1.0.0.0 Introduction
The American Diabetes Association (ADA) “Standards of Medical Care in Diabetes” includes ADA’s current clinical practice recommendations and is intended to provide the components of diabetes care, general treatment goals and guidelines, and tools to evaluate quality of care. Members of the ADA Professional Practice Committee, a multidisciplinary expert committee, are responsible for updating the Standards of Care annually, or more frequently as warranted. For a detailed description of ADA standards, statements, and reports, as well as the evidence-grading system for ADA’s clinical practice recommendations, please refer to the Standards of Care Introduction. Readers who wish to comment on the Standards of Care are invited to do so at professional.diabetes.org/SOC.
For prevention and management of diabetes complications in children and adolescents, please refer to Section 13 “Children and Adolescents.”
Atherosclerotic cardiovascular disease (ASCVD)ddefined as coronary heart disease, cerebrovascular disease, or peripheral arterial disease presumed to be of atherosclerotic origindis the leading cause of morbidity and mortality for individuals with diabetes and results in an estimated $37.3 billion in cardiovascular-related spending per year associated with diabetes (1). Common conditions coexisting with type 2 diabetes (e.g., hypertension and dyslipidemia) are clear risk factors for ASCVD, and diabetes itself confers independent risk. Numerous studies have shown the efficacy of controlling individual cardiovascular risk factors in preventing or slowing ASCVD in people with diabetes. Furthermore, large benefits are seen when multiple cardiovascular risk factors are addressed simultaneously. Under the current paradigm of aggressive risk factor modification in patients with diabetes, there is evidence that measures of 10-year coronary heart disease (CHD) risk among U.S. adults with diabetes have improved significantly over the past decade (2) and that ASCVD morbidity and mortality have decreased (3,4).
Heart failure is another major cause of morbidity and mortality from cardiovascular disease. Recent studies have found that rates of incident heart failure hospitalization (adjusted for age and sex) were twofold higher in patients with diabetes compared with those without (5,6). People with diabetes may have heart failure with preserved ejection fraction (HFpEF) or with reduced ejection fraction (HFrEF). Hypertension is often a precursor of heart failure of either type, and ASCVD can coexist with either type (7), whereas prior myocardial infarction (MI) is often a major factor in HFrEF. Rates of heart failure hospitalization have been improved in recent trials including patients with type 2 diabetes, most of whom also had ASCVD, with sodium– glucose cotransporter 2 (SGLT2) inhibitors (8-10).
For prevention and managementof both ASCVD and heart failure, cardiovascular risk factors should be systematically assessed at least annually in all patients with diabetes. These risk factors include obesity/overweight, hypertension, dyslipidemia, smoking, a family history of premature coronary disease, chronic kidney disease, and the presence of albuminuria. Modifiable abnormal risk factors should be treated as described in these guidelines.
This section has received endorsement from the American College of Cardiology. Suggested citation: American Diabetes Association. 10. Cardiovascular disease and risk management: Standards of Medical Care in Diabetesd2019. Diabetes Care 2019;42(Suppl. 1): S103–S123 © 2018 by the American Diabetes Association. Readers may use this article as long as the work is properly cited, the use is educational and not for profit, and the work is not altered. More information is available at http://www.diabetesjournals .org/content/license.
The Risk Calculator
The American College of Cardiology/ American Heart Association ASCVD risk calculator (Risk Estimator Plus) is generally a useful tool to estimate 10-year ASCVD risk (http://tools.acc.org/ASCVD-Risk-Estimator-Plus). Thesecalculatorshave diabetes as a risk factor, since diabetes itself confers increased risk for ASCVD, although it should be acknowledged that these risk calculators do not account for the duration of diabetes or the presence of diabetes complications, such as albumin-uria. Although some variability in calibration exists in various subgroups, including by sex, race, and diabetes, the overall risk prediction does not differ in those with or without diabetes (11-14), validating the use of risk calculators in people with diabetes. The 10-year risk of a first ASCVD event should be assessed to better stratify ASCVD risk and help guide therapy, as described below.
Recently, risk scores and other cardiovascular biomarkers have been developed for risk stratification of secondary prevention patients (i.e., those who are already high risk because they have ASCVD) but are not yet in widespread use (15,16). With newer, more expensive lipid-lowering therapies now available, use of these risk assessments may help target these new therapies to “higher risk” ASCVD patients in the future.
2.0.0.0 HYPERTENSION/BLOOD PRESSURE CONTROL
2.1.0.0 Overview
Hypertension, defined as a sustained blood pressure ≥140/90 mmHg, is common among patients with either type 1 or type 2 diabetes. Hypertension is a major risk factor for both ASCVD and microvascular complications. Moreover, numerous studies have shown that antihypertensive therapy reduces ASCVD events, heart failure, and microvascular complications. Please refer to the American Diabetes Association (ADA) position statement “Diabetes and Hypertension” for a detailed review of the epidemiology, diagnosis, and treatment of hypertension (17). The recommendations presented here reflect ADA’s updated stance on blood pressure.
2.2.0.0 Screening and Diagnosis
Recommendations
10.1 Blood pressure should be measured at every routine clinical visit. Patients found to have elevated blood pressure (≥140/ 90 mmHg) should have blood pressure confirmed using multiple readings, including measurements on a separate day, to diagnose hypertension. B
10.2 All hypertensive patients with diabetes should monitor their blood pressure at home. B
Blood pressure should be measured by a trained individual and should follow the guidelines established for the general population: measurement in the seated position, with feet on the floor and arm supported at heart level, after 5 min of rest. Cuff size should be appropriate for the upper-arm circumference. Elevated values should be confirmed on a separate day. Postural changes in blood pressure and pulse may be evidence of autonomic neuropathy and therefore require adjustment of blood pressure targets. Orthostatic blood pressure measurements should be checked on initial visit and as indicated.
Home blood pressure self-monitoring and 24-h ambulatory blood pressure monitoring may provide evidence of white coat hypertension, masked hypertension, or other discrepancies between office and “true” blood pressure (17). In addition to confirming or refuting a diagnosis of hypertension, home blood pressure assessment may be useful to monitor antihypertensive treatment. Studies of individuals without diabetes found that home measurements may better correlate with ASCVD risk than office measurements (18,19). Moreover, home blood pressure monitoring may improve patient medication adherence and thus help reduce cardiovascular risk (20).
2.3.0.0 Treatment Goals
2.3.1.0 Recommendations
Recommendations
10.3 For patients with diabetes and hypertension, blood pressure targets should be individualized through a shared decision-making process that addresses cardiovascular risk, potential adverse effects of antihypertensive medications, and patient preferences. C
10.4 For individuals with diabetes and hypertension at higher cardiovascular risk (existing atherosclerotic cardiovascular disease or 10-year atherosclerotic cardiovascular disease risk >15%), a blood pressure targetof,130/ 80 mmHg may be appropriate, if it can be safely attained. C
10.5 For individuals with diabetes and hypertension at lower risk for cardiovascular disease (10-year atherosclerotic cardiovascular disease risk <15%), treat to a blood pressure target of,140/90 mmHg. A
10.6 In pregnant patients with diabetes and preexisting hypertension who are treated with antihypertensive therapy, blood pressure targets of 120–160/80– 105 mmHg are suggested in the interest of optimizing long-term maternal health and minimizing impaired fetal growth. E
Randomized clinical trials have demonstrated unequivocally that treatment of hypertension to blood pressure <140/90 mmHg reduces cardiovascular events as well as microvascular complications (21-27). Therefore, patients with type 1 or type 2 diabetes who have hypertension should, at a minimum, be treated to blood pressure targets of <140/90 mmHg. The benefits and risks of intensifying antihypertensive therapy to target blood pressures lower than <140/90 mmHg (e.g., <130/80 or <120/80 mmHg) have been evaluated in large randomized clinical trials and metaanalyses of clinical trials. Notably, there is an absence of high-quality data available to guide blood pressure targets in type 1 diabetes.
