Recommendations
A reasonable A1C goal for many nonpregnant adults is <7% (53 mmol/mol). A
Providers might reasonably suggest more stringent A1C goals (such as <6.5% [48 mmol/mol]) for selected individual patients if this can be achieved without significant hypoglycemia or other adverse effects of treatment (i.e., polypharmacy). Appropriate patients might include those with short duration of diabetes, type 2 diabetes treated with lifestyle or metformin only, long life expectancy, or no significant cardiovascular disease. C
Less stringent A1C goals (such as <8% [64 mmol/mol]) may be appropriate for patients with a history of severe hypoglycemia, limited life expectancy, advanced microvascular or macrovascular complications, extensive comorbid conditions, or long-standing diabetes in whom the goal is difficult to achieve despite diabetes self-management education, appropriate glucose monitoring, and effective doses of multiple glucose-lowering agents including insulin. B
Reassess glycemic targets over time based on the criteria in Fig. 6.1 or, in older adults,Table 12.1. E
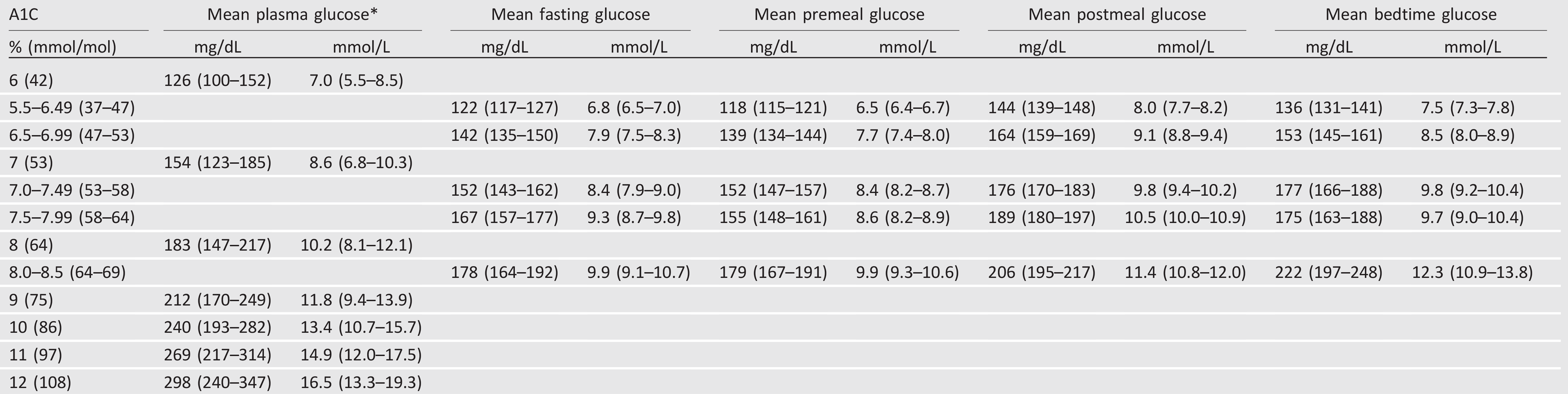
Table 6.1—Mean glucose levels for specified A1C levels (6,7)
xxxxx
4.2.0.0 A1C and Microvascular Complications
Hyperglycemia defines diabetes, and glycemic control is fundamental to diabetes management. The Diabetes Control and Complications Trial (DCCT) (16), a prospective randomized controlled trial of intensive (mean A1C about 7% [53 mmol/mol]) versus standard (mean A1C about 9% [75 mmol/mol]) glycemic control in patients with type 1 diabetes, showed definitively that better glycemic control is associated with 50–76% reductions in rates of development and progression of microvascular (retinopathy, neuropathy, and diabetic kidney disease) complications. Follow-up of the DCCT cohorts in the Epidemiology of Diabetes Interventions and Complications (EDIC) study (17,18) demonstrated persistence of these microvascular benefits over two decades despite the fact that the glycemic separation between the treatment groups diminished and disappeared during follow-up.
The Kumamoto Study (19) and UK Prospective Diabetes Study (UKPDS) (20,21) confirmed that intensive glycemic control significantly decreased rates of microvascular complications in patients with short-duration type 2 diabetes. Long-term follow-up of the UKPDS cohorts showed enduring effects of early glycemic control on most microvascular complications (22).
Therefore, achieving A1C targets of <7% (53 mmol/mol) has been shown to reduce microvascular complications of type 1 and type 2 diabetes when instituted early in the course of disease. Epidemiologic analyses of the DCCT (16) and UKPDS (23) demonstrate a curvilinear relationship between A1C and microvascular complications. Such analyses suggest that, on a population level, the greatest number of complications will be averted by taking patients from very poor control to fair/good control. These analyses also suggest that further lowering of A1C from 7% to 6% [53 mmol/mol to 42 mmol/mol] is associated with further reduction in the risk of microvascular complications, although the absolute risk reductions become much smaller. Given the substantially increased risk of hypoglycemia in type 1 diabetes trials and with polypharmacy in type 2 diabetes, the risks of lower glycemic targets may outweigh the potential benefits on microvascular complications.
