5.5.0.0 Antihyperglycemic Therapies and Cardiovascular Outcomes
In 2008, the FDA issued a guidance for industry to perform cardiovascular outcomes trials for all new medications for the treatment for type 2 diabetes amid concerns of increased cardiovascular risk (157). Previously approved diabetes medications were not subject to the guidance. Recently published cardiovascular outcomes trials have provided additional data on cardiovascular outcomes in patients with type 2 diabetes with cardiovascular disease or at high risk for cardiovascular disease (see Table 10.4). Cardiovascular outcomes trials of dipeptidyl peptidase 4 (DPP-4) inhibitors have all, so far, not shown cardiovascular benefits relative to placebo. However, results from other new agents have provided a mix of results.
The BI 10773 (Empagliflozin) Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients (EMPA-REG OUTCOME) trial was a randomized, double-blind trial that assessed the effect of empagliflozin, an SGLT2 inhibitor, versus placebo on cardiovascular outcomes in 7,020 patients with type 2 diabetes and existing cardiovascular disease. Study participants had a mean age of 63 years, 57% had diabetes for more than 10 years, and 99% had established cardiovascular disease. EMPA-REG OUTCOME showed that over a median follow-up of 3.1 years, treatment reduced the composite outcome of MI, stroke, and cardiovascular death by 14% (absolute rate 10.5% vs. 12.1% in the placebo group, HR in the empagliflozin group 0.86; 95% CI 0.74–0.99; P = 0.04 for superiority) and cardiovascular death by 38% (absolute rate 3.7% vs. 5.9%, HR 0.62; 95% CI 0.49–0.77; P < 0.001) (8). The FDA added an indication for empagliflozin to reduce the risk of major adverse cardiovascular death in adults with type 2 diabetes and cardiovascular disease.
A second large cardiovascular outcomes trial program of an SGLT2 inhibitor, canagliflozin, has been reported (9). The Canagliflozin Cardiovascular Assessment Study (CANVAS) integrated data from two trials, including the CANVAS trial that started in 2009 before the approval of canagliflozin and the CANVAS-Renal (CANVAS-R) trial that started in 2014 after the approval of canagliflozin. Combining both these trials, 10,142 participants with type 2 diabetes (two-thirds with established CVD) were randomized to canagliflozin or placebo and were followed for an average 3.6 years. The mean age of patients was 63 years and 66% had a history of cardiovascular disease. The combined analysis of the two trials found that canagliflozin significantly reduced the composite outcome of cardiovascular death, MI, or stroke versus placebo (occurring in 26.9 vs. 31.5 participants per 1,000 patient-years; HR 0.86 [95% CI 0.75–0.97]; P < 0.001 for noninferiority; P = 0.02 for superiority). The specific estimates for canagliflozin versus placebo on the primary composite cardiovascular outcome were HR 0.88 (0.75–1.03) for the CANVAS trial and 0.82 (0.66–1.01) for CANVAS-R, with no heterogeneity found between trials. In the combined analysis, there was not a statistically significant difference in cardiovascular death (HR 0.87 [95% CI 0.72–1.06]). The initial CANVAS trial was partially unblinded prior to completion because of the need to file interim cardiovascular outcomes data for regulatory approval of the drug (158). Of note, there was an increased risk of lower-limb amputation with canaglifozin (6.3 vs. 3.4 participants per 1,000 patient-years; HR 1.97 [95% CI 1.41–2.75]) (9).
The Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results (LEADER) trial was a randomized, double-blind trial that assessedtheeffectof liraglutide, a glucagonlike peptide 1 (GLP-1) receptor agonist, versus placebo on cardiovascular outcomes in 9,340 patients with type 2 diabetes at high risk for cardiovascular disease or with cardiovascular disease. Study participants had a mean age of 64 years and a mean duration of diabetes of nearly 13 years. Over 80% of study participants had established cardiovascular disease. After a median follow-up of 3.8 years, LEADER showed that the primary composite outcome (MI, stroke, or cardiovascular death) occurred in fewer participants in the treatment group (13.0%) when compared with the placebo group (14.9%) (HR 0.87; 95% CI 0.78–0.97; P < 0.001 for non-inferiority; P = 0.01 for superiority). Deaths from cardiovascular causes were significantly reduced in the liraglutide group (4.7%) compared with the placebo group (6.0%) (HR 0.78; 95% CI 0.66–0.93; P = 0.007) (159). The FDA approved the use of liraglutide to reduce the risk of major adverse cardiovascular events, including heart attack, stroke, and cardiovascular death, in adults with type 2 diabetes and established cardiovascular disease.