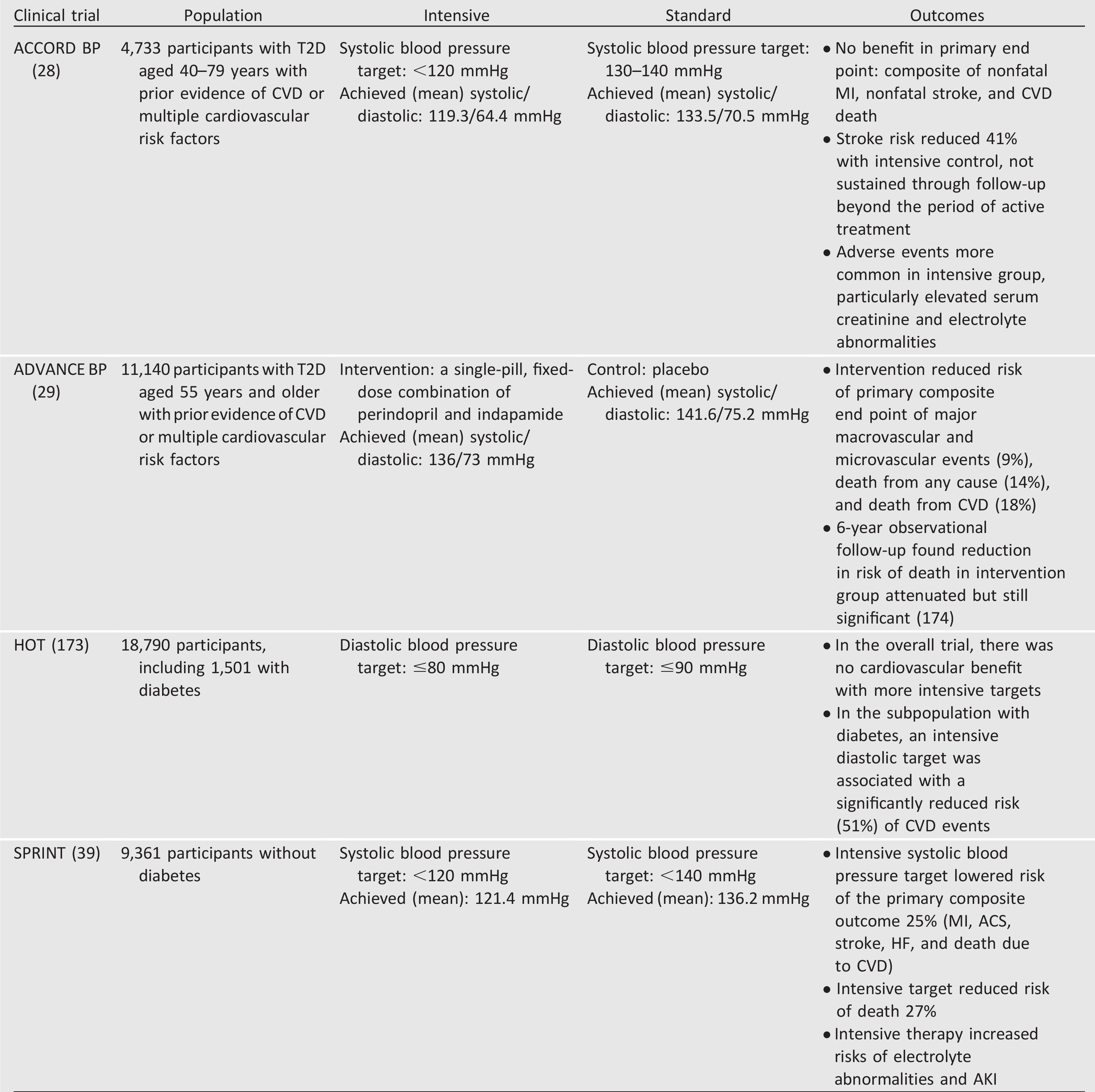
2.3.2.0 Randomized Controlled Trials of Intensive Versus Standard Blood Pressure Control
The Action to Control Cardiovascular Riskin Diabetes blood pressure (ACCORD BP) trial provides the strongest direct assessment of the benefits and risks of intensive blood pressure control among people with type 2 diabetes (28). In ACCORD BP, compared with standard blood pressure control (target systolic blood pressure <140 mmHg), intensive blood pressure control (target systolic blood pressure <120 mmHg) did not reduce total major atherosclerotic cardiovascular events but did reduce the risk of stroke, at the expense of increased adverse events (Table 10.1). The ACCORD BP results suggest that blood pressure targets more intensive than <140/90 mmHg are not likely to improve cardiovascular outcomes among most people with type 2 diabetes but may be reasonable for patients who may derive the most benefit and have been educated about added treatment burden, side effects, and costs, as discussed below.
Additional studies, such as the Systolic Blood Pressure Intervention Trial (SPRINT) and the Hypertension Optimal Treatment (HOT) trial, also examined effects of intensive versus standard control (Table 10.1), though the relevance of their results to people with diabetes is less clear. The Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation–Blood Pressure (ADVANCE BP) trial did not explicitly test blood pressure targets (29); the achieved blood pressure in the intervention group was higher than that achieved in the ACCORD BP intensive arm and would be consistent with a target blood pressure of <140/90 mmHg. Notably, ACCORD BP and SPRINT measured blood pressure using automated office blood pressure measurement, which yields values that are generally lower than typical office blood pressure readings by approximately 5–10 mmHg (30), suggesting that implementing the ACCORD BP or SPRINT protocols in an outpatient clinic might require a systolic blood pressure target higher than <120 mmHg, such as <130 mmHg.
A number of post hoc analyses have attempted to explain the apparently divergent results of ACCORD BP and SPRINT. Some investigators have argued that the divergent results are not due to differences between people with and without diabetes but rather are due to differences in study design or to characteristics other than diabetes (31-33). Others have opined that the divergent results are most readily explained by the lack of benefit of intensive blood pressure control on cardiovascular mortality in ACCORD BP, which may be due to differential mechanisms underlying cardiovascular disease in type 2 diabetes, to chance, or both (34).
Table 10.1—Randomized controlled trials of intensive versus standard hypertension treatment strategies

ACS, acute coronary syndrome; AKI, acute kidney injury; CVD, cardiovascular disease; HF, heart failure; MI, myocardial infarction; T2D, type 2 diabetes. Data from this table can also be found in the ADA position statement “Diabetes and Hypertension” (17).
2.3.3.0 Meta-analyses of Trials
To clarify optimal blood pressure targets in patients with diabetes, meta-analyses have stratified clinical trials by mean baseline blood pressure or mean blood pressure attained in the intervention (or intensive treatment) arm. Based on these analyses, antihypertensive treatment appears to be beneficial when mean baseline blood pressure is ≥140/90 mmHg or mean attained intensive blood pressure is ≥130/80 mmHg (17,21,22,24–26). Among trials with lower baseline or attained blood pressure, antihypertensive treatment reduced the risk of stroke, retinopathy, and albuminuria, but effects on other ASCVD outcomes and heart failure were not evident. Taken together, these meta-analyses consistently show that treating patients with baseline blood pressure ≥140 mmHg to targets <140 mmHg is beneficial, while more intensive targets may offer additional (though probably less robust) benefits.
2.3.4.0 Individualization of Treatment Targets
Patients and clinicians should engage in a shared decision-making process to determine individual blood pressure targets (17). This approach acknowledges that the benefits and risks of intensive blood pressure targets are uncertain and may vary across patients and is consistent with a patient-focused approach to care that values patient priorities and provider judgment (35). Secondary analyses of ACCORD BP and SPRINT suggest that clinical factors can help determine individuals more likely to benefit and less likely to be harmed by intensive blood pressure control (36).
Absolute benefit from blood pressure reduction correlated with absolute baseline cardiovascular risk in SPRINT and in earlier clinical trials conducted at higher baseline blood pressure levels (11,37). Extrapolation of these studies suggests that patients with diabetes may also be more likely to benefit from intensive blood pressure control when they have high absolute cardiovascular risk. Therefore, it may be reasonable to target blood pressure <130/80 mmHg among patients with diabetes and either clinically diagnosed cardiovascular disease (particularly stroke, which was significantly reduced in ACCORD BP) or 10-year ASCVD risk ≥15%, if it can be attained safely. This approach is consistent with guidelines from the American College of Cardiology/American Heart Association, which advocate a blood pressure target <130/80 mmHg for all patients, with or without diabetes (38).
Potential adverse effects of antihypertensive therapy (e.g., hypotension, syncope, falls, acute kidney injury, and electrolyte abnormalities) should also be taken into account (28,39–41). Patients with older age, chronic kidney disease, and frailty have been shown to be at higher risk of adverse effects of intensive blood pressure control (41). In addition, patients with orthostatic hypotension, substantial comorbidity, functional limitations, or polypharmacy may be at high risk of adverse effects, and some patients may prefer higher blood pressure targets to enhance quality of life. Patients with low absolute cardiovascular risk (10-year ASCVD risk <15%) or with a history of adverse effects of intensive blood pressure control or at high risk of such adverse effects should have a higher blood pressure target. In such patients, a blood pressure target of <140/90 mmHg is recommended, if it can be safely attained.
2.3.5.0 Pregnancy and Antihypertensive Medications
Since there is a lack of randomized controlled trials of antihypertensive therapy in pregnant women with diabetes, recommendations for the management of hypertension in pregnant women with diabetes should be similar to those for all pregnant women. The American College of Obstetricians and Gynecologists (ACOG) has recommended that women with mild to moderate gestational hypertension (systolic blood pressure <160 mmHg or diastolic blood pressure <110 mmHg) do not need to be treated with antihypertensive medications as there is no benefit identified that clearly outweighs potential risks of therapy (42). A 2014 Cochrane systematic review of antihypertensive therapy for mild to moderate chronic hypertension that included 49 trials and over 4,700 women did not find any conclusive evidence for or against blood pressure treatment to reduce the risk of preeclampsia for the mother or effects on perinatal outcomes such as preterm birth, small-for-gestational-age infants, or fetal death (43). For pregnant women who require antihypertensive therapy, systolic blood pressure levels of 120– 160 mmHg and diastolic blood pressure levels of 80–105 mmHg are suggested to optimize maternal health without risking fetal harm. Lower targets (systolic blood pressure 110–119 mmHg and diastolic blood pressure 65–79 mmHg) may contribute to improved long-term maternal health; however, they may be associated with impaired fetal growth. Pregnant women with hypertension and evidence of end-organ damage from cardiovascular and/or renal disease may be considered for lower blood pressure targets to avoid progression of these conditions during pregnancy.