Three landmark trials (Action to Control Cardiovascular Risk in Diabetes [ACCORD], Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation [ADVANCE], and Veterans Affairs Diabetes Trial [VADT]) were conducted to test the effects of near normalization of blood glucose on cardiovascular outcomes in individuals with long-standing type 2 diabetes and either known cardiovascular disease (CVD) or high cardiovascular risk. These trials showed that lower A1C levels were associated with reduced onset or progression of some microvascular complications (24-26).
The concerning mortality findings in the ACCORD trial (27), discussed below, and the relatively intense efforts required to achieve near euglycemia should also be considered when setting glycemic targets for individuals with long-standing diabetes such as those studied in ACCORD, ADVANCE, and VADT.
Findings from these studies suggest caution is needed in treating diabetes aggressively to near-normal A1C goals in people with long-standing type 2 diabetes with or at significant risk of CVD. However, on the basis of physician judgment and patient preferences, select patients, especially those with little comorbidity and long life expectancy, may benefit from adopting more intensive glycemic targets (e.g., A1C target <6.5% [48 mmol/mol]) if they can achieve it safely without hypoglycemia or significant therapeutic burden.
Figure 6.1—Depicted are patient and disease factors used to determine optimal A1C targets. Characteristics and predicaments toward the left justify more stringent efforts to lower A1C; those toward the right suggest less stringent efforts. A1C 7% = 53 mmol/mol. Adapted with permission from Inzucchi et al. (40).
xxxxx
4.3.0.0 A1C and Cardiovascular Disease Outcomes
4.3.1.0 Cardiovascular Disease and Type 1 Diabetes
CVD is a more common cause of death than microvascular complications in populations with diabetes. There is evidence for a cardiovascular benefit of intensive glycemic control after long-term follow-up of cohorts treated early in the course of type 1 diabetes. In the DCCT, there was a trend toward lower risk of CVD events with intensive control. In the 9-year post-DCCT follow-up of the EDIC cohort, participants previously randomized to the intensive arm had a significant 57% reduction in the risk of nonfatal myocardial infarction (MI), stroke, or cardiovascular death compared with those previously randomized to the standard arm (28). The benefit of intensive glycemic control in this cohort with type 1 diabetes has been shown to persist for several decades (29) and to be associated with a modest reduction in all-cause mortality (30).
xxxxx
4.3.2.0 Cardiovascular Disease and Type 2 Diabetes
In type 2 diabetes, there is evidence that more intensive treatment of glycemia in newly diagnosed patients may reduce long-term CVD rates. During the UKPDS, there was a 16% reduction in CVD events (combined fatal or nonfatal MI and sudden death) in the intensive glycemic control arm that did not reach statistical significance (P = 0.052), and there was no suggestion of benefit on other CVD outcomes (e.g., stroke). However, after 10 years of observational follow-up, those originally randomized to intensive glycemic control had significant long-term reductions in MI (15% with sulfonylurea or insulin as initial pharmacotherapy, 33% with metformin as initial pharmacotherapy) and in all-cause mortality (13% and 27%, respectively) (22). ACCORD, ADVANCE, and VADT suggested no significant reduction in CVD outcomes with intensive glycemic control in participants followed for shorter durations (3.5–5.6 years) and who had more advanced type 2 diabetes than UKPDS participants. All three trials were conducted in relatively older participants with longer known duration of diabetes (mean duration 8–11 years) and either CVD or multiple cardiovascular risk factors. The target A1C among intensive-control subjects was <6% (42 mmol/mol) in ACCORD, <6.5% (48 mmol/mol) in ADVANCE, and a 1.5% reduction in A1C compared with control subjects in VADT, with achieved A1C of 6.4% vs. 7.5% (46 mmol/mol vs. 58 mmol/mol) in ACCORD, 6.5% vs. 7.3% (48 mmol/mol vs. 56 mmol/mol) in ADVANCE, and 6.9% vs. 8.4% (52 mmol/mol vs. 68 mmol/mol) in VADT. Details of these studies are reviewed extensively in “Intensive Glycemic Control and the Prevention of Cardiovascular Events: Implications of the ACCORD, ADVANCE, and VA Diabetes Trials” (31).
The glycemic control comparison in ACCORD was halted early due to an increased mortality rate in the intensive compared with the standard treatment arm (1.41% vs. 1.14% per year; hazard ratio 1.22 [95% CI 1.01–1.46]), with a similar increase in cardiovascular deaths. Analysis of the ACCORD data did not identify a clear explanation for the excess mortality in the intensive treatment arm (27).
Longer-term follow-up has shown no evidence of cardiovascular benefit or harm in the ADVANCE trial (32). The end-stage renal disease rate was lower in the intensive treatment group over follow-up. However, 10-year follow-up of the VADT cohort (33) showed a reduction in the risk of cardiovascular events (52.7 [control group] vs. 44.1 [intervention group] events per 1,000 person-years) with no benefit in cardiovascular or overall mortality. Heterogeneity of mortality effects across studies was noted, which may reflect differences in glycemic targets, therapeutic approaches, and population characteristics (34).
