% of children with type 1 diabetes have two or more atherosclerotic cardiovascular disease (ASCVD) risk factors (92-94), and the prevalence of cardiovascular disease (CVD) risk factors increases with age (94) and among racial/ ethnic minorities (21), with girls having a higher risk burden than boys (93).
Pathophysiology.
The atherosclerotic process begins in childhood, and although ASCVD events are not expected to occur during childhood, observations using a variety of methodologies show that youth with type 1 diabetes may have subclinical CVD within the first decade of diagnosis (95-97). Studies of carotid intima-media thickness have yielded inconsistent results (90,91).
Treatment.
Pediatric lipid guidelines provide some guidance relevant to children with type 1 diabetes (90,98–100); however, there are few studies on modifying lipid levels in children with type 1 diabetes. A 6-month trial of dietary counseling produced a significant improvement in lipid levels (101); likewise, a lifestyle intervention trial with 6 months of exercise in adolescents demonstrated improvement in lipid levels (102).
Although intervention data are sparse, the American Heart Association categorizes children with type 1 diabetes in the highest tier for cardiovascular risk and recommends both lifestyle and pharmacologic treatment for those with elevated LDL cholesterol levels (100,103). Initial therapy should be with a nutrition plan that restricts saturated fat to 7% of total calories and dietary cholesterol to 200 mg/day. Data from randomized clinical trials in children as young as 7 months of age indicate that this diet is safe and does not interfere with normal growth and development (104).
For children with a significant family history of CVD, the National Heart, Lung, and Blood Institute recommends obtaining a fasting lipid panel beginning at 2 years of age (98). Abnormal results from a random lipid panel should be confirmed with a fasting lipid panel. Data from the SEARCH for Diabetes in Youth (SEARCH) study show that improved glucose control over a 2-year period is associated with a more favorable lipid profile; however, improved glycemic control alone will not normalize lipids in youth with type 1 diabetes and dyslipidemia (105).
Neither long-term safety nor cardiovascular outcome efficacy of statin therapy has been established for children; however, studies have shown short-term safety equivalent to that seen in adults and efficacy in lowering LDL cholesterol levels in familial hypercholesterolemiaorsevere hyperlipidemia, improving endothelial function and causing regression of carotid intimal thickening (106,107). Statins are not approved for patients aged <10 years, and statin treatment should generally not be used in children with type 1 diabetes before this age. Statins are contraindicated in pregnancy; therefore, prevention of unplanned pregnancies is of paramount importance for postpubertal girls (see Section 14 “Management of Diabetes in Pregnancy” for more information). The multicenter, randomized, placebo-controlled Adolescent Type 1 Diabetes Cardio-Renal Intervention Trial (AdDIT) provides safety data on pharmacologic treatment with an ACE inhibitor and statin in adolescents with type 1 diabetes.
1.9.3.0 Smoking
Recommendations
13.38 Elicit a smoking history at initial and follow-up diabetes visits; discourage smoking in youth who do not smoke, and encourage smoking cessation in those who do smoke. A
13.39 e-Cigarette use should be discouraged. B
The adverse health effects of smoking are well recognized with respect to future cancer and CVD risk. Despite this, smoking rates are significantly higher among youth with diabetes than among youth without diabetes (108,109). In youth with diabetes, it is important to avoid additional CVD risk factors. Smoking increases the risk of onset of albumin-uria; therefore, smoking avoidance is important to prevent both microvascular and macrovascular complications (98, 110). Discouraging cigarette smoking, including e-cigarettes (111,112), is an important part of routine diabetes care. In younger children, it is important to assess exposure to cigarette smoke in the home because of the adverse effects of secondhand smoke and to discourage youth from ever smoking if exposed to smokers in childhood.