During pregnancy, treatment with ACE inhibitors, angiotensin receptor blockers (ARBs), and spironolactone are contraindicated as they may cause fetal damage. Antihypertensive drugs known to be effective and safe in pregnancy include methyldopa, labetalol, and long-acting nifedipine, while hydralzine may be considered in the acute management of hypertension in pregnancy or severe preeclampsia (42). Diuretics are not recommended for blood pressure control in pregnancy but may be used during late-stage pregnancy if needed for volume control (42,44). ACOG also recommends that postpartum patients with gestational hypertension, preeclampsia, and superimposed preeclampsia have their blood pressures observed for 72 h in the hospital and for 7–10 days postpartum. Long-term follow-up is recommended for these women as they have increased life-time cardiovascular risk (45). See Section 14 “Management of Diabetes in Pregnancy” for additional information.
2.4.0.0 Treatment Strategies
2.4.1.0 Lifestyle Intervention
Recommendations
10.7 For patients with blood pressure >120/80 mmHg, lifestyle intervention consists of weight loss if overweight or obese, a Dietary Approaches to Stop Hypertension (DASH)-style dietary pattern including reducing sodium and increasing potassium intake, moderation of alcohol intake, and increased physical activity. B
Lifestyle management is an important component of hypertension treatment because it lowers blood pressure, enhances the effectiveness of some antihypertensive medications, promotes other aspects of metabolic and vascular health, and generally leads to few adverse effects. Lifestyle therapy consists of reducing excess body weight through caloric restriction, restricting sodium intake ( <2,300 mg/day), increasing consumption of fruits and vegetables (8–10 servings per day) and low-fat dairy products (2–3 servings per day), avoiding excessive alcohol consumption (no more than 2 servings per day in men and no more than 1 serving per day in women) (46), and increasing activity levels (47).
These lifestyle interventions are reasonable for individuals with diabetes and mildly elevated blood pressure (systolic >120 mmHg or diastolic >80 mmHg) and should be initiated along with pharmacologic therapy when hypertension is diagnosed (Fig. 10.1) (47). A lifestyle therapy plan should be developed in collaboration with the patient and discussed as part of diabetes management.
2.4.2.0 Pharmacologic Interventions
2.4.2.1 Recommendations
Recommendations
10.8 Patients with confirmed office-based blood pressure ≥140/90 mmHg should, in addition to lifestyle therapy, have prompt initiation and timely titration of pharmacologic therapy to achieve blood pressure goals. A
10.9 Patients with confirmed office-based blood pressure ≥160/ 100 mmHg should, in addition to lifestyle therapy, have prompt initiation and timely titration of two drugs or a single-pill combination of drugs demonstrated to reduce cardiovascular events in patients with diabetes. A
10.10 Treatment for hypertension should include drug classes demonstrated to reduce cardiovascular events in patients with diabetes (ACE inhibitors, angiotensin receptor blockers, thiazide-like diuretics, or dihydropyridine calcium channel blockers). A
10.11 Multiple-drug therapy is generally required to achieve blood pressure targets. However, combinations of ACE inhibitors and angiotensin receptor blockers and combinations of ACE inhibitors or angiotensin receptor blockers with direct renin inhibitors should not be used. A
10.12 An ACE inhibitor or angiotensin receptor blocker, at the maximum tolerated dose indicated for blood pressure treatment, is the recommended first-line treatment for hypertension in patients with diabetes and urinary albuminto-creatinine ratio ≥300 mg/g creatinine A or 30–299 mg/g creatinine. B If one class is not tolerated, the other should be substituted. B
10.13 For patients treated with an ACE inhibitor, angiotensin receptor blocker, or diuretic, serum creatinine/estimated glomerular filtration rate and serum potassium levels should be monitored at least annually. B
2.4.2.2 Initial Number of Antihypertensive Medications.
Initial treatment for people with diabetes depends on the severity ofhypertension (Fig. 10.1). Those with blood pressure between 140/90 mmHg and 159/99 mmHg may begin with a single drug. For patients with blood pressure ≥160/100 mmHg, initial pharmacologic treatment with two antihypertensive medications is recommended in order to more effectively achieve adequate blood pressure control (48-50). Single-pill antihypertensive combinations may improve medication adherence in some patients (51).
2.4.2.3 Classes of Antihypertensive Medications.
Initial treatment for hypertension should include any of the drug classes demonstrated to reduce cardiovascular events in patients with diabetes: ACE inhibitors (52,53), ARBs (52,53), thiazide-like diuretics (54), or dihydropyridine calcium channel blockers (55). For patients with albuminuria (urine albumin-to-creatinine ratio ≥30 mg/g), initial treatment should include an ACE inhibitor or ARB in order to reduce the risk of progressive kidney disease (17) (Fig. 10.1). In the absence of albuminuria, risk of progressive kidney disease is low, and ACE inhibitors and ARBs have not been found to afford superior cardioprotection when compared with thiazide-like diuretics or dihydropyridine calcium channel blockers (56). b-Blockers may be used for the treatment of prior MI, active angina, or heart failure but have not been shown to reduce mortality as blood pressure-lowering agents in the absence of these conditions (23,57).
2.4.2.4 Multiple-Drug Therapy.
Multiple-drug therapy is often required to achieve blood pressure targets (Fig. 10.1), particularly in the setting of diabetic kidney disease. However, the use of both ACE inhibitors and ARBs in combination, or the combination of an ACE inhibitor or ARB and a directrenininhibitor, isnotrecommended given the lack of added ASCVD benefit and increased rate of adverse eventsdnamely, hyperkalemia, syncope, and acute kidney injury (AKI) (58-60). Titration of and/or addition of further blood pressure medications should be made in a timely fashion to overcome clinical inertia in achieving blood pressure targets.
2.4.2.5 Bedtime Dosing.
Growing evidence suggests that there is an association between the absence of nocturnal blood pressure dipping and the incidence of ASCVD. A meta-analysis of randomized clinical trials found a small benefit of evening versus morning dosing of antihypertensive medications with regard toblood pressure control but had no data on clinical effects (61). In two subgroup analyses of a single subsequent randomized controlled trial, moving at least one antihypertensive medication to bedtime significantly reduced cardiovascular events, but results were based on a small number of events (62).
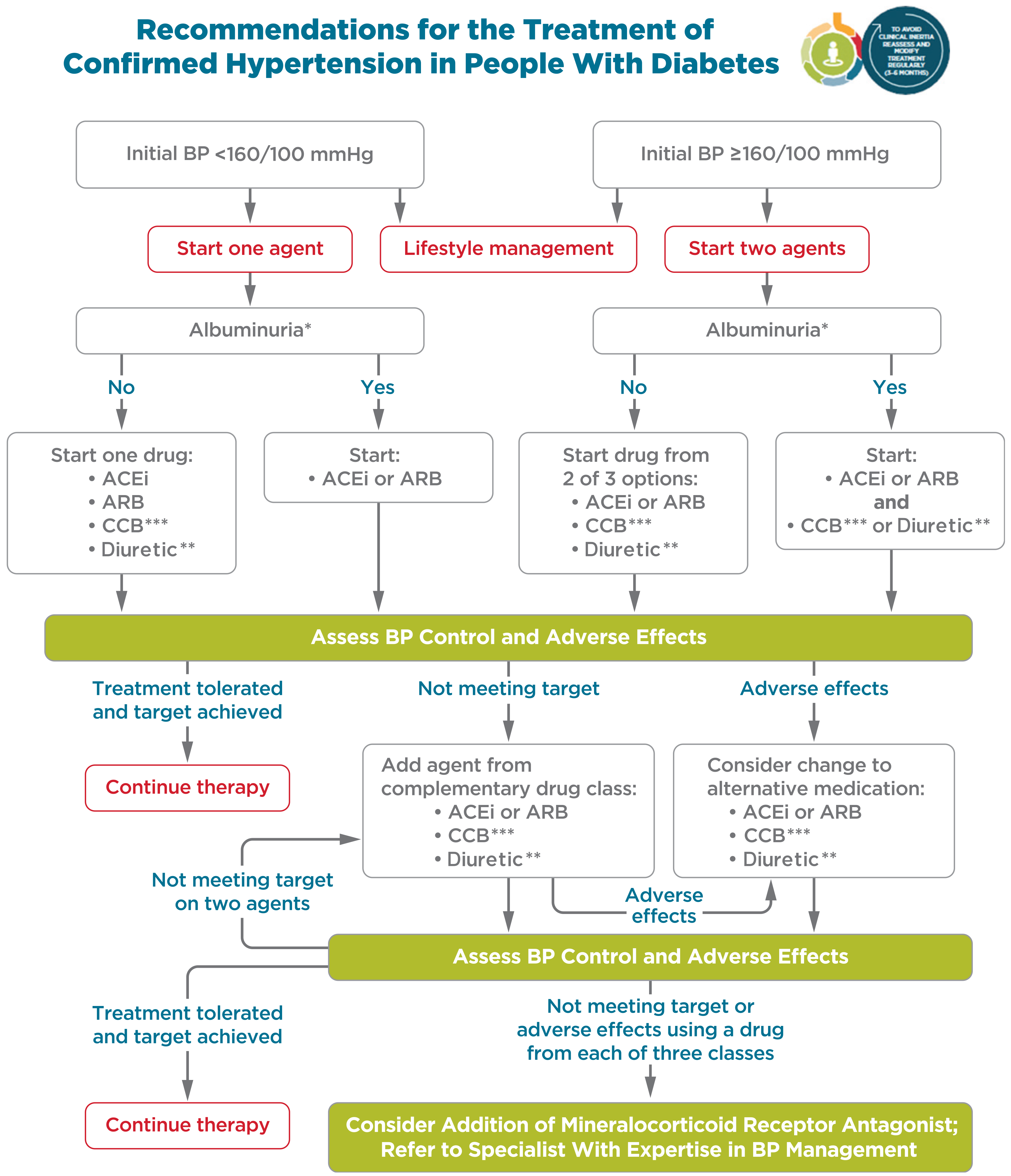
Figure 10.1—Recommendations for the treatment of confirmed hypertension in people with diabetes. *An ACE inhibitor (ACEi) or angiotensin receptor blocker (ARB) is suggested to treat hypertension for patients with urine albumin-to-creatinine ratio 30–299 mg/g creatinine and strongly recommended for patients with urine albumin-to-creatinine ratio ≥300 mg/g creatinine. **Thiazide-like diuretic; long-acting agents shown to reduce cardiovascular events, such as chlorthalidone and indapamide, are preferred. ***Dihydropyridine calcium channel blocker (CCB). BP, blood pressure. Adapted from de Boer et al. (17).