Mortality findings in ACCORD (27) and subgroup analyses of VADT (35) suggest that the potential risks of intensive glycemic control may outweigh its benefits in higher-risk patients. In all three trials, severe hypoglycemia was significantly more likely in participants who were randomly assigned to the intensive glycemic control arm. Those patients with long duration of diabetes, a known history of hypoglycemia, advanced atherosclerosis, or advanced age/frailty may benefit from less aggressive targets (36,37).
As discussed further below, severe hypoglycemia is a potent marker of high absolute risk of cardiovascular events and mortality (38). Providers should be vigilant in preventing hypoglycemia and should not aggressively attempt to achieve near-normal A1C levels in patients in whom such targets cannot be safely and reasonably achieved. As discussed in Section 9 “Pharmaco-logic Approaches to Glycemic Treatment,” addition of specific sodium–glucose cotransporter 2 inhibitors (SGLT2i) or glucagon-like peptide 1 receptor agonists (GLP-1 RA) to improve cardiovascular outcomes in patients with established CVD is indicated with consideration of glycemic goals. If the patient is not at A1C target, continue metformin unless contraindicated and add SGLT2i or GLP-1 RA with proven cardiovascular benefit. If the patient is meeting A1C target, considerone of three strategies (39):
If already on dual therapy or multiple glucose-lowering therapies and not on an SGLT2i or GLP-1 RA, consider switching to one of these agents with proven cardiovascular benefit.
Reconsider/lower individualized A1C target and introduce SGLT2i or GLP-1 RA.
Reassess A1C at 3-month intervals and add SGLT2i or GLP-1 RA if A1C goes above target.
xxxxx
4.4.0.0 Setting and Modifying A1C Goals
Numerous factors must be considered when setting glycemic targets. The ADA proposes general targets appropriate for many patients but emphasizes the importance of individualization based on key patient characteristics. Glycemic targets must be individualized in the context of shared decision making to address the needs and preferences of each patient and the individual characteristics that influence risks and benefits of therapy for each patient.
The factors to consider in individualizing goals are depicted in Fig. 6.1. Figure 6.1 is not designed to be applied rigidly but to be used as a broad construct to guide clinical decision making (40) in both type 1 and type 2 diabetes. More stringent control (such as an A1C of 6.5% [48 mmol/mol] or <7% [53 mmol/mol]) may be recommended if it can be achieved safely and with acceptable burden of therapy and if life expectancy is sufficient to reap benefits of tight control. Less stringent control (A1C up to 8% [64 mmol/mol]) may be recommended if the life expectancy of the patient is such that the benefits of an intensive goal may not be realized, or if the risks and burdens outweigh the potential benefits. Severe or frequent hypoglycemia is an absolute indication for the modification of treatment regimens, including setting higher glycemic goals.
Diabetes is a chronic disease that progresses over decades. Thus, a goal that might be appropriate for an individual early in the course of the disease may change over time. Newly diagnosed patients and/or those without comorbidities that limit life expectancy may benefit from intensive control proven to prevent microvascular complications. Both DCCT/EDIC and UKPDS demonstrated metabolic memory, or a legacy effect, in which a finite period of intensive control yielded benefits that extended for decades after that control ended. Thus, a finite period of intensive control to near-normal A1C may yield enduring benefits even if control is subsequently deintensified as patient characteristics change. Over time, comorbidities may emerge, decreasing life expectancy and the potential to reap benefits from intensive control. Also, with longer duration of disease, diabetes may become more difficult to control, with increasing risks and burdens of therapy. Thus, A1C targets should be reevaluated over time to balance the risks and benefits as patient factors change.
Recommended glycemic targets for many nonpregnant adults are shown in Table 6.2. The recommendations include blood glucose levels that appear to correlate with achievement of an A1C of <7% (53 mmol/mol). The issue of preprandial versus postprandial SMBG targets is complex (41). Elevated post-challenge (2-h oral glucose tolerance test) glucose values have been associated with increased cardiovascular risk independent of fasting plasma glucose in some epidemiologic studies, but intervention trials have not shown postpran-dial glucose to be a cardiovascular risk factor independent of A1C. In subjects with diabetes, surrogate measures of vascular pathology, such as endothelial dysfunction, are negatively affected by postprandial hyperglycemia. It is clear that postprandial hyperglycemia, like preprandial hyperglycemia, contributes to elevated A1C levels, with its relative contribution being greater at A1C levels that are closer to 7% (53 mmol/mol). However, outcome studies have clearly shown A1C to be the primary predictor of complications, and landmark trials of glycemic control such as the DCCT and UKPDS relied overwhelmingly on pre-prandial SMBG. Additionally, a randomized controlled trial in patients with known CVD found no CVD benefit of insulin regimens targeting postprandial glucose compared with those targeting preprandial glucose (42). Therefore, it is reasonable for postprandial testing to be recommended for individuals who have premeal glucose values within target but have A1C values above target. Measuring postprandial plasma glucose 1–2 h after the start of a meal and using treatments aimed at reducing postprandial plasma glucose values to <180 mg/dL (10.0 mmol/L) may help to lower A1C.