1.10.0.0 Microvascular Complications
1.10.1.0 Nephropathy
Recommendations
Screening
13.40 Annual screening for albumin-uria with a random (morning sample preferred to avoid effects of exercise) spot urine sample for albumin-to-creatinine ratio should be considered at puberty or at age >10 years, whichever is earlier, once the child has had diabetes for 5 years. B
Treatment
13.41 An ACE inhibitor or an angiotensin receptor blocker, titrated to normalization of albumin excretion, may be considered when elevated urinary albumin-to-creatinine ratio (>30 mg/g) is documented (two of three urine samples obtained over a 6-month interval following efforts to improve glycemic control and normalize blood pressure). E
Data from 7,549 participants <20 years of age in the T1D Exchange clinic registry emphasize the importance of good glycemic and blood pressure control, particularly as diabetes duration increases, in order to reduce the risk of diabetic kidney disease. The data also underscore the importance of routine screening to ensure early diagnosis and timely treatment of albuminuria (113). An estimation of glomerular filtration rate (GFR), calculated using GFR estimating equations from the serum creatinine, height, age, and sex (114), should be considered at baseline and repeated as indicated based on clinical status, age, diabetes duration, and therapies. Improved methods are needed to screen for early GFR loss, since estimated GFR is inaccurate at GFR >60 mL/min/1.73 m2 (114,115). The AdDIT study in adolescents with type 1 diabetes demonstrated safety of ACE inhibitor treatment, but the treatment did not change the albumin-to-creatinine ratio over the course of the study (90).
1.10.2 Retinopathy
Recommendations
13.42 An initial dilated and comprehensive eye examination is recommended once youth have had type 1 diabetes for 3–5 years, provided they are age ≥10 years or puberty has started, whichever is earlier. B
13.43 After the initial examination, annual routine follow-up is generally recommended. Less-frequent examinations, every 2 years, may be acceptable on the advice of an eye care professional and based on risk factor assessment. E
13.44 Retinopathy (like albuminuria) most commonly occurs after the onset of puberty and after 5–10 years of diabetes duration (116). Referrals should be made to eye care professionals with expertise in diabetic retinopathy and experience in counseling the pediatric patient and family on the importance of prevention, early detection, and intervention.
1.10.3.0 Neuropathy
Recommendation
13.45 Consider an annual comprehensive foot exam at the start of puberty or at age ≥10 years, whichever is earlier, once the youth has had type 1 diabetes for 5 years. B
13.46 Diabetic neuropathy rarely occurs in prepubertal children or after only 1–2 years of diabetes (116), although data suggest a prevalence of distal peripheral neuropathy of 7% in 1,734 youth with type 1 diabetes and associated with the presence of CVD risk factors (117,118). A comprehensive foot exam, including inspection, palpation of dorsalis pedis and posterior tibial pulses, and determination of proprioception, vibration, and monofilament sensation, should be performed annually along with an assessment of symptoms of neuropathic pain (118). Foot inspection can be performed at each visit to educate youth regarding the importance of foot care (see Section 11 “Microvascular Complications and Foot Care”).
2.0.0.0 TYPE 2 DIABETES
2.1.0.0 Introduction
For information on testing for type 2 diabetes and prediabetes in children and adolescents, please refer to Section 2 “Classification and Diagnosis of Diabetes.” For additional support for these recommendations, see the ADA position statement “Evaluation and Management of Youth-Onset Type 2 Diabetes” (2).
Type 2 diabetes in youth has increased over the past 20 years, and recent estimates suggest an incidence of ;5,000 new cases per year in the U.S. (119). The Centers for Disease Control and Prevention published projections for type 2 diabetes prevalence using the SEARCH database; assuming a 2.3% annual increase, the prevalence in those under 20 years of age will quadruple in 40 years (120,121).
Evidence suggests that type 2 diabetes in youth is different not only from type 1 diabetes but also from type 2 diabetes in adults and has unique features, such as a more rapidly progressive decline in β-cell function and accelerated development of diabetes complications (2,122). Type 2 diabetes disproportionately impacts youth of ethnic and racial minorities and can occur in complex psychosocial and cultural environments, which may make it difficult to sustain healthy lifestyle changes and self-management behaviors (22,123–126). Additional risk factors associated with type 2 diabetes in youth include adiposity, family history of diabetes, female sex, and low socioeconomic status (122).
As with type 1 diabetes, youth with type 2 diabetes spend much of the day in school. Therefore, close communication with and the cooperation of school personnel are essential for optimal diabetes management, safety, and maximal academic opportunities.
2.2.0.0 Screening and Diagnosis
Recommendations
13.45 Risk-based screening for prediabetes and/or type 2 diabetes should be considered in children and adolescents after the onset of puberty or ≥10 years of age, whichever occurs earlier, who are overweight (BMI ≥85th percentile) or obese (BMI ≥95th percentile) and who have one or more additional risk factors for diabetes (see Table 2.4 for evidence grading of other risk factors).