2.4.3.0 Hyperkalemia and Acute Kidney Injury.
Treatment with ACE inhibitors or ARBs can cause AKI and hyperkalemia, while diuretics can cause AKI and either hypokalemia or hyperkalemia (depending on mechanism of action) (63,64). Detection and management of these abnormalities is important because AKI and hyperkalemia each increase the risks of cardiovascular events and death (65). Therefore, serum creatinine and potassium should be monitored during treatment with an ACE inhibitor, ARB, or diuretic, particularly among patients with reduced glomerular filtration who are at increased risk of hyperkalemia and AKI (63,64,66).
2.4.4.0 Resistant Hypertension
Recommendation
10.14 Patients with hypertension who are not meeting blood pressure targets on three classes of antihypertensive medications (including a diuretic) should be considered for mineralocorticoid receptor antagonist therapy. B
Resistant hypertension is defined as blood pressure ≥140/90 mmHg despite a therapeutic strategy that includes appropriate lifestyle management plus a diuretic and two other antihypertensive drugs belonging to different classes at adequate doses. Prior to diagnosing resistant hypertension, a number of other conditions should be excluded, including medication nonadherence, white coat hypertension, and secondary hypertension. In general, barriers to medication adherence (such as cost and side effects) should be identified and addressed (Fig. 10.1). Mineralocorticoid receptor antagonists are effective for management of resistant hypertension in patients with type 2 diabetes when added to existing treatment with an ACE inhibitor or ARB, thiazide-like diuretic, and dihydropyridine calcium channel blocker (67). Mineralocorticoid receptor antagonists also reduce albuminuria and have additional cardiovascular benefits (68-71). However, adding a mineralocorticoidreceptorantagonisttoa regimen including an ACE inhibitor or ARB may increase the risk for hyperkalemia, emphasizing the importance of regular monitoring for serum creatinine and potassium in these patients, and long-term outcome studies are needed to better evaluate the role of mineralocorticoid receptor antagonists in blood pressure management.
3.0.0.0 LIPID MANAGEMENT
3.1.0.0 Lifestyle Intervention
Recommendations
10.15 Lifestyle modification focusing on weight loss (if indicated); application of a Mediterranean diet or Dietary Approaches to Stop Hypertension (DASH) dietary pattern; reduction of saturated fat and trans fat; increase of dietary n-3 fatty acids, viscous fiber, and plant stanols/ sterols intake; and increased physical activity should be recommended to improve the lipid profile and reduce the risk of developing atherosclerotic cardiovascular disease in patients with diabetes. A
10.16 Intensify lifestyle therapy and optimize glycemic control for patients with elevated triglyceride levels (≥150 mg/dL [1.7 mmol/L]) and/or low HDL cholesterol (,40 mg/dL [1.0 mmol/L] for men, <50 mg/dL [1.3 mmol/L] for women). C
Lifestyle intervention, including weight loss (72), increased physical activity, and medical nutrition therapy, allows some patients to reduce ASCVD risk factors. Nutrition intervention should be tailored according to each patient’s age, diabetes type, pharmacologic treatment, lipid levels, and medical conditions.
Recommendations should focus on application of a Mediterranean diet (73) or Dietary Approaches to Stop Hypertension (DASH) dietary pattern, reducing saturated and trans fat intake and increasing plant stanols/sterols, n-3 fatty acids, and viscous fiber (such as in oats, legumes, and citrus) intake (74). Glycemic control may also beneficially modify plasma lipid levels, particularly in patients with very high triglycerides and poor glycemic control. See Section 5 “Lifestyle Management” for additional nutrition information.
3.2.0.0 Ongoing Therapy and Monitoring With Lipid Panel
Recommendations
10.17 In adults not taking statins or other lipid-lowering therapy, it is reasonable to obtain a lipid profile at the time of diabetes diagnosis, at an initial medical evaluation, and every 5 years thereafter if under the age of 40 years, or more frequently if indicated. E
10.18 Obtain a lipid profile at initiation of statins or other lipidlowering therapy, 4–12 weeks after initiation or a change in dose, and annually thereafter as it may help to monitor the response to therapy and inform medication adherence. E
In adults with diabetes, it is reasonable to obtain a lipid profile (total cholesterol, LDL cholesterol, HDL cholesterol, and triglycerides) at the time of diagnosis, at the initial medical evaluation, and at least every 5 years thereafter in patients under the age of 40 years. In younger patients with longer duration of disease (such as those with youth-onset type 1 diabetes), more frequent lipid profiles may be reasonable. A lipid panel should also be obtained immediately before initiating statin therapy. Once a patient is taking a statin, LDL cholesterol levels should be assessed 4–12 weeks after initiation of statin therapy, after any change in dose, and on an individual basis (e.g., to monitor for medication adherence and efficacy). If LDL cholesterol levels are not responding in spite of medication adherence, clinical judgment is recommended to determine the need for and timing of lipid panels. In individual patients, the highly variable LDL cholesterol–lowering response seen with statins is poorly understood (75). Clinicians should attempt to find a dose or alternative statin that is tolerable if side effects occur. There is evidence for benefit from even extremely low, less than daily statin doses (76).
3.3.0.0 Statin Treatment
3.3.1.0 Recommendations
Recommendations
10.19 For patients of all ages with diabetes and atherosclerotic cardiovascular disease or 10- year atherosclerotic cardiovascular disease risk >20%, high-intensity statin therapy should be added to lifestyle therapy. A
10.20 For patients with diabetes aged <40 years with additional atherosclerotic cardiovascular disease risk factors, the patient and provider should consider using moderate-intensity statin in addition to lifestyle therapy. C
10.21 For patients with diabetes aged 40–75 years A and >75 years B without atherosclerotic cardiovascular disease, use moderate-intensity statin in addition to lifestyle therapy.
10.22 In patients with diabetes who have multiple atherosclerotic cardiovascular disease risk factors, it is reasonable to consider high-intensity statin therapy. C
10.23 For patients who do not tolerate the intended intensity, the maximally tolerated statin dose should be used. E
10.24 For patients with diabetes and atherosclerotic cardiovascular disease, if LDL cholesterol is ≥70 mg/dL on maximally tolerated statin dose, consider adding additional LDL-lowering therapy (such as ezetimibe or PCSK9 inhibitor). A Ezetimibe may be preferred due to lower cost.
10.25 Statin therapy is contraindicated in pregnancy. B
3.3.2.0 Initiating Statin Therapy Based on Risk
Patients with type 2 diabetes have an increased prevalence of lipid abnormalities, contributing to their high risk of ASCVD. Multiple clinical trials have demonstrated the beneficial effects of statin therapy on ASCVD outcomes in subjects with and without CHD (77,78). Subgroup analyses of patients with diabetes in larger trials (79-83) and trials in patients with diabetes (84,85) showed significant primary and secondary prevention of ASCVD events and CHD death in patients with diabetes. Meta-analyses, including data from over 18,000 patients with diabetesfrom 14 randomized trials of statin therapy (mean follow-up 4.3 years), demonstrate a 9% proportional reduction in all-cause mortality and 13% reduction in vascular mortality for each mmol/L (39 mg/dL) reduction in LDL cholesterol (86). Accordingly, statins are the drugs of choice for LDL cholesterol lowering and cardioprotection. Table 10.2 shows recommended lipid-lowering strategies, and Table 10.3 shows the two statin dosing intensities that are recommended for use in clinical practice: high-intensity statin therapy will achieve approximately a 50% reduction in LDL cholesterol, and moderate-intensity statin regimens achieve 30–50% reductions in LDL cholesterol. Low-dose statin therapy is generally not recommended in patients with diabetes but is sometimes the only dose of statin that a patient can tolerate. For patients who do nottolerate the intended intensity of statin, the maximally tolerated statin dose should be used.
As in those without diabetes, absolute reductions in ASCVD outcomes (CHD death and nonfatal MI) are greatest in people with high baseline ASCVD risk (known ASCVD and/or very high LDL cholesterol levels), but the overall bene- fits of statin therapy in people with diabetes at moderate or even low risk for ASCVDareconvincing (87,88). The relative benefit of lipid-lowering therapy has been uniform across most subgroups tested (78,86), including subgroups that varied with respect to age and other risk factors.