An analysis of data from 470 participants in the ADAG study (237 with type 1 diabetes and 147 with type 2 diabetes) found that actual average glucose levels associated with conventional A1C targets were higher than older DCCT and ADA targets (Table 6.1) (7,43). These findings support that premeal glucose targets may be relaxed without undermining overall glycemic control as measured by A1C. These data prompted the revision in the ADA-recommended premeal glucose target to 80–130 mg/dL (4.4–7.2 mmol/L) but did not affect the definition of hypoglycemia.
Table 6.2—Summary of glycemic recommendations for many nonpregnant adults with diabetes

xxxxx
5.0.0.0 HYPOGLYCEMIA
5.1.0.0 Recommendations
Recommendations
Individuals at risk for hypoglycemia should be asked about symptomatic and asymptomatic hypoglycemia at each encounter. C
Glucose(15–20 g) is thepreferred treatment for the conscious individual with blood glucose
<70 mg/dL [3.9 mmol/L]), although any form of carbohydrate that contains glucose may be used. Fifteen minutes after treatment, if SMBG shows continued hypoglycemia, the treatment should be repeated. Once SMBG returns to normal, the individual should consume a meal or snack to prevent recurrence of hypoglycemia. E
Glucagon should be prescribed for all individuals at increased risk of level 2 hypoglycemia, defined as blood glucose <54 mg/dL (3.0 mmol/L), so it is available should it be needed. Caregivers, school personnel, or family members of these individuals should know where it is and when and how to administer it. Glucagon administration is not limited to health care professionals. E
Hypoglycemia unawareness or one or more episodes of level 3 hypoglycemia should trigger reevaluation of the treatment regimen. E
Insulin-treated patients with hypoglycemia unawareness or an episode of level 2 hypoglycemia should be advised to raise their glycemic targets to strictly avoid hypoglycemia for at least several weeks in order to partially reverse hypoglycemia unawareness and reduce risk of future episodes. A
Ongoing assessment of cognitive function is suggested with
increased vigilance for hypoglycemia by the clinician, patient, and caregivers if low cognition or declining cognition is found. B
Table 6.3—Classification of hypoglycemia (44)
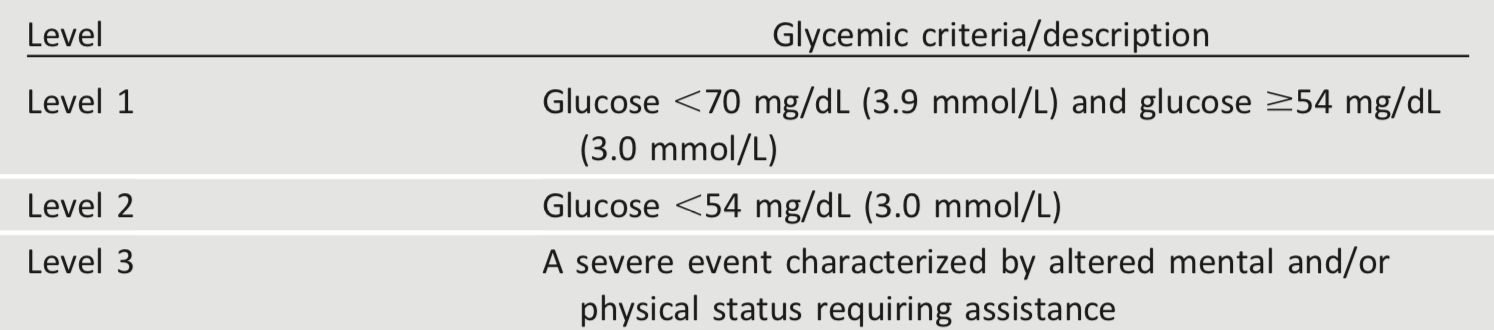
Hypoglycemia is the major limiting factor in the glycemic management of type 1 and type 2 diabetes. Recommendations regarding the classification of hypoglycemia are outlined in Table 6.3 (44). Level 1 hypoglycemia is defined as a measurable glucose concentration <70 mg/dL (3.9 mmol/L) but ≥54 mg/dL (3.0 mmol/L). A blood glucose concentration of 70 mg/dL (3.9 mmol/L) has been recognized as a threshold for neuroendocrine responses to falling glucose in people without diabetes. Because many people with diabetes demonstrate impaired counterregulatory responses to hypoglycemia and/or experience hypoglycemia unawareness, a measured glucose level <70 mg/dL (3.9 mmol/L) is considered clinically important, independent of the severity of acute hypoglycemic symptoms. Level 2 hypoglycemia (defined as a blood glucose concentration <54 mg/dL [3.0 mmol/L]) is the threshold at which neuroglycopenic symptoms begin to occur and requires immediate action to resolve the hypoglycemic event. Lastly, level 3 hypoglycemia is defined as a severe event characterized by altered mental and/or physical functioning that requires assistance from another person for recovery. Studies of rates of level 3 hypoglycemia that rely on claims data for hospitalization, emergency department visits, and ambulance use substantially underestimate rates of level 3 hypoglycemia (45), yet find high burden of hypoglycemia in adults over 60 years of age in the community (46). African Americans are at substantially increased risk of level 3 hypoglycemia (46,47). In addition to age and race, other important risk factors found in a community-based epidemiologic cohort of older black and white adults with type 2 diabetes include insulin use, poor or moderate versus good glycemic control, albuminuria, and poor cognitive function (46).