13.46 If tests are normal, repeat testing at a minimum of 3-year intervals E, or more frequently if BMI is increasing. C
13.47 Fasting plasma glucose, 2-h plasma glucose during a 75-g oral glucose tolerance test, and A1C can be used to test for prediabetes or diabetes in children and adolescents. B
13.48 Children and adolescents with overweight/obesity in whom the diagnosis of type 2 diabetes is being considered should have a panel of pancreatic autoantibodies tested to exclude the possibility of autoimmune type 1 diabetes. B
In the last decade, the incidence and prevalence of type 2 diabetes in adolescents has increased dramatically, especially in racial and ethnic minority populations (98,127). A few recent studies suggest oral glucose tolerance tests or fasting plasma glucose values as more suitable diagnostic tests than A1C in the pediatric population, especially among certain ethnicities (128), although fasting glucose alone may overdiagnose diabetes in children (129,130). In addition, many of these studies do not recognize that diabetes diagnostic criteria are based on long-term health outcomes, and validations are not currently available in the pediatric population (131). ADA acknowledges the limited data supporting A1C for diagnosing type 2 diabetes in children and adolescents. Although A1C is not recommended for diagnosis of diabetes in children with cystic fibrosis or symptoms suggestive of acute onset of type 1 diabetes, and only A1C assays without interference are appropriate for children with hemoglobinopathies, ADA continues to recommend A1C for diagnosis of type 2 diabetes in this population (132,133).
2.3.0.0 Diagnostic Challenges
Given the current obesity epidemic, distinguishing between type 1 and type 2 diabetes in children can be difficult. Overweight and obesity are common in children with type 1 diabetes (23), and diabetes-associated autoantibodies and ketosis may be present in pediatric patients with features of type 2 diabetes (including obesity and acanthosis nigricans) (129). The presence of islet auto-antibodies has been associated with faster progression to insulin deficiency (129). At onset, DKA occurs in ˜6% of youth aged 10–19 years with type 2 diabetes (134). Although uncommon, type 2 diabetes has been observed in prepubertal children under the age of 10, and thus it should be part of the differential in children with suggestive symptoms (135). Finally, obesity (136) and type 2 diabetes–associated genetic factors may (137) contribute to the development of type 1 diabetes in some individuals, which further blurs the lines between diabetes types. However, accurate diagnosis is critical, as treatment regimens, educational approaches, dietary advice, and outcomes differ markedly between patients with the two diagnoses.
2.4.0.0 Management
2.4.1.0 Recommendations
Recommendations
Lifestyle Management
13.49 All youth with type 2 diabetes and their families should receive comprehensive diabetes self-management education and support that is specific to youth with type 2 diabetes and is culturally competent. B
13.50 Youth with overweight/obesity and type 2 diabetes and their families should be provided with developmentally and culturally appropriate comprehensive lifestyle programs that are integrated with diabetes management to achieve 7–10% decrease in excess weight. C
13.51 Given the necessity of long-term weight management for children and adolescents with type 2 diabetes, lifestyle intervention should be based on a chronic care model and offered in the context of diabetes care. E