Table 10.2—Recommendations for statin and combination treatment in adults with diabetes
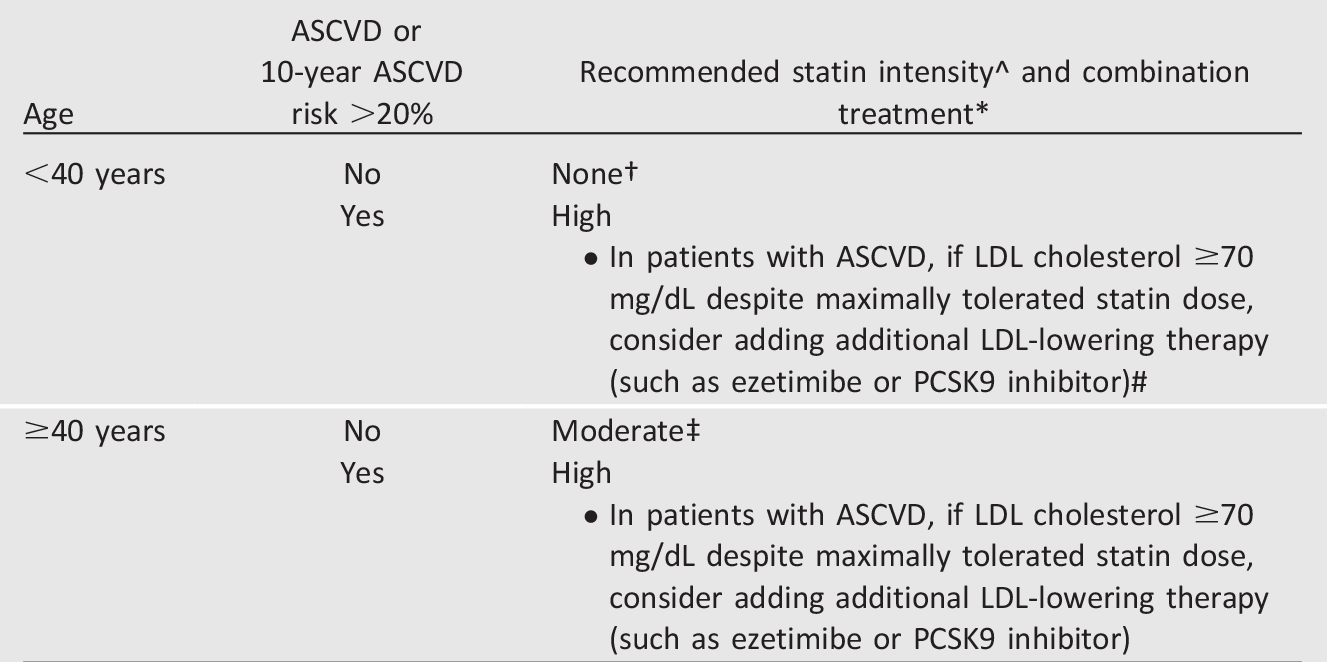
ASCVD, atherosclerotic cardiovascular disease; PCSK9, proprotein convertase subtilisin/kexin type 9. *In addition to lifestyle therapy. ^For patients who do not tolerate the intended intensity of statin, the maximally tolerated statin dose should be used. †Moderate-intensity statin may be considered based on risk-benefit profile and presence of ASCVD risk factors. ASCVD risk factors include LDL cholesterol ≥100 mg/dL (2.6 mmol/L), high blood pressure, smoking, chronic kidney disease, albuminuria, and family history of premature ASCVD. ‡High-intensity statin may be considered based on risk-benefit profile and presence of ASCVD risk factors. #Adults aged <40 years with prevalent ASCVD were not well represented in clinical trials of non-statin–based LDL reduction. Before initiating combination lipid-lowering therapy, consider the potential for further ASCVD risk reduction, drug-specific adverse effects, and patient preferences.
Table 10.3—High-intensity and moderate-intensity statin therapy*
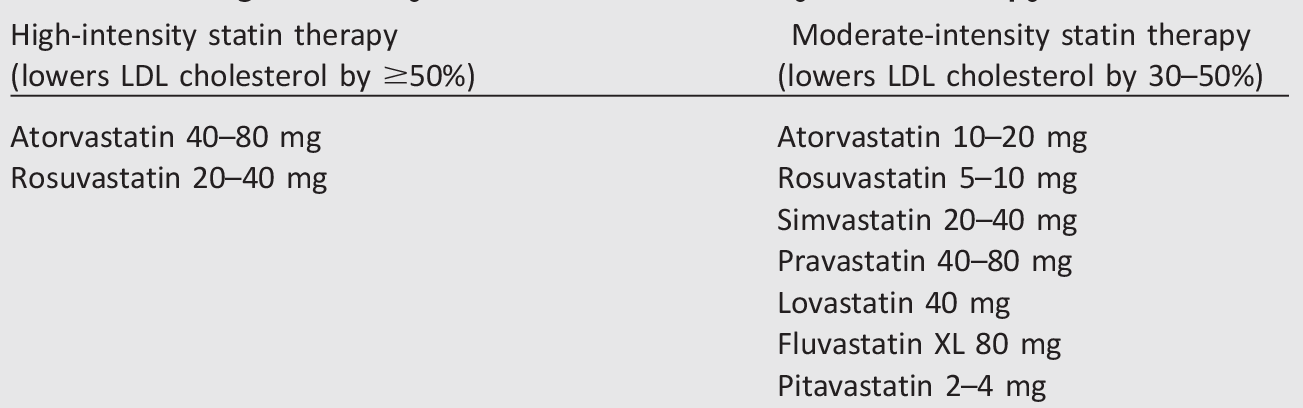
*Once-daily dosing. XL, extended release.
3.3.3.0 Primary Prevention (Patients Without ASCVD)
3.3.3.1 Primary Prevention (Patients Without ASCVD)
For primary prevention, moderate-dose statin therapy is recommended for those 40 years and older (80,87,88), though high-intensity therapy may be considered on an individual basis in the context of additional ASCVD risk factors. The evidence is strong for patients with diabetes aged 40–75 years, an age-group well represented in statin trials showing benefit. Since risk is enhanced in patients with diabetes, as noted above, patients who also have multiple other coronary risk factors have increased risk, equivalent to that of those with ASCVD. As such, recent guidelines recommend that in patients with diabetes who have multiple ASCVD risk factors, it is reasonable to prescribe high-intensity statin therapy (12,89). Furthermore, for patients with diabetes whose ASCVD risk is >20%, i.e., an ASCVD risk equivalent, the same high-intensity statin therapy is recommended asforthosewithdocumented ASCVD(12). The evidence is lower for patients aged >75 years; relatively few older patients with diabetes have been enrolled in primary prevention trials. However, heterogeneity by age has not been seen in the relative benefit of lipid-lowering therapy in trials that included older participants (78,85,86), and because older age confers higher risk, the absolute benefits are actually greater (78,90). Moderate-intensitystatintherapy is recommended in patients with diabetes that are 75 years or older. However, the risk-benefit profile should be routinely evaluated in this population, with downward titration of dose performed as needed. See Section 12 “Older Adults” for more details on clinical considerations for this population.
3.3.3.2 Age <40 Years and/or Type 1 Diabetes.
Very little clinical trial evidence exists for patients with type 2 diabetes under the age of 40 years or for patients with type 1 diabetes of any age. For pediatric recommendations, see Section 13 “Children and Adolescents.” In the Heart Protection Study (lower age limit 40 years), the subgroup of ;600 patients with type 1 diabetes had a proportionately similar, although not statistically significant, reduction in risk as patients with type 2 diabetes (80). Even though the data are not definitive, similar statin treatment approaches should be considered for patients with type 1 or type 2 diabetes, particularly in the presence of other cardiovascular risk factors. Patients below the age of 40 have lower risk of developing a cardiovascular event over a 10-year horizon; however, their lifetime risk of developing cardiovascular disease and suffering an MI, stroke, or cardiovascular death is high. For patients under the age of 40 years and/or who have type 1 diabetes with other ASCVD risk factors, we recommend that the patient and health care provider discuss the relative benefits and risks and consider the use of moderate-intensity statin therapy. Please refer to “Type 1 Diabetes Mellitus and Cardiovascular Disease: A Scientific Statement From the American Heart Association and American Diabetes Association” (91) for additional discussion.
3.3.4.0 Secondary Prevention (Patients With ASCVD)
Because risk is high in patients with ASCVD, intensive therapy is indicated and has been shown to be of benefit in multiple large randomized cardiovascular outcomes trials (86,90,92,93). High-intensity statin therapy is recommended for all patients with diabetes and ASCVD. This recommendation is based on the Cholesterol Treatment Trialists’ Collaboration involving 26 statin trials, of which 5 compared high-intensity versus moderate-intensity statins. Together, they found reductions in nonfatal cardiovascular events with more intensive therapy, in patients with and without diabetes (78,82,92).
Over the past few years, there have been multiple large randomized trials investigating the benefits of adding nonstatin agents to statin therapy, including those that evaluated further lowering of LDL cholesterol with ezetimibe (90,94) and proprotein convertase subtilisin/ kexin type 9 (PCSK9) inhibitors (93). Each trial found a significant benefit in the reduction of ASCVD events that was directly related to the degree of further LDL cholesterol lowering. These large trials included a significant number of participants with diabetes. For patients with ASCVD who are on high-intensity (and maximally tolerated) statin therapy and have an LDL cholesterol ≥70 mg/dL, the addition of nonstatin LDL-lowering therapy is recommended following a clinician-patient discussion about the net benefit, safety, and cost (Table 10.2).