Symptoms of hypoglycemia include, but are not limited to, shakiness, irritability, confusion, tachycardia, and hunger. Hypoglycemia may be inconvenient or frightening to patients with diabetes. Level 3 hypoglycemia may be recognized or unrecognized and can progress to loss of consciousness, seizure, coma, or death. It is reversed by administration of rapid-acting glucose or glucagon. Hypoglycemia can cause acute harm to the person with diabetes or others, especially if it causes falls, motor vehicle accidents, or other injury. A large cohort study suggested that among older adults with type 2 diabetes, a history of level 3 hypoglycemia was associated with greater risk of dementia (48). Conversely, in a substudy of the ACCORD trial, cognitive impairment at baseline or decline in cognitive function during the trial was significantly associated with subsequent episodes of level 3 hypoglycemia (49). Evidence from DCCT/EDIC, which involved adolescents and younger adults with type 1 diabetes, found no association between frequency of level 3 hypoglycemia and cognitive decline (50), as discussed in Section 13 “Children and Adolescents.”
Level 3 hypoglycemia was associated with mortality in participants in both the standard and the intensive glycemia arms of the ACCORD trial, but the relationships between hypoglycemia, achieved A1C, and treatment intensity were not straight-forward. An association of level 3 hypoglycemia with mortality was also found in the ADVANCE trial (51). An association between self-reported level 3 hypoglycemia and 5-year mortality has also been reported in clinical practice (52).
Young children with type 1 diabetes and the elderly, including those with type 1 and type 2 diabetes (48,53), are noted as particularly vulnerable to hypoglycemia because of their reduced ability to recognize hypoglycemic symptoms and effectively communicate their needs. Individualized glucose targets, patient education, dietary intervention (e.g., bedtime snack to prevent overnight hypoglycemia when specifically needed to treat low blood glucose), exercise management, medication adjustment, glucose monitoring, and routine clinical surveillance may improve patient outcomes (54). CGM with automated low glucose suspend has been shown to be effective in reducing hypoglycemia in type 1 diabetes (55). For patients with type 1 diabetes with level 3 hypoglycemia and hypoglycemia unawareness that persists despite medical treatment, human islet transplantation may be an option, but the approach remains experimental (56,57).
In 2015, the ADA changed its preprandial glycemic target from 70–130 mg/dL (3.9–7.2 mmol/L) to 80–130 mg/dL (4.4–7.2 mmol/L). This change reflects the results of the ADAG study, which demonstrated that higher glycemic targets corresponded to A1C goals (7). An additional goal of raising the lower range of the glycemic target was to limit overtreatment and provide a safety margin in patients titrating glucose-lowering drugs such as insulin to glycemic targets.
xxxxx
5.2.0.0 Hypoglycemia Treatment
xxxxx
5.2.1.0 Overview
Providers should continue to counsel patients to treat hypoglycemia with fast-acting carbohydrates at the hypoglycemia alert value of 70 mg/dL (3.9 mmol/L) or less. Hypoglycemia treatment requires ingestion of glucose- or carbohydrate-containing foods. The acute glycemic response correlates better with the glucose content of food than with the carbohydrate content of food. Pure glucose is the preferred treatment, but any form of carbohydrate that contains glucose will raise blood glucose. Added fat may retard and then prolong the acute glycemic response. In type 2 diabetes, ingested protein may increase insulin response without increasing plasma glucose concentrations (58). Therefore, carbohydrate sources high in protein should not be used to treat or prevent hypoglycemia. Ongoing insulin activity or insulin secretagogues may lead to recurrent hypoglycemia unless more food is ingested after recovery. Once the glucose returns to normal, the individual should be counseled to eat a meal or snack to prevent recurrent hypoglycemia.