13.52 Youth with diabetes, like all children, should be encouraged to participate in at least 30–60 min of moderate to vigorous physical activity at least 5 days per week (and strength training on at least 3 days/week) B and to decrease sedentary behavior. C
13.53 Nutrition for youth with type 2 diabetes, like all children, should focus on healthy eating patterns that emphasize consumption of nutrient-dense, high-quality foods and decreased consumption of calorie-dense, nutrient-poor foods, particularly sugar-added beverages. B
Glycemic Targets
13.54 Home self-monitoring of blood glucose regimens should be individualized, taking into consideration the pharmacologic treatment of the patient. E
13.55 A1C should be measured every 3 months. E
13.56 A reasonable A1C target for most children and adolescents with type 2 diabetes treated with oral agents alone is <7% (53 mmol/mol). More stringent A1C targets (such as <6.5% [48 mmol/mol]) may be appropriate for selected individual patients if this can be achieved without significant hypoglycemia or other adverse effects of treatment. Appropriate patients might include those with short duration of diabetes and lesser degrees of β-cell dysfunction and patients treated with lifestyle or metformin only who achieve signifi- cant weight improvement. E
13.57 A1C targets for patients on insulin should be individualized, taking into account the relatively low rates of hypoglycemia in youth-onset type 2 diabetes. E
Pharmacologic Management
13.58 Initiate pharmacologic therapy, in addition to lifestyle therapy, at diagnosis of type 2 diabetes. A
13.59 In incidentally diagnosed or metabolically stable patients (A1C <8.5% [69 mmol/mol] and asymptomatic), metformin is the initial pharmacologic treatment of choice if renal function is normal. A
13.60 Youth with marked hyperglycemia (blood glucose ≥250 mg/dL [13.9 mmol/L], A1C ≥8.5% [69 mmol/mol]) without acidosis at diagnosis who are symptomatic with polyuria, polydipsia, nocturia, and/or weight loss should be treated initially with basal insulin while metformin is initiated and titrated. B
13.61 In patients with ketosis/ ketoacidosis, treatment with subcutaneous or intravenous insulin should be initiated to rapidly correct the hyperglycemia and the metabolic derangement. Once acidosis is resolved, metformin should be initiated while subcutaneous insulin therapy is continued. A
13.62 In individuals presenting with severe hyperglycemia (blood glucose ≥600 mg/dL [33.3 mmol/L]), consider assessment for hyperglycemic hyperosmolar nonketotic syndrome. A
13.63 If the A1C target is no longer met with metformin monotherapy, or if contraindications or intolerable side effects of metformin develop, basal insulin therapy should be initiated. B
13.64 Patients treated with basal insulin up to 1.5 units/kg/day who do not meet A1C target should be moved to multiple daily injections with basal and premeal bolus insulins. E
13.65 In patients initially treated with insulin and metformin who are meeting glucose targets based on home blood glucose monitoring, insulin can be tapered over 2–6 weeks by decreasing the insulin dose 10–30% every few days. B
13.66 Use of medications not approved by the U.S. Food and Drug Administration for youth with type 2 diabetes is not recommended outside of research trials. B
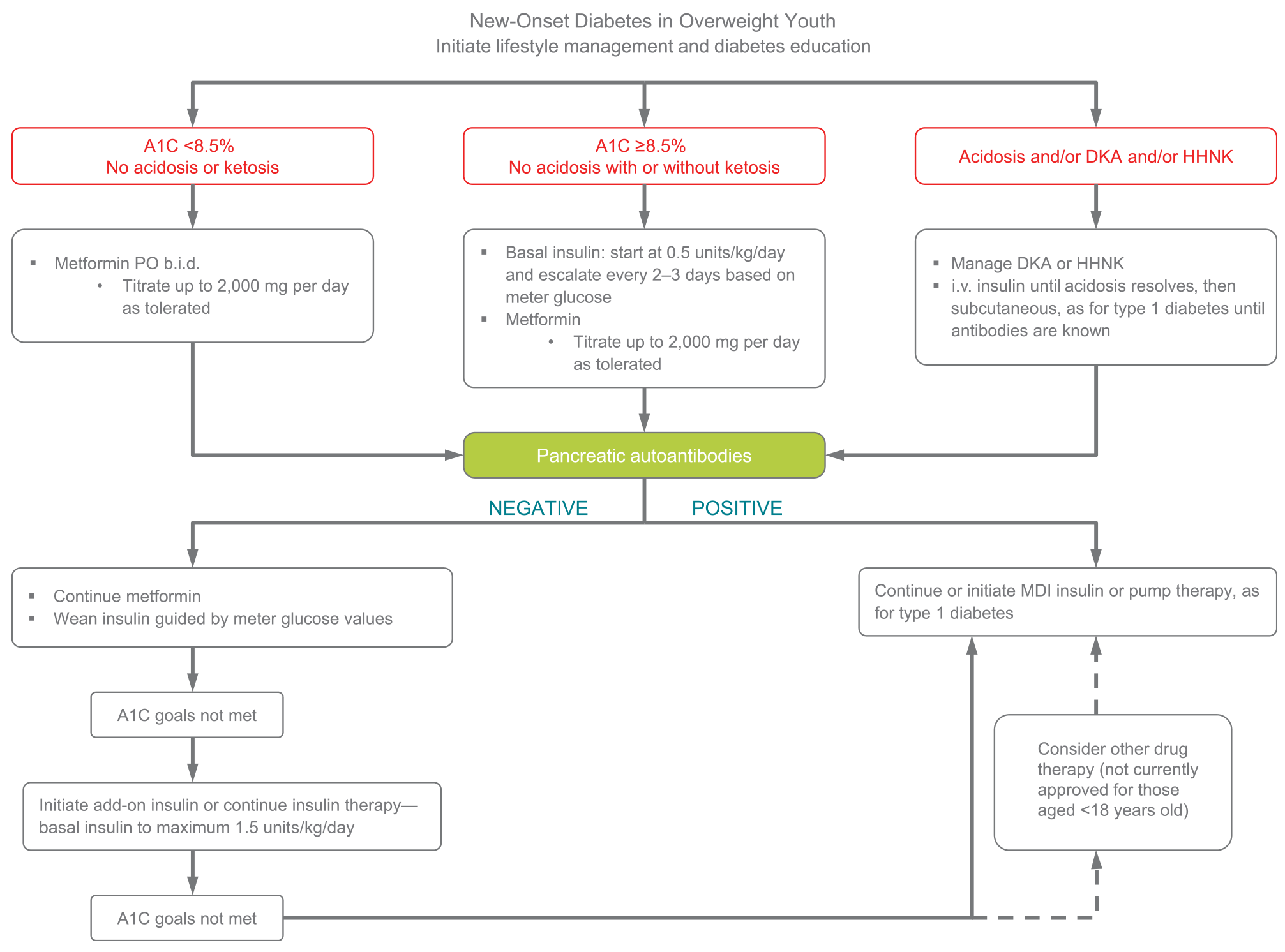
Figure 13.1—Management of new-onset diabetes in overweight youth (2). A1C 8.5% 5 69 mmol/mol. DKA, diabetic ketoacidosis; HHNK, hyperosmolar hyperglycemic nonketotic syndrome; MDI, multiple daily injections.
2.4.2 Overview