Results from a moderate-sized trial of another GLP-1 receptor agonist, semaglutide, were consistent with the LEADER trial (160). Semaglutide is a once-weekly GLP-1 receptor agonist approved by the FDA for the treatment of type 2 diabetes. The Trial to Evaluate Cardiovascular and Other Long-term Outcomes With Semaglutide in Subjects With Type 2 Diabetes (SUSTAIN-6) was the initial randomized trial powered to test noninferiority of semaglutide for the purpose of initial regulatory approval. In this study, 3,297 patients with type 2 diabetes were randomized to receive once-weekly semaglutide (0.5 mg or 1.0 mg) or placebo for 2 years. The primary outcome (the first occurrence of cardiovascular death, non-fatal MI, or nonfatal stroke) occurred in 108 patients (6.6%) in the semaglutide group vs. 146 patients (8.9%) in the placebo group (HR 0.74 [95% CI 0.58–0.95]; P < 0.001). More patients discontinued treatment in the semaglutide group because of adverse events, mainly gastrointestinal.
The Evaluation of Lixisenatide in Acute Coronary Syndrome (ELIXA) trial studied the once-daily GLP-1 receptor agonist lixisenatide on cardiovascular outcomes in patients with type 2 diabetes who had had a recent acute coronary event (161). A total of 6,068 patients with type 2 diabetes with a recent hospitalization for MI or unstable angina within the previous 180 days were randomized to receive lixisenatide or placebo in addition to standard care and were followed for a median of approximately 2.1 years. The primary outcome of cardiovascular death, MI, stroke, or hospitalization for unstable angina occurred in 406 patients (13.4%) in the lixisenatide group vs. 399 (13.2%) in the placebo group (HR 1.2 [95% CI 0.89–1.17]), which demonstrated the noninferiority of lixisenatide to placebo (P < 0.001) but did not show superiority (P = 0.81).
The Exenatide Study of Cardiovascular Event Lowering (EXSCEL) trial also reported results with the once-weekly GLP-1 receptor agonist extended-release exenatide and found that major adverse cardiovascular events were numerically lower with use of extended-release exenatide compared with placebo, although this difference was not statistically significant(162). A total of 14,752 patients with type 2 diabetes (of whom 10,782 [73.1%] had previous cardiovascular disease) were randomized to receive extended-release exenatide 2 mg or placebo and followed for a median of 3.2 years. The primary end point of cardiovascular death, MI, or stroke occurred in 839 patients (11.4%; 3.7 events per 100 person-years) in the exenatide group and in 905 patients (12.2%; 4.0 events per 100 person-years) in the placebo group (HR 0.91 [95% CI 0.83– 1.00]; P <0.001 for noninferiority) but was not superior to placebo with respect to the primary end point (P = 0.06 for superiority). However, all-cause mortality was lower in the exenatide group (HR 0.86 [95% CI 0.77–0.97]. The incidence of acute pancreatitis, pancreatic cancer, medullary thyroid carcinoma, and serious adverse events did not differ significantly between the two groups.
The Harmony Outcomes trial randomized 9,463 patients with type 2 diabetes and cardiovascular disease to once-weekly subcutaneous albiglutide or matching placebo, in addition to their standard care. Over a median duration of 1.6 years, the GLP-1 receptor agonist reduced the risk of cardiovascular death, MI, or stroke to an incidence rate of 4.6 events per 100 person-years in the albiglutide group vs. 5.9 events in the placebo group (HR ratio 0.78, P = 0.0006 for superiority) (163). This agent is not currently available for clinical use.