5.2.2.0 Glucagon
The use of glucagon is indicated for the treatment of hypoglycemia in people unable or unwilling to consume carbohydrates by mouth. Those in close contact with, or having custodial care of, people with hypoglycemia-prone diabetes (family members, roommates, school personnel, child care providers, correctional institution staff, or coworkers) should be instructed on the use of glucagon kits, including where the kit is and when and how to administer glucagon. An individual does not need to be a health care professional to safely administer glucagon. Care should be taken to ensure that glucagon kits are not expired.
xxxxx
5.3.0.0 Hypoglycemia Prevention
Hypoglycemia prevention is a critical component of diabetes management. SMBG and, for some patients, CGM are essential tools to assess therapy and detect incipient hypoglycemia. Patients should understand situations that increase their risk of hypoglycemia, such as when fasting for tests or procedures, when meals are delayed, during and after the consumption of alcohol, during and after intense exercise, and during sleep. Hypoglycemia may increase the risk of harm to self or others, such as with driving. Teaching people with diabetes to balance insulin use and carbohydrate intake and exercise are necessary, but these strategies are not always sufficient for prevention.
In type 1 diabetes and severely insulindeficient type 2 diabetes, hypoglycemia unawareness (or hypoglycemia-associated autonomic failure) can severely compromise stringent diabetes control and quality of life. This syndrome is characterized by deficient counterregulatory hormone release, especially in older adults, and a diminished autonomic response, which both are risk factors for, and caused by, hypoglycemia. A corollary to this “vicious cycle” is that several weeks of avoidance of hypoglycemia has been demonstrated to improve counterregulation and hypoglycemia awareness in many patients (59). Hence, patients with one or more episodes of clinically significant hypoglycemia may benefit from at least short-term relaxation of glycemic targets.
xxxxx
6.0.0.0 INTERCURRENT ILLNESS
For further information on management of patients with hyperglycemia in the hospital, please refer to Section 15 “Diabetes Care in the Hospital.
Stressful events (e.g., illness, trauma, surgery, etc.) may worsen glycemic control and precipitate diabetic ketoacidosis or nonketotic hyperglycemic hyperosmolar state, life-threatening conditions that require immediate medical care to prevent complications and death. Any condition leading to deterioration in glycemic control necessitates more frequent monitoring of blood glucose; ketosis-prone patients also require urine or blood ketone monitoring. If accompanied by ketosis, vomiting, or alteration in the level of consciousness, marked hyperglycemia requires temporary adjustment of the treatment regimen and immediate interaction with the diabetes care team. The patient treated with noninsulin therapies or medical nutrition therapy alone may require insulin. Adequate fluid and caloric intake must be ensured. Infection or dehydration is more likely to necessitate hospitalization of the person with diabetes than the person without diabetes.
A physician with expertise in diabetes management should treat the hospitalized patient. For further information on the management of diabetic ketoacidosis and the nonketotic hyperglycemic hyperosmolar state, please refer to the ADA consensus report “Hyperglycemic Crises in Adult Patients With Diabetes” (60).
xxxxx
7.0.0.0 References
- Laiteerapong N, Ham SA, Gao Y, et al. The legacy effect in type 2 diabetes: impact of early glycemic control on future complications (the Diabetes & Aging Study). Diabetes Care. In press
- Stratton IM, Adler AI, Neil HAW, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ 2000;321:405–412
- Little RR, Rohlfing CL, Sacks DB; National Glycohemoglobin Standardization Program (NGSP) Steering Committee. Status of hemoglobin A1c measurement and goals for improvement: from chaos to order for improving diabetes care. Clin Chem 2011;57:205–214
- Jovanovicˇ L, Savas H, Mehta M, Trujillo A,
Pettitt DJ. Frequent monitoring of A1C during pregnancy as a treatment tool to guide therapy. Diabetes Care 2011;34:53–54
- Beck RW, Connor CG, Mullen DM, Wesley DM, Bergenstal RM. The fallacy of average: how using HbA1c alone to assess glycemic control can be misleading. Diabetes Care 2017;40:994–999
- Nathan DM, Kuenen J, Borg R, Zheng H, Schoenfeld D, Heine RJ; A1c-Derived Average Glucose Study Group. Translating the A1C assay into estimated average glucose values. Diabetes Care 2008;31:1473–1478
- Wei N, Zheng H, Nathan DM. Empirically establishing blood glucose targets to achieve HbA1c goals. Diabetes Care 2014;37:1048–1051
- Selvin E. Are there clinical implications of racial differences in HbA1c? A difference, to be a difference, must make a difference. Diabetes Care 2016;39:1462–1467
- Bergenstal RM, Gal RL, Connor CG, et al.; T1D Exchange Racial Differences Study Group. Racial differences in the relationship of glucose concentrations and hemoglobin A1c levels. Ann Intern Med 2017;167:95–102