Treatment of youth-onset type 2 diabetes should include lifestyle management, diabetes self-management education, and pharmacologic treatment. Initial treatment of youth with obesity and diabetes must take into account that diabetes type is often uncertain in the first few weeks of treatment, due to overlap in presentation, and that a substantial percentage of youth with type 2 diabetes will present with clinically significant ketoacidosis (138). Therefore, initial therapy should address the hyperglycemia and associated metabolic derangements irrespective of ultimate diabetes type, with adjustment of therapy once metabolic compensation has been established and subsequent information, such as islet autoantibody results, becomes available. Figure 13.1 provides an approach to initial treatment of new-onset diabetes in overweight youth.
Glycemic targets should be individualized, taking into consideration long-term health benefits of more stringent targets as well as risk for adverse effects, such as hypoglycemia. A lower target A1C in youth with type 2 diabetes when compared with those recommended in type 1 diabetes is justified by lower risk of hypoglycemia and higher risk of complications (139-142).
Patients and their families must prioritize lifestyle modifications such as eating a balanced diet, achieving and maintaining a healthy weight, and exercising regularly. A family-centered approach to nutrition and lifestyle modi- fication is essential in children with type 2 diabetes, and nutition recommendations should be culturally appropriate and sensitive to family resources (see Section “Lifestyle Management”). Given the complex social and environmental context surrounding youth with type 2 diabetes, individual-level lifestyle interventions may not be sufficient to target the complex interplay of family dynamics, mental health, community readiness, and the broader environmental system (2).
A multidisciplinary diabetes team, including a physician, diabetes nurse educator, registered dietitian, and psychologist or social worker, is essential. In addition to blood glucose control and self-management education (143-145), initial treatment must include management of comorbidities such as obesity, dyslipidemia, hypertension, and microvascular complications.
Current pharmacologic treatment options for youth-onset type 2 diabetes are limited to two approved drugsdinsulin and metformin (2). Presentation with ketoacidosis or marked ketosis requires a period of insulin therapy until fasting and postprandial glycemia have been restored to normal or near-normal levels. Metformin therapy may be used as an adjunct after resolution of ketosis/ ketoacidosis. Initial treatment should also be with insulin when the distinction between type 1 diabetes and type 2 diabetes is unclear and in patients who have random blood glucose concentrations ≥250 mg/dL (13.9 mmol/L) and/or A1C ≥8.5% (69 mmol/mol) (146). Insulin is needed when the glycemic target is not met on metformin alone, or if there is metformin intolerance or renal or hepatic insufficiency (147).
When insulin treatment is not required, initiation of metformin is recommended. The Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) study found that metformin alone provided durable glycemic control (A1C ≤8% [64 mmol/mol] for 6 months) in approximately half of the subjects (148). To date, the TODAY study is the only trial combining lifestyle and metformin therapy in youth with type 2 diabetes; the combination did not perform better than metformin alone in achieving durable glycemic control (148).