3.4.0.0 Combination Therapy for LDL Cholesterol Lowering
3.4.1.0 Statins and Ezetimibe
The IMProved Reduction of Outcomes: Vytorin Efficacy International Trial (IMPROVE-IT) was a randomized controlled trial in 18,144 patients comparing the addition of ezetimibe to simvastatin therapy versus simvastatin alone. Individuals were ≥50 years of age, had experienced a recent acute coronary syndrome (ACS), and were treated for an averageof 6 years. Overall, theaddition of ezetimibe led to a 6.4% relative benefit and a 2% absolute reduction in major adverse cardiovascular events, with the degree of benefit being directly proportional to the change in LDL cholesterol, which was 70 mg/dL in the statin group on average and 54 mg/dL in the combination group (90). In those with diabetes (27% of participants), the combination of moderate-intensity simvastatin (40 mg) and ezetimibe (10 mg) showed a significant reduction of major adverse cardiovascular events with an absolute risk reduction of 5% (40% vs. 45% cumulative incidence at 7 years) and relative risk reduction of 14% (hazard ratio [HR] 0.86 [95% CI 0.78–0.94]) over moderate-intensity simvastatin (40 mg) alone (94).
3.4.2.0 Statins and PCSK9 Inhibitors
Placebo-controlled trials evaluating the addition of the PCSK9 inhibitors evolocumab and alirocumab to maximally tolerated doses of statin therapy in participants who were at high risk for ASCVD demonstrated an average reduction in LDL cholesterol ranging from 36% to 59%. These agents have been approved as adjunctive therapy for patients with ASCVD or familial hypercholesterolemia who are receiving maximally tolerated statin therapy but require additional lowering of LDL cholesterol (95,96).
The effects of PCSK9 inhibition on ASCVD outcomes was investigated in the Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk (FOURIER) trial, which enrolled 27,564 patients with prior ASCVD and an additional high-risk feature who were receiving their maximally tolerated statin therapy (two-thirds were on high-intensity statin) but who still had an LDL cholesterol ≥70 mg/dL or a non-HDL cholesterol ≥100 mg/dL (93). Patients were randomized to receive subcutaneous injections of evolocumab (either 140 mg every 2 weeks or 420 mg every month based on patient preference) versus placebo. Evolocumab reduced LDL cholesterol by 59% from a median of 92 to 30 mg/dL in the treatment arm.
During the median follow-up of 2.2 years, the composite outcome of cardiovascular death, MI, stroke, hospitalization for angina, or revascularization occurred in 11.3% vs. 9.8% of the placebo and evolocumab groups, respectively, representing a 15% relative risk reduction (P <0.001). The combined end point of cardiovascular death, MI, or stroke was reduced by 20%, from 7.4% to 5.9% (P <0.001). Importantly, similar benefits were seen in prespecified subgroup of patients with diabetes, comprising 11,031 patients (40% of the trial) (97).
3.5.0.0 Treatment of Other Lipoprotein Fractions or Targets
Recommendations
10.26 For patients with fasting triglyceride levels ≥500 mg/dL (5.7 mmol/L), evaluate for secondary causes of hypertriglyceridemia and consider medical therapy to reduce the risk of pancreatitis. C
10.27 In adults with moderate hypertriglyceridemia (fasting or nonfasting triglycerides 175–499 mg/dL), clinicians should address and treat lifestyle factors (obesity and metabolic syndrome), secondary factors (diabetes, chronic liver or kidney disease and/or nephrotic syndrome, hypothyroidism), and medications that raise triglycerides. C
Hypertriglyceridemia should be addressed with dietary and lifestyle changes including weight loss and abstinence from alcohol (98). Severe hypertriglyceridemia (fasting triglycerides ≥500 mg/dL and especially >1,000 mg/dL) may warrant pharmacologic therapy (fibric acid derivatives and/or fish oil) to reduce the risk of acute pancreatitis. In addition, if 10-year ASCVD risk is ≥7.5%, it is reasonable to initiate moderate-intensity statin therapy or increase statin intensity from moderate to high. In patients with moderate hypertriglyceridemia, lifestyle interventions, treatment of secondary factors, and avoidance of medications that might raise triglycerides are recommended.
Low levels of HDL cholesterol, often associated with elevated triglyceride levels, are the most prevalent pattern of dyslipidemia in individuals with type 2 diabetes. However, the evidence for the use of drugs that target these lipid fractions is substantially less robust than that for statin therapy (99). In a large trial in patients with diabetes, fenofibrate failed to reduce overall cardiovascular outcomes (100).
3.6.0.0 Other Combination Therapy
3.6.1.0 Recommendations
Recommendations
10.28 Combination therapy (statin/ fibrate) has not been shown to improve atherosclerotic cardio- vascular disease outcomes and is generally not recommended. A
10.29 Combination therapy (statin/ niacin) has not been shown to provide additional cardiovascular benefit above statin therapy alone, may increase the risk of stroke with additional sideeffects, and is generallynot recommended. A
3.6.2.0 Statin and Fibrate
Combination therapy (statin and fibrate) is associated with an increased risk for abnormal transaminase levels, myositis, and rhabdomyolysis. The risk of rhabdomyolysis is more common with higher doses of statins and renal insufficiency and appears tobe higher when statins are combined with gemfibrozil (compared with fenofibrate) (101).
In the ACCORD study, in patients with type 2 diabetes who were at high risk for ASCVD, the combination of fenofibrate and simvastatin did not reduce the rate of fatal cardiovascular events, nonfatal MI, or nonfatal stroke as compared with simvastatin alone. Prespecified subgroup analyses suggested heterogeneity in treatment effects with possible benefit for men with both a triglyceride level ≥204 mg/dL (2.3 mmol/L) and an HDL cholesterol level ≤34 mg/dL (0.9 mmol/L) (102). A prospective trial of a newer fibrate in this specific population of patients is ongoing (103).
3.6.3.0 Statin and Niacin
The Atherothrombosis Intervention in Metabolic Syndrome With Low HDL/ High Triglycerides: Impact on Global Health Outcomes (AIM-HIGH) trial randomized over 3,000 patients (about one-third with diabetes) with established ASCVD, low LDL cholesterol levels (<180 mg/dL [4.7 mmol/L]), low HDL cholesterol levels (men <40 mg/dL [1.0 mmol/L] and women <50 mg/dL [1.3 mmol/L]), and triglyceride levels of 150–400 mg/dL (1.7–4.5 mmol/L) to statin therapy plus extended-release niacin or placebo. The trial was halted early due to lack of efficacy on the primary ASCVD outcome (first event of the composite of death from CHD, nonfatal MI, ischemic stroke, hospitalization for an ACS, or symptom-driven coronary or cerebral revascularization) and a possible increase in ischemic stroke in those on combination therapy (104).
The much larger Heart Protection Study 2–Treatment of HDL to Reduce the Incidence of Vascular Events (HPS2- THRIVE) trial also failed to show a benefit of adding niacin to background statin therapy (105). A total of 25,673 patients with prior vascular disease were randomized to receive 2 g of extended-release niacin and 40 mg of laropiprant (an antagonist of the prostaglandin D2 receptor DP1 that has been shown to improve adherence to niacin therapy) versus a matching placebo daily and followed for a median follow-up period of 3.9 years. There was no significant difference in the rate of coronary death, MI, stroke, or coronary revascularization with the addition of niacin–laropiprant versus placebo (13.2% vs. 13.7%; rate ratio 0.96; P = 0.29). Niacin–laropiprant was associated with an increased incidence of new-onset diabetes (absolute excess, 1.3 percentage points; P <0.001) and disturbances in diabetes control among those with diabetes. In addition, there was an increase in serious adverse events associated with the gastrointestinal system, musculoskeletal system, skin, and, unexpectedly, infection and bleeding.
Therefore, combination therapy with a statin and niacin is not recommended given the lack of efficacy on major ASCVD outcomes and increased side effects.
3.7.0.0 Diabetes With Statin Use
Several studies have reported a modestly increased risk of incident diabetes with statin use (106,107), which may be limited to those with diabetes risk factors. An analysis of one of the initial studies suggested that although statin use was associated with diabetes risk, the cardiovascular event rate reduction with statins far outweighed the risk of incident diabetes even for patients at highest risk for diabetes (108). The absolute risk increase was small (over 5 years of follow-up, 1.2% of participants on placebo developed diabetes and 1.5% on rosuvastatin developed diabetes) (108). A meta-analysis of 13 randomized statin trials with 91,140 participants showed an odds ratio of 1.09 for a new diagnosis of diabetes, so that (on average) treatment of 255 patients with statins for 4 years resulted in one additional case of diabetes while simultaneously preventing 5.4 vascular events among those 255 patients (107).
3.8.0.0 Lipid-Lowering Agents and Cognitive Function
Although this issue has been raised, several lines of evidence point against this association, as detailed in a 2018 European Atherosclerosis Society Consensus Panel statement (109). First, there are three large randomized trials of statin versus placebo where specific cognitive tests were performed, and no differences were seen between statin and placebo (110-113). In addition, no change in cognitive function has been reported in studies with the addition of ezetimibe (90) or PCSK9 inhibitors (93,114) to statin therapy, including among patients treated to very low LDL cholesterol levels. In addition, the most recent systematic review of the U.S. Food and Drug Administration’s (FDA’s) postmarketing surveillance databases, randomized controlled trials, and cohort, casecontrol, and cross-sectional studies evaluating cognition in patients receiving statins found that published data do not reveal an adverse effect of statins on cognition (115). Therefore, a concern that statins or other lipid-lowering agents might cause cognitive dysfunction or dementia is not currently supported by evidence and should not deter their use in individuals with diabetes at high risk for ASCVD (115).