- Lacy ME, Wellenius GA, Sumner AE, et al. Association of sickle cell trait with hemoglobin A1c in African Americans. JAMA 2017;317:507– 515
- Rohlfing C, Hanson S, Little RR. Measurement of hemoglobin A1c in patients with sickle cell trait. JAMA 2017;317:2237
- Wheeler E, Leong A, Liu C-T, et al.; EPIC-CVD Consortium; EPIC-InterAct Consortium; Lifelines Cohort Study. Impact of common genetic determinants of hemoglobin A1c on type 2 diabetes risk and diagnosis in ancestrally diverse populations: a transethnic genome-wide metaanalysis. PLoS Med 2017;14:e1002383
- Wilson DM, Kollman; Diabetes Research in Children Network (DirecNet) Study Group. Relationship of A1C to glucose concentrations in children with type 1 diabetes: assessments by high-frequency glucose determinations by sensors. Diabetes Care 2008;31:381–385
- Buse JB, Kaufman FR, Linder B, Hirst K, El Ghormli L, Willi S; HEALTHY Study Group. Diabetes screening with hemoglobin A1c versus fasting plasma glucose in a multiethnic middle-school cohort. Diabetes Care 2013;36:429–435
- Kamps JL, Hempe JM, Chalew SA. Racial disparity in A1C independent of mean blood glucose in children with type 1 diabetes. Diabetes Care 2010;33:1025–1027
- Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977–986
- Diabetes Control and Complications Trial (DCCT)/Epidemiology of Diabetes Interventions and Complications (EDIC) Research Group. Effect of intensive diabetes therapy on the progression of diabetic retinopathy in patients with type 1 diabetes: 18 years of follow-up in the DCCT/EDIC. Diabetes 2015;64:631–642
- Lachin JM, Genuth S, Cleary P, Davis MD, Nathan DM; Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. Retinopathy and nephropathy in patients with type 1 diabetes four years after a trial of intensive therapy [published correction appears in N Engl J Med 2000;342:1376]. N Engl J Med 2000;342:381–389
- Ohkubo Y, Kishikawa H, Araki E, et al. Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non-insulin-dependent diabetes mellitus: a randomized prospective 6-year study. Diabetes Res Clin Pract 1995;28: 103–117
- UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998;352:854–865
- UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998;352:837–853
- Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HAW. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008;359:1577–1589
- Adler AI, Stratton IM, Neil HAW, et al. Association of systolic blood pressure with macrovascular and microvascular complications of type 2 diabetes (UKPDS 36): prospective observational study. BMJ 2000;321:412–419
- Duckworth W, Abraira C, Moritz T, et al.; VADT Investigators. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 2009;360:129–139
- Patel A, MacMahon S, Chalmers J, et al.; ADVANCE Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008; 358:2560–2572
- Ismail-Beigi F, Craven T, Banerji MA, et al.; ACCORD trial group. Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet 2010; 376:419–430
- Gerstein HC, Miller ME, Byington RP, et al.; Action to Control Cardiovascular Risk in Diabetes Study Group. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008; 358:2545–2559
- Nathan DM, Cleary PA, Backlund J-YC, et al.; Diabetes Control and Complications Trial/ Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study Research Group. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med 2005;353:2643–2653
- Nathan DM, Zinman B, Cleary PA, et al.; Diabetes Control and Complications Trial/ Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Research Group. Modern-day clinical course of type 1 diabetes mellitus after 30 years’ duration: the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications and Pittsburgh Epidemiology of Diabetes Complications experience (1983-2005). Arch Intern Med 2009;169:1307–1316
- Orchard TJ, Nathan DM, Zinman B, et al.; Writing Group for the DCCT/EDIC Research Group. Association between 7 years of intensive treatment of type 1 diabetes and long-term mortality. JAMA 2015;313:45–53
- Skyler JS, Bergenstal R, Bonow RO, et al.; American Diabetes Association; American
College of Cardiology Foundation; American Heart Association. Intensive glycemic control and the prevention of cardiovascular events: implications of the ACCORD, ADVANCE, and VA diabetes trials: a position statement of the American Diabetes Association and a scientific statement of the American College of Cardiology Foundation and the American Heart Association. Diabetes Care 2009;32:187–192
- Zoungas S, Chalmers J, Neal B, et al.; ADVANCE-ON Collaborative Group. Follow-up of blood-pressure lowering and glucose control in type 2 diabetes. N Engl J Med 2014;371:1392– 1406
- Hayward RA, Reaven PD, Wiitala WL, et al.; VADT Investigators. Follow-up of glycemic control and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2015;372:2197–2206
- Turnbull FM, Abraira C, Anderson RJ, et al.; Control Group. Intensive glucose control and macrovascular outcomes in type 2 diabetes [published correction appears in Diabetologia 2009;52:2470]. Diabetologia 2009;52:2288–2298