2.4.3.0 Metabolic Surgery
Recommendations
13.67 Metabolic surgery may be considered for the treatment of adolescents with type 2 diabetes who are markedly obese (BMI >35 kg/m2) and who have uncontrolled glycemia and/or serious comorbidities despite lifestyle and pharmacologic intervention. A
13.68 Metabolic surgery should be performed only by an experienced surgeon working as part of a well-organized and engaged multidisciplinary team including surgeon, endocrinologist, nutritionist, behavioral health specialist, and nurse. A
The results of weight-loss and lifestyle interventions for obesity in children and adolescents have been disappointing, and no effective and safe pharmacologic intervention is available or approved by the U.S. Food and Drug Administration in youth. Over the last decade, weight-loss surgery has been increasingly performed in adolescents with obesity. Small retrospective analyses and a recent prospective multicenter nonrandomized study suggest that bariatric or metabolic surgery may have benefits in obese adolescents with type 2 diabetes similar to those observed in adults. Teenagers experience similar degrees of weight loss, diabetes remission, and improvement of cardiometabolic risk factors for at least 3 years after surgery (149). No randomized trials, however, have yet compared the effectiveness and safety of surgery to those of conventional treatment options in adolescents (150). The guidelines used as an indication for metabolic surgery in adolescents generally include BMI >35 kg/m2 with comorbidities or BMI >40 kg/m2 with or without comorbidities (151-162). A number of groups, including the Pediatric Bariatric Study Group and the Teen Longitudinal Assessment of Bariatric Surgery (Teen-LABS) Study have demonstrated the effectiveness of metabolic surgery in adolescents (155-161).
2.5.0.0 Prevention and Management of Diabetes Complications
Recommendations
Nephropathy
13.69 Blood pressure should be measured at every visit. A
13.70 Blood pressure should be optimized to reduce risk and/or slow the progression of diabetic kidney disease. A
13.71 If blood pressure is >95th percentile for age, sex, and height, increased emphasis should be placed on lifestyle management to promote weight loss. If blood pressure remains above the 95th percentile after 6 months, antihypertensive therapy should be initiated. C
13.72 Initial therapeutic options include ACE inhibitors or angiotensin receptor blockers. Other blood pressure–lowering agents may be added as needed. C
13.73 Protein intake should be at the recommended daily allowance of 0.8 g/kg/day. E
13.75 Urine albumin-to-creatinine ratio should be obtained at the time of diagnosis and annually thereafter. An elevated urine albumin-to-creatinine ratio (>30 mg/g creatinine) should be confirmed on two of three samples. B
13.76 Estimated glomerular filtration rate should be determined at the time of diagnosis and annually thereafter. E
13.77 In nonpregnant patients with diabetes and hypertension, either an ACE inhibitor or an angiotensin receptor blocker is recommended for those with modestly elevated urinary albumin-to-creatinine ratio (30–299 mg/g creatinine) D and is strongly recommended for those with urinary albumin-to-creatinine ratio >300 mg/g creatinine and/or estimated glomerular filtration rate <60 mL/min/1.73 m2. E
13.78 For those with nephropathy, continued monitoring (yearly urinary albumin-to-creatinine ratio, estimated glomerular filtration rate, and serum potassium) may aid in assessing adherence and detecting progression of disease. E
13.79 Referral to nephrology is recommended in case of uncertainty of etiology, worsening urinary albumin-to-creatinine ratio, or decrease in estimated glomerular filtration rate. E
Neuropathy
13.79 Youth with type 2 diabetes should be screened for the presence of neuropathy by foot examination at diagnosis and annually. The examination should include inspection, assessment of foot pulses, pinprick and 10-g monofilament sensation tests, testing of vibration sensation using 128-Hz tuning fork, and ankle reflexes. C
13.80 Prevention should focus on achieving glycemic targets. C
Retinopathy
13.81 Screening for retinopathy should be performed by dilated fundoscopy or retinal photography at or soon after diagnosis and annually thereafter. C
13.82 Optimizing glycemia is recommended to decrease the risk or slow the progression of retinopathy. B