4.0.0.0 ANTIPLATELET AGENTS
4.1.0.0 Recommendations
Recommendations
10.30 Use aspirin therapy (75–162 mg/day) as a secondary prevention strategy in those with diabetes and a history of atherosclerotic cardiovascular disease. A
10.31 For patients with atherosclerotic cardiovascular disease and documented aspirin allergy, clopidogrel (75 mg/day) should be used. B
10.32 Dual antiplatelet therapy (with low-dose aspirin and a P2Y12 inhibitor) is reasonable for a year after an acute coronary syndrome A and may have benefits beyond this period. B
10.33 Aspirin therapy (75–162 mg/day) may be considered as a primary prevention strategy in those with diabetes who are at increased cardiovascular risk, after a discussion with the patient on the benefits versus increased risk of bleeding. C
4.2.0.0 Risk Reduction
Aspirin has been shown to be effective in reducing cardiovascular morbidity and mortality in high-risk patients with previous MI or stroke (secondary prevention) and is strongly recommended. In primary prevention, however, among patients with no previous cardiovascular events, its net benefit is more controversial (116,117).
Previous randomized controlled trials of aspirin specifically in patients with diabetes failed to consistently show a significant reduction in overall ASCVD end points, raising questions about the ef- ficacy of aspirin for primary prevention in people with diabetes, although some sex differences were suggested (118-120).
The Antithrombotic Trialists’ Collaboration published an individual patient– level meta-analysis (116) of the six large trials of aspirin for primary prevention in the general population. These trials collectively enrolled over 95,000 participants, including almost 4,000 with diabetes. Overall, they found that aspirin reduced the risk of serious vascular events by 12% (RR 0.88 [95% CI 0.82–0.94]). The largest reduction was for nonfatal MI, with little effect on CHD death (RR 0.95 [95% CI 0.78–1.15]) or total stroke. Most recently, the ASCEND (A Study of Cardiovascular Events iN Diabetes) trial randomized 15,480 patients with diabetes but no evident cardiovascular disease to aspirin 100 mg daily or placebo (121). The primary efficacy end point was vascular death, MI, or stroke or transient ischemic attack. The primary safety outcome was major bleeding (i.e., intracranial hemorrhage, sight-threatening bleeding in the eye, gastrointestinal bleeding, or other serious bleeding). During a mean follow-up of 7.4 years, there was a significant 12% reduction in the primary efficacy end point (8.5% vs. 9.6%; P = 0.01). In contrast, major bleeding was significantly increased from 3.2% to 4.1% in the aspirin group (rate ratio 1.29; P = 0.003), with most of the excess being gastrointestinal bleeding and other extra-cranial bleeding. There were no significant differences by sex, weight, or duration of diabetes or other baseline factors including ASCVD risk score.
Two other large randomized trials of aspirin for primary prevention, in patients without diabetes (ARRIVE [Aspirin to Reduce Risk of Initial Vascular Events]) (122) and in the elderly (ASPREE [Aspirin in Reducing Events in the Elderly]) (123), including 11% with diabetes, found no benefit of aspirin on the primary efficacy end point and an increased risk of bleeding. In ARRIVE, with 12,546 patients over a period of 60 months follow-up, the primary end point occurred in 4.29% vs. 4.48% of patients in the aspirin versus placebo groups (HR 0.96; 95% CI 0.81– 1.13; P = 0.60). Gastrointestinal bleeding events (characterized as mild) occurred in 0.97% of patients in the aspirin group vs. 0.46% in the placebo group (HR 2.11; 95% CI 1.36–3.28; P = 0.0007). In ASPREE, including 19,114 persons, for the rate of cardiovascular disease (fatal CHD, MI, stroke, or hospitalization for heart failure) after a median of 4.7 years of follow-up, the rates per 1,000 person-years were 10.7 vs. 11.3 events in aspirin vs. placebo groups (HR 0.95; 95% CI 0.83–1.08). The rate of major hemorrhage per 1,000 person-years was 8.6 events vs. 6.2 events, respectively (HR 1.38; 95% CI 1.18–1.62; P <0.001).
Thus, aspirin appears to have a modest effect on ischemic vascular events, with the absolute decrease in events depending on the underlying ASCVD risk. The main adverse effect is an increased risk of gastrointestinal bleeding. The excess risk may be as high as 5 per 1,000 per year in real-world settings. However, for adults with ASCVD risk >1% per year, the number of ASCVD events prevented will be similar to the number of episodes of bleeding induced, although these complications do not have equal effects on long-term health (124).
4.3.0.0 Treatment Considerations
In 2010, a position statement of the ADA, the American Heart Association, and the American College of Cardiology Foundation recommended that low-dose (75–162 mg/day) aspirin for primary prevention is reasonable for adults with diabetes and no previous history of vascular disease who are at increased ASCVD risk and who are not at increased risk for bleeding (125). These recommendations for using aspirin as primary prevention include both men and women aged ≥50 years with diabetes and at least one additional major risk factor (family history of premature ASCVD, hypertension, dyslipidemia, smoking, or chronic kidney disease/albuminuria) who are not at increased risk of bleeding (e.g., older age, anemia, renal disease) (126-129). Non-invasive imaging techniques such as coronary computed tomography angiography may potentially help further tailor aspirin therapy, particularly in those at low risk (130), but are not generally recommended. For patients over the age of 70 years (with or without diabetes), the balance appears to have greater risk than benefit (121,123). Thus, for primary prevention, the use of aspirin needs to be carefully considered and may generally not be recommended. Aspirin may be considered in the context of high cardiovascular risk with low bleeding risk, but generally not in older adults. For patients with documented ASCVD, use of aspirin for secondary prevention has far greater benefit than risk; for this indication, aspirin is still recommended (116).
4.4.0.0 Aspirin Use in People <50 Years of Age
Aspirin is not recommended for those at low risk of ASCVD (such as men and women aged <50 years with diabetes with no other major ASCVD risk factors) as the low benefit is likely to be out-weighed by the risks of bleeding. Clinical judgment should be used for those at intermediate risk (younger patients with one or more risk factors or older patients with no risk factors) until further research is available. Patients’ willingness to undergo long-term aspirin therapy should also be considered (131). Aspirin use in patients aged <21 years is generally contraindicated due to the associated risk of Reye syndrome.
4.5.0.0 Aspirin Dosing
Average daily dosages used in most clinical trials involving patients with diabetes ranged from 50 mg to 650 mg but were mostly in the range of 100–325 mg/day. There is little evidence tosupport any specific dose, but using the lowest possible dose may help to reduce side effects (132). In the U.S., the most common low-dose tablet is 81 mg. Although platelets from patients with diabetes have altered function, it is unclear what, if any, effect that finding has on the required dose of aspirin for cardioprotective effects in the patient with diabetes. Many alternate pathways for platelet activation exist that are independent of thromboxane A2 and thus are not sensitive to the effects of aspirin (133). “Aspirin resistance” has been described in patients with diabetes when measured by a variety of ex vivo and in vitro methods (platelet aggregometry, measurement of thromboxane B2) (134), but other studies suggest no impairment in aspirin response among patients with diabetes (135). A recent trial suggested that more frequent dosing regimens of aspirin may reduce platelet reactivity in individuals with diabetes (136); however, these observations alone are insufficient to empirically recommend that higher doses of aspirin be used in this group at this time. Another recent metaanalysis raised the hypothesis that low-dose aspirin efficacy is reduced in those weighing more than 70kg (137); however, the ASCEND trial found benefit of low dose aspirin in those in this weight range, which would thus not validate this suggested hypothesis (121). It appears that 75–162 mg/day is optimal.
4.6.0.0 Indications for P2Y12 Receptor Antagonist Use
A P2Y12 receptor antagonist in combination with aspirin should be used for at least 1 year in patients following an ACS and may have benefits beyond this period. Evidence supports use of either ticagrelor or clopidogrel if no percutaneous coronary intervention was performed and clopidogrel, ticagrelor, or prasugrel if a percutaneous coronary intervention was performed (138). In patients with diabetes and prior MI (1–3 years before), adding ticagrelor to aspirin significantly reduces the risk of recurrent ischemic events including cardiovascular and CHD death (139).