- Duckworth WC, Abraira C, Moritz TE, et al.; Investigators of the VADT. The duration of diabetes affects the response to intensive glucose control in type 2 subjects: the VA Diabetes Trial. J Diabetes Complications 2011;25:355–361
- Lipska KJ, Ross JS, Miao Y, Shah ND, Lee SJ, Steinman MA. Potential overtreatment of diabetes mellitus in older adults with tight glycemic control. JAMA Intern Med 2015;175: 356–362
- Vijan S, Sussman JB, Yudkin JS, Hayward RA. Effect of patients’ risks and preferences on health gains with plasma glucose level lowering in type 2 diabetes mellitus. JAMA Intern Med 2014;174:1227–1234
- Lee AK, Warren B, Lee CJ, et al. The association of severe hypoglycemia with incident cardiovascular events and mortality in adults with type 2 diabetes. Diabetes Care 2018;41: 104–111
- Davies MJ, D’Alessio DA, Fradkin J, et al. Management of hyperglycemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2018;41:2669–2701
- Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycemia in type 2 diabetes, 2015: a patient-centered approach: update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2015;38:140– 149
- American Diabetes Association. Postprandial blood glucose. Diabetes Care 2001;24:775– 778
- Raz I, Wilson PWF, Strojek K, et al. Effects of prandial versus fasting glycemia on cardiovascular outcomes in type 2 diabetes: the HEART2D trial. Diabetes Care 2009;32:381– 386
- Albers JW, Herman WH, Pop-Busui R, et al.; Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. Effect of prior intensive insulin treatment during the Diabetes Control and Complications Trial (DCCT) on peripheral neuropathy in type 1 diabetes during the Epidemiology of Diabetes Interventions and
Complications (EDIC) study. Diabetes Care 2010;33:1090–1096
- Agiostratidou G, Anhalt H, Ball D, et al. Standardizing clinically meaningful outcome measures beyond HbA1c for type 1 diabetes: a consensus report of the American Association of Clinical Endocrinologists, the American Association of Diabetes Educators, the American Diabetes Association, the Endocrine Society, JDRF International, The Leona M. and Harry B. Helmsley Charitable Trust, the Pediatric Endocrine Society, and the T1D Exchange. Diabetes Care 2017;40:1622–1630
- Karter AJ, Moffet HH, Liu JY, Lipska KJ. Surveillance of hypoglycemiadlimitations of emergency department and hospital utilization data. JAMA Intern Med 2018;178:987–988
- Lee AK, Lee CJ, Huang ES, Sharrett AR, Coresh J, Selvin E. Risk factors for severe hypoglycemia in black and white adults with diabetes: the Atherosclerosis Risk in Communities (ARIC) study. Diabetes Care 2017;40:1661–1667
- Karter AJ, Lipska KJ, O’Connor PJ, et al.; SUPREME-DM Study Group. High rates of severe hypoglycemia among African American patients with diabetes: the SUrveillance, PREvention, and ManagEment of Diabetes Mellitus (SUPREME-DM) network. J Diabetes Complications 2017;31: 869–873
- Whitmer RA, Karter AJ, Yaffe K, Quesenberry CP Jr, Selby JV. Hypoglycemic episodes and risk of dementia in older patients with type 2 diabetes mellitus. JAMA 2009;301:1565–1572
- Punthakee Z, Miller ME, Launer LJ, et al.; ACCORD Group of Investigators; ACCORD-MIND Investigators. Poor cognitive function and risk of severe hypoglycemia in type 2 diabetes: post hoc epidemiologic analysis of the ACCORD trial. Diabetes Care 2012;35: 787–793
- Jacobson AM, Musen G, Ryan CM, et al.; Diabetes Control and Complications Trial/ Epidemiology of Diabetes Interventions and Complications Study Research Group. Longterm effect of diabetes and its treatment on cognitive function. N Engl J Med 2007;356: 1842–1852
- Zoungas S, Patel A, Chalmers J, et al.; ADVANCE Collaborative Group. Severe hypoglycemia and risks of vascular events and death. N Engl J Med 2010;363:1410–1418
- McCoy RG, Van Houten HK, Ziegenfuss JY, Shah ND, Wermers RA, Smith SA. Increased mortality of patients with diabetes reporting severe hypoglycemia. Diabetes Care 2012;35: 1897–1901
- DuBose SN, Weinstock RS, Beck RW, et al. Hypoglycemia in older adults with type 1 diabetes. Diabetes Technol Ther 2016;18:765– 771
- Seaquist ER, Anderson J, Childs B, et al. Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association and the Endocrine Society. Diabetes Care 2013;36: 1384–1395
- Bergenstal RM, Klonoff DC, Garg SK, et al.; ASPIRE In-Home Study Group. Threshold-based insulin-pump interruption for reduction of hypoglycemia. N Engl J Med 2013;369:224– 232
- Hering BJ, Clarke WR, Bridges ND, et al.; Clinical Islet Transplantation Consortium.
Phase 3 trial of transplantation of human islets in type 1 diabetes complicated by severe hypoglycemia. Diabetes Care 2016;39:1230– 1240
- Harlan DM. Islet transplantation for hypoglycemia unawareness/severe hypoglycemia:
caveat emptor. Diabetes Care 2016;39:1072– 1074
- Layman DK, Clifton P, Gannon MC, Krauss RM, Nuttall FQ. Protein in optimal health: heart disease and type 2 diabetes. Am J Clin Nutr 2008; 87:1571S–1575S
- Cryer PE. Diverse causes of hypoglycemia-associated autonomic failure in diabetes. N Engl J Med 2004;350:2272–2279
- Kitabchi AE, Umpierrez GE, Miles JM, Fisher JN. Hyperglycemic crises in adult patients with diabetes. Diabetes Care 2009;32:1335–1343