13.83 Less frequent examination (every 2 years) may be considered if there is adequate glycemic control and a normal eye exam. C
Nonalcoholic Fatty Liver Disease
13.84 Evaluation for nonalcoholic fatty liver disease (by measuring aspartate aminotransferase and alanine aminotransferase) should be done at diagnosis and annually thereafter. B
13.85 Referral to gastroenterology should be considered for persistently elevated or worsening transaminases. B
Obstructive Sleep Apnea
13.86 Screening for symptoms of sleep apnea should be done at each visit, and referral to a pediatric sleep specialist for evaluation and a polysomnogram, if indicated, is recommended. Obstructive sleep apnea should be treated when documented. B
Polycystic Ovary Syndrome
13.87 Evaluate for polycystic ovary syndrome in female adolescents with type 2 diabetes, including laboratory studies when indicated. B
Oral contraceptive pills for treatment of polycystic ovary syndrome are not contraindicated for girls with type 2 diabetes. C
13.88 Metformin in addition to lifestyle modification is likely to improve the menstrual cyclicity and hyperandrogenism in girls with type 2 diabetes. E
Cardiovascular Disease
13.89 Intensive lifestyle interventions focusing on weight loss, dyslipidemia, hypertension, and dysglycemia are important to prevent overt macrovascular disease in early adulthood. E
Dyslipidemia
13.90 Lipid testing should be performed when initial glycemic control has been achieved and annually thereafter. B
13.91 Optimal goals are LDL cholesterol <100 mg/dL (2.6 mmol/L), HDL cholesterol >35 mg/dL (0.905 mmol/L), and triglycerides <150 mg/dL (1.7 mmol/L). E
13.92 If LDL cholesterol is >130 mg/dL, blood glucose control should be maximized and dietary counseling should be provided using the American Heart Association Step 2 diet. E
13.93 If LDL cholesterol remains above goal after 6 months of dietary intervention, initiate therapy with statin, with goal of LDL <100 mg/dL. B
13.94 If triglycerides are >400 mg/dL (4.7 mmol/L) fasting or >1,000 mg/dL (11.6 mmol/L) nonfasting, optimize glycemia and begin fibrate, with a goal of <400 mg/dL (4.7 mmol/L) fasting (to reduce risk for pancreatitis). C
Cardiac Function Testing
13.95 Routine screening for heart disease with electrocardiogram, echocardiogram, or stress testing is not recommended in asymptomatic youth with type 2 diabetes. B
Comorbidities may already be present at the time of diagnosis of type 2 diabetes in youth (122,163). Therefore, blood pressure measurement, a fasting lipid panel, assessment of random urine albumin-to-creatinine ratio, and a dilated eye examination should be performed at diagnosis. Thereafter, screening guidelines and treatment recommendations for hypertension, dyslipidemia, urine albumin excretion, and retinopathy are similar to those for youth with type 1 diabetes. Additional problems that may need to be addressed include polycystic ovary disease and other comorbidities associated with pediatric obesity, such as sleep apnea, hepatic steatosis, orthopedic complications, and psychosocial concerns. The ADA position statement “Evaluation and Management of Youth-Onset Type 2 Diabetes” (2) provides guidance on the prevention, screening, and treatment of type 2 diabetes and its comorbidities in children and adolescents.
Youth-onset type 2 diabetes is associated with significant microvascular and macrovascular risk burden and a substantial increase in the risk of cardiovascular morbidity and mortality at an earlier age than those diagnosed later in life (164). The higher complication risk in earlier-onset type 2 diabetes is likely related to prolonged lifetime exposure to hyperglycemia and other atherogenic risk factors, including insulin resistance, dyslipidemia, hypertension, and chronic inflammation. There is low risk of hypoglycemia in youth with type 2 diabetes, even if they are being treated with insulin (165), and there are high rates of complications (139-142). These diabetes comorbidities also appear to be higher than in youth with type 1 diabetes despite shorter diabetes duration and lower A1C (163). In addition, the progression of vascular abnormalities appears to be more pronounced in youth-onset type 2 diabetes compared with type 1 diabetes of similar duration, including ischemic heart disease and stroke (166).
