4.2.0.0 Initial Therapy
Metformin should be started at the time type 2 diabetes is diagnosed unless there are contraindications; for most patients this will be monotherapy in combination with lifestyle modifications. Metformin is effective and safe, is inexpensive, and may reduce risk of cardiovascular events and death (40). Metformin is available in an immediate-release form for twicedaily dosing or as an extended-release form that can be given once daily. Compared with sulfonylureas, metformin as first-line therapy has beneficial effects on A1C, weight, and cardiovascular mortality (41); there is little systematic data available for other oral agents as initial therapy of type 2 diabetes. The principal side effects of metformin are gastrointestinal intolerance due to bloating, abdominal discomfort, and diarrhea. The drug is cleared by renal filtration, and very high circulating levels (e.g., as a result of overdose or acute renal failure) have been associated with lactic acidosis. However, the occurrence of this complication is now known to be very rare, and metformin may be safely used in patients with reduced estimated glomerular filtration rates (eGFR); the FDA has revised the label for metformin to reflect its safety in patients with eGFR ≥30 mL/min/1.73 m2 (42). A recent randomized trial confirmed previous observations that metformin use is associated with vitamin B12 deficiency and worsening of symptoms of neuropathy (43). This is compatible with a recent report from the Diabetes Prevention Program Outcomes Study (DPPOS) suggesting periodic testing of vitamin B12 (44).
In patients with contraindications or intolerance of metformin, initial therapy should be based on patient factors; consider a drug from another class depicted in Fig. 9.1. When A1C is ≥1.5% (12.5 mmol/mol) above glycemic target (see Section 6 “Glycemic Targets” for more information on selecting appropriate targets), many patients will require dual combination therapy to achieve their target A1C level (45). Insulin has the advantage of being effective where other agents are not and should be considered as part of any combination regimen when hyperglycemia is severe, especially if catabolic features (weight loss, hypertriglyceridemia, ketosis) are present. Consider initiating insulin therapy when blood glucose is ≥300 mg/dL (16.7 mmol/L) or A1C is ≥10% (86 mmol/mol) or if the patient has symptoms of hyperglycemia (i.e., polyuria or polydipsia), even at diagnosis or early in the course of treatment (Fig. 9.2). As glucose toxicity resolves, simplifying the regimen and/or changing to oral agents is often possible.
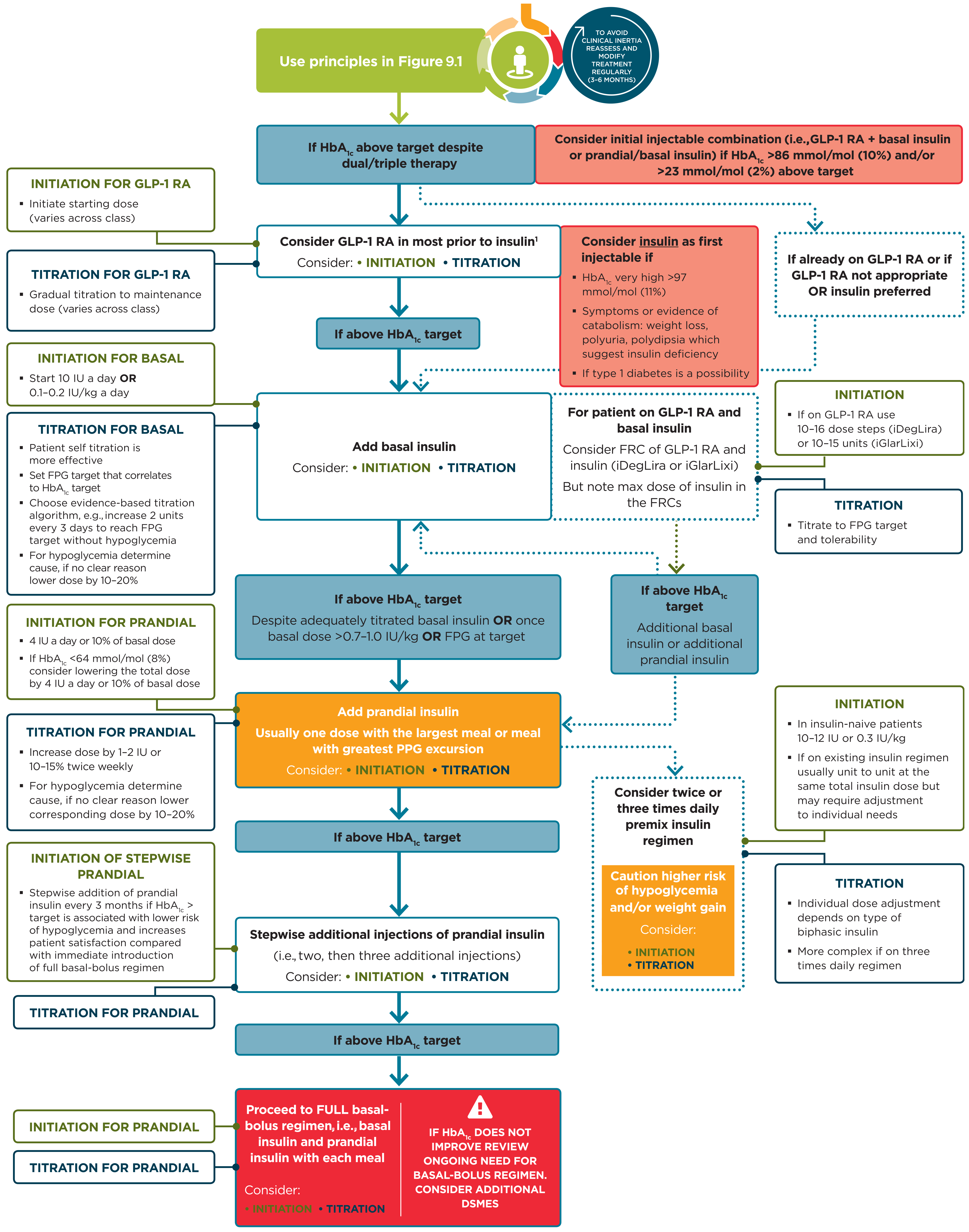
Figure 9.2 Intensifying to injectable therapies

Figure 9.2—Intensifying to injectable therapies. For appropriate context, see Fig. 4.1.
DSMES, diabetes self-management education and support; FPG, fasting plasma glucose; FRC, fixed-ratio combination; GLP-1 RA, glucagon-like peptide 1 receptor agonist; max, maximum; PPG, postprandial glucose. Adapted from Davies et al. (39).
For interactive tool, See here