In summary, there are now several large randomized controlled trials reporting statistically significant reductions in cardiovascular events for two of the FDA- approved SGLT2 inhibitors (empagliflozin and canagliflozin) and three FDA-approved GLP-1 receptor agonists (liraglutide, albiglutide [although that agent was removed from the market for business reasons], and semaglutide [lower risk of cardiovascular events in a moderate-sized clinical trial but one not powered as a cardiovascular outcomes trial]). In these trials, the majority, if not all, patients in the trial had ASCVD. The empagliflozin and liraglutide trials further demonstrated significant reductions in cardiovascular death. Once-weekly exenatide did not have statistically significant reductions in major adverse cardiovascular events or cardiovascular mortality but did have a significant reduction in all-cause mortality. In contrast, other GLP-1 receptor agonists have not shown similar reductions in cardiovascular events (Table 10.4). Additional large randomized trials of other agents in these classes are ongoing.
Of note, these studies examined the drugs in combination with metformin (Table 10.4) in the great majority of patients for whom metformin was not contraindicated or was tolerated. For patients with type 2 diabetes who have ASCVD, on lifestyle and metformin therapy, it is recommended to incorporate an agent with strong evidence for cardiovascular risk reduction, especially those with proven reduction of cardiovascular death, after consideration of drug-specific patient factors (Table 9.1). See Fig. 9.1 for additional recommendations on antihyperglycemic treatment in adults with type 2 diabetes.
Table 10.4—Cardiovascular outcomes trials of available antihyperglycemic medications completed after the issuance of the FDA 2008 guidelines
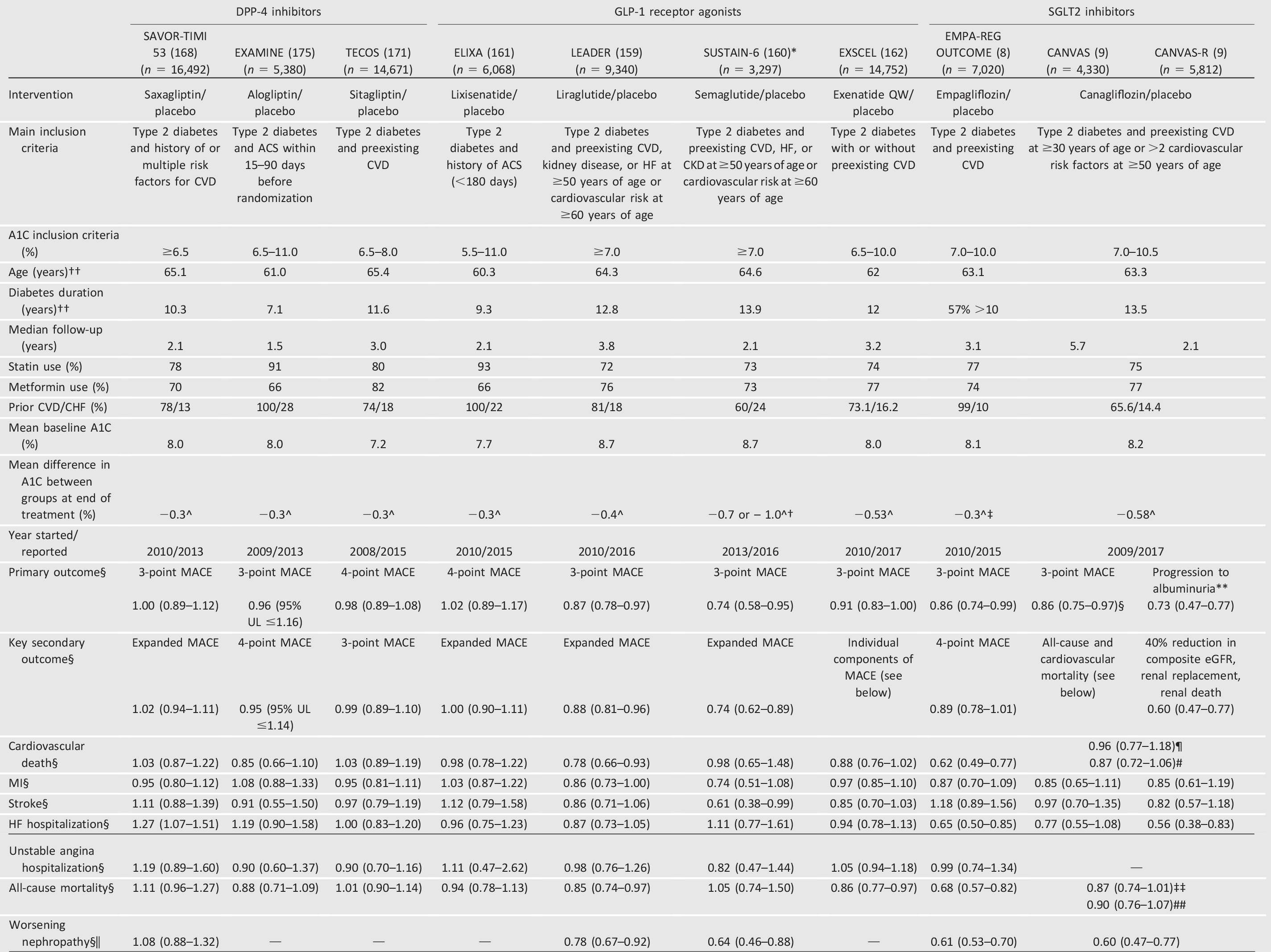
—, not assessed/reported; ACS, acute coronary syndrome; CHF, congestive heart failure; CKD, chronic kidney disease; CVD, cardiovascular disease; DPP-4, dipeptidyl peptidase 4; eGFR, estimated glomerular filtration rate; GLP-1, glucagon-like peptide 1; HF, heart failure; MACE, major adverse cardiac event; MI, myocardial infarction; SGLT2, sodium–glucose cotransporter 2; UL, upper limit. Data from this table was adapted from Cefalu et al. (176) in the January 2018 issue of Diabetes Care.