2.6.0.0 Psychosocial Factors
Recommendations
13.97 Providers should assess social context, including potential food insecurity, housing stability, and financial barriers, and apply that information to treatment decisions. E
13.98 Use patient-appropriate standardized and validated tools to assess for diabetes distress and mental/behavioral health in youth with type 2 diabetes, with attention to symptoms of depression and eating disorders, and refer to specialty care when indicated. B
13.99 When choosing glucose-lowering or other medications for youth with overweight/ obesity and type 2 diabetes, consider medication-taking behavior and their effect on weight. E
13.100 Starting at puberty, preconception counseling should be incorporated into routine diabetes clinic visits for all females of childbearing potential because of the adverse pregnancy outcomes in this population. A
13.101 Patients should be screened for smoking and alcohol use at diagnosis and regularly thereafter. C
Most youth with type 2 diabetes come from racial/ethnic minority groups, have low socioeconomic status, and often experience multiple psychosocial stressors (22,35,123–126). Consideration of the sociocultural context and efforts to personalize diabetes management are of critical importance to minimize barriers to care, enhance adherence, and maximize response to treatment.
Evidence about psychiatric disorders and symptoms in youth with type 2 diabetes is limited (167-171), but given the sociocultural context for many youth and the medical burden and obesity associated with type 2 diabetes, ongoing surveillance of mental health/behavioral health is indicated. Symptoms of depression and disordered eating are common and associated with poorer glycemic control (168,172,173).
Many of the drugs prescribed for diabetes and psychiatric disorders are associated with weight gain and can increase patients’ concerns about eating, body shape, and weight (174,175). The TODAY study documented (176) that despite disease- and age-specific counseling, 10.2% of the females in the cohort became pregnant over an average of 3.8 years of study participation. Of note, 26.4% of pregnancies ended in a miscarriage, stillbirth, or intrauterine death, and 20.5% of the live-born infants had a major congenital anomaly.
3.0.0.0 TRANSITION FROM PEDIATRIC TO ADULT CARE
Recommendations
13.102 Pediatric diabetes providers should begin to prepare youth for transition to adult health care in early adolescence and, at the latest, at least 1 year before the transition. E
13.103 Both pediatric and adult diabetes care providers should provide support and resources for transitioning young adults. E
13.104 Youth with type 2 diabetes should be transferred to an adult-oriented diabetes specialist when deemed appropriate by the patient and provider. E
Care and close supervision of diabetes management are increasingly shifted from parents and other adults to the youth with type 1 or type 2 diabetes throughout childhood and adolescence. The shift from pediatric to adult health care providers, however, often occurs abruptly as the older teen enters the next developmental stage, referred to as emerging adulthood (177), which is a critical period for young people who have diabetes. During this period of major life transitions, youth begin to move out of their parents’ homes and must become fully responsible for their diabetes care. Their new responsibilities include self-management of their diabetes, making medical appointments, and financing health care, once they are no longer covered by their parents’ health insurance plans (ongoing coverage until age 26 years is currently available under provisions of the U.S. Affordable Care Act). In addition to lapses in health care, this is also a period associated with deterioration in glycemic control; increased occurrence of acute complications; psychosocial, emotional, and behavioral challenges; and the emergence of chronic complications (178-181). The transition period from pediatric to adult care is prone to fragmentation in health care delivery, which may adversely impact health care quality, cost, and outcomes (182).
Although scientific evidence is limited, it is clear that comprehensive and coordinated planning that begins in early adolescence is necessary to facilitate a seamless transition from pediatric to adult health care (178,179,183,184). A comprehensive discussion regarding the challenges faced during this period, including specific recommendations, is found in the ADA position statement “Diabetes Care for Emerging Adults: Recommendations for Transition From Pediatric to Adult Diabetes Care Systems” (179).
The Endocrine Society in collaboration with the ADA and other organizations has developed transition tools for clinicians and youth and families (184).
4.0.0.0 References
White NH, Zeitler P. Evaluation and management of youth-onset type 2 diabetes: a position
statementby the American Diabetes Association. Diabetes Care 2018;41:2648–2668
new carbohydrate intake algorithm: an observational field study. Diabetes Technol Ther 2011; 13:819–825
[published correction appears in Diabetes Care 2018;41:640]. Diabetes Care 2017;40:
1002–1009
Darcan S¸, Go¨ks¸en D. Reliability and validity of the diabeteseating problemsurvey in Turkish children and adolescents with type 1 diabetes mellitus. J Clin Res Pediatr Endocrinol 2017;9:323–328
Betts VT, et al. Rethinking funding priorities in mental health research. Br J Psychiatry 2016;208: 507–509
baseline data from the TODAY study. Diabetes Care 2011;34:858–860