*Powered to rule out a hazard ratio of 1.8; superiority hypothesis not prespecified.
**On the basis of prespecified outcomes, the renal outcomes are not viewed as statistically significant.
††Age was reported as means in all trials except EXAMINE, which reported medians; diabetes duration was reported as means in all but four trials, with SAVOR- TIMI 58, EXAMINE, and EXSCEL reporting medians and EMPA-REG OUTCOME reporting as percentage of population with diabetes duration >10 years. †A1C change of 0.66% with 0.5 mg and 1.05% with 1 mg dose of semaglutide.
‡AlC change of 0.30 in EMPA-REG OUTCOME is based on pooled results for both doses (i.e., 0.24% for 10 mg and 0.36% for 25 mg of empagliflozin). §Outcomes reported as hazard ratio (95% CI).
||Worsening nephropathy is defined as the new onset of urine albumin-to-creatinine ratio >300 mg/g creatinine or a doubling of the serum creatinine level and an estimated glomerular filtration rate of <45 mL/min/1.73 m2, the need for continuous renal-replacement therapy, or death from renal disease in EMPA-REG OUTCOME, LEADER, and SUSTAIN-6 and as doubling of creatinine level, initiation of dialysis, renal transplantation, or creatinine >6.0 mg/dL (530 mmol/L) in SAVOR-TIMI 53. Worsening nephropathy was a prespecified exploratory adjudicated outcome in SAVOR-TIMI 53, LEADER, and SUSTAIN-6 but not in EMPA-REG OUTCOME.
¶Truncated data set (prespecified in treating hierarchy as the principal data set for analysis for superiority of all-cause mortality and cardiovascular death in the CANVAS Program).
^Significant difference in A1C between groups (P <0.05).
#Nontruncated data set.
‡‡Truncated integrated data set (refers to pooled data from CANVAS after 20 November 2012 plus CANVAS-R; prespecified in treating hierarchy as the principal data set for analysis for superiority of all-cause mortality and cardiovascular death in the CANVAS Program).
##Nontruncated integrated data (refers to pooled data from CANVAS, including before 20 November 2012 plus CANVAS-R).