5.0.0.0 CARDIOVASCULAR DISEASE
5.1.0.0 Recommendations
Recommendations
Screening
10.34 In asymptomatic patients, routine screening for coronary artery disease is not recommended as it does not improve outcomes as long as atherosclerotic cardiovascular disease risk factors are treated. A
10.35 Consider investigations for coronary artery disease in the presence of any of the following: atypical cardiac symptoms (e.g., unexplained dyspnea, chest discomfort); signs or symptoms of associated vascular disease including carotid bruits, transient ischemic attack, stroke, claudication, or peripheral arterial disease; or electrocardiogram abnormalities (e.g., Q waves). E
Treatment
10.36 In patients with known atherosclerotic cardiovascular disease, consider ACE inhibitor or angiotensin receptor blocker therapy to reduce the risk of cardiovascular events. B
10.37 In patients with prior myocardial infarction, b-blockers should be continued for at least 2 years after the event. B
10.38 In patients with type 2 diabetes with stable congestive heart failure, metformin may be used if estimated glomerular filtration rate remains >30 mL/min but should be avoided in unstable or hospitalized patients with congestive heart failure. B
10.39 Among patients with type 2 diabetes who have established atherosclerotic cardiovascular disease, sodium–glucose cotransporter 2 inhibitors or glucagon-like peptide 1 receptor agonists with demonstrated cardiovascular disease benefit (Table 9.1) are recommended as part of the antihyperglycemic regimen. A
10.40 Among patients with atherosclerotic cardiovascular disease at high risk of heart failure or in whom heart failure coexists, sodium–glucose cotransporter 2 inhibitors are preferred. C
5.2.0.0 Cardiac Testing
Candidates for advanced or invasive cardiac testing include those with 1) typical or atypical cardiac symptoms and 2) an abnormal resting electrocardiogram (ECG). Exercise ECG testing without or with echocardiography may be used as the initial test. In adults with diabetes ≥40 years of age, measurement of coronary artery calcium is also reasonable for cardiovascular risk assessment. Pharmacologic stress echocardiography or nuclear imaging should be considered in individuals with diabetes in whom resting ECG abnormalities preclude exercise stress testing (e.g., left bundle branch block or ST-T abnormalities). In addition, individuals who require stress testing and are unable to exercise should undergo pharmacologic stress echocardiography or nuclear imaging.
5.3.0.0 Screening Asymptomatic Patients
The screening of asymptomatic patients with high ASCVD risk is not recommended (140), in part because these high-risk patients should already be receiving intensive medical therapydan approach that provides similar benefit as invasive revascularization (141,142). There is also some evidence that silent ischemia may reverse over time, adding to the controversy concerning aggressive screening strategies (143). In prospective studies, coronary artery calcium has been established as an independent predictor of future ASCVD events in patients with diabetes and is consistently superior to both the UK Prospective Diabetes Study (UKPDS) risk engine and the Framingham Risk Score in predicting risk in this population (144-146). However, a randomized observational trial demonstrated no clinical benefit to routine screening of asymptomatic patients with type 2 diabetes and normal ECGs (147). Despite abnormal myocardial perfusion imaging in more than one in five patients, cardiac outcomes were essentially equal (and very low) in screened versus unscreened patients. Accordingly, indiscriminate screening is not considered cost-effective. Studies have found that a risk factor–based approach to the initial diagnostic evaluation and subsequent follow-upfor coronary artery disease fails to identify which patients with type 2 diabetes will have silent ischemia on screening tests (148,149).
Any benefit of newer noninvasive coronary artery disease screening methods, such as computed tomography calcium scoring and computed tomography angiography, to identify patient subgroups for different treatment strategies remains unproven in asymptomatic patients with diabetes, though research is ongoing. Although asymptomatic patients with diabetes with higher coronary disease burden have more future cardiac events (144,150,151), the role of these tests beyond risk stratification is not clear.
While coronary artery screening methods, such as calcium scoring, may improve cardiovascular risk assessment in people with type 2 diabetes (152), their routine use leads to radiation exposure and may result in unnecessary invasive testing such as coronary angiography and revascularization procedures. The ultimate balance of benefit, cost, and risks of such an approach in asymptomatic patients remains controversial, particularly in the modern setting of aggressive ASCVD risk factor control.
5.4.0.0 Lifestyle and Pharmacologic Interventions
Intensive lifestyle intervention focusing on weight loss through decreased caloric intake and increased physical activity as performed in the Action for Health in Diabetes (Look AHEAD) trial may be considered for improving glucose control, fitness, and some ASCVD risk factors (153). Patients at increased ASCVD risk should receive aspirin and a statin and ACE inhibitor or ARB therapy if the patient has hypertension, unless there are contraindications to a particular drug class. While clear benefit exists for ACE inhibitor or ARB therapy in patients with diabetic kidney disease or hypertension, the benefits in patients with ASCVD in the absence of these conditions are less clear, especially when LDL cholesterol is concomitantly controlled (154,155). In patients with prior MI, active angina, or HFrEF, b-blockers should be used (156).
5.5.0.0 Antihyperglycemic Therapies and Cardiovascular Outcomes
In 2008, the FDA issued a guidance for industry to perform cardiovascular outcomes trials for all new medications for the treatment for type 2 diabetes amid concerns of increased cardiovascular risk (157). Previously approved diabetes medications were not subject to the guidance. Recently published cardiovascular outcomes trials have provided additional data on cardiovascular outcomes in patients with type 2 diabetes with cardiovascular disease or at high risk for cardiovascular disease (see Table 10.4). Cardiovascular outcomes trials of dipeptidyl peptidase 4 (DPP-4) inhibitors have all, so far, not shown cardiovascular benefits relative to placebo. However, results from other new agents have provided a mix of results.
The BI 10773 (Empagliflozin) Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients (EMPA-REG OUTCOME) trial was a randomized, double-blind trial that assessed the effect of empagliflozin, an SGLT2 inhibitor, versus placebo on cardiovascular outcomes in 7,020 patients with type 2 diabetes and existing cardiovascular disease. Study participants had a mean age of 63 years, 57% had diabetes for more than 10 years, and 99% had established cardiovascular disease. EMPA-REG OUTCOME showed that over a median follow-up of 3.1 years, treatment reduced the composite outcome of MI, stroke, and cardiovascular death by 14% (absolute rate 10.5% vs. 12.1% in the placebo group, HR in the empagliflozin group 0.86; 95% CI 0.74–0.99; P = 0.04 for superiority) and cardiovascular death by 38% (absolute rate 3.7% vs. 5.9%, HR 0.62; 95% CI 0.49–0.77; P <0.001) (8). The FDA added an indication for empagliflozin to reduce the risk of major adverse cardiovascular death in adults with type 2 diabetes and cardiovascular disease.
A second large cardiovascular outcomes trial program of an SGLT2 inhibitor, canagliflozin, has been reported (9). The Canagliflozin Cardiovascular Assessment Study (CANVAS) integrated data from two trials, including the CANVAS trial that started in 2009 before the approval of canagliflozin and the CANVAS-Renal (CANVAS-R) trial that started in 2014 after the approval of canagliflozin. Combining both these trials, 10,142 participants with type 2 diabetes (two-thirds with established CVD) were randomized to canagliflozin or placebo and were followed for an average 3.6 years. The mean age of patients was 63 years and 66% had a history of cardiovascular disease. The combined analysis of the two trials found that canagliflozin significantly reduced the composite outcome of cardiovascular death, MI, or stroke versus placebo (occurring in 26.9 vs. 31.5 participants per 1,000 patient-years; HR 0.86 [95% CI 0.75–0.97]; P <0.001 for noninferiority; P = 0.02 for superiority). The specific estimates for canagliflozin versus placebo on the primary composite cardiovascular outcome were HR 0.88 (0.75–1.03) for the CANVAS trial and 0.82 (0.66–1.01) for CANVAS-R, with no heterogeneity found between trials. In the combined analysis, there was not a statistically significant difference in cardiovascular death (HR 0.87 [95% CI 0.72–1.06]). The initial CANVAS trial was partially unblinded prior to completion because of the need to file interim cardiovascular outcomes data for regulatory approval of the drug (158). Of note, there was an increased risk of lower-limb amputation with canaglifozin (6.3 vs. 3.4 participants per 1,000 patient-years; HR 1.97 [95% CI 1.41–2.75]) (9).
The Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results (LEADER) trial was a randomized, double-blind trial that assessedtheeffectof liraglutide, a glucagonlike peptide 1 (GLP-1) receptor agonist, versus placebo on cardiovascular outcomes in 9,340 patients with type 2 diabetes at high risk for cardiovascular disease or with cardiovascular disease. Study participants had a mean age of 64 years and a mean duration of diabetes of nearly 13 years. Over 80% of study participants had established cardiovascular disease. After a median follow-up of 3.8 years, LEADER showed that the primary composite outcome (MI, stroke, or cardiovascular death) occurred in fewer participants in the treatment group (13.0%) when compared with the placebo group (14.9%) (HR 0.87; 95% CI 0.78–0.97; P <0.001 for non-inferiority; P = 0.01 for superiority). Deaths from cardiovascular causes were significantly reduced in the liraglutide group (4.7%) compared with the placebo group (6.0%) (HR 0.78; 95% CI 0.66–0.93; P = 0.007) (159). The FDA approved the use of liraglutide to reduce the risk of major adverse cardiovascular events, including heart attack, stroke, and cardiovascular death, in adults with type 2 diabetes and established cardiovascular disease.
Results from a moderate-sized trial of another GLP-1 receptor agonist, semaglutide, were consistent with the LEADER trial (160). Semaglutide is a once-weekly GLP-1 receptor agonist approved by the FDA for the treatment of type 2 diabetes. The Trial to Evaluate Cardiovascular and Other Long-term Outcomes With Semaglutide in Subjects With Type 2 Diabetes (SUSTAIN-6) was the initial randomized trial powered to test noninferiority of semaglutide for the purpose of initial regulatory approval. In this study, 3,297 patients with type 2 diabetes were randomized to receive once-weekly semaglutide (0.5 mg or 1.0 mg) or placebo for 2 years. The primary outcome (the first occurrence of cardiovascular death, non-fatal MI, or nonfatal stroke) occurred in 108 patients (6.6%) in the semaglutide group vs. 146 patients (8.9%) in the placebo group (HR 0.74 [95% CI 0.58–0.95]; P <0.001). More patients discontinued treatment in the semaglutide group because of adverse events, mainly gastrointestinal.
The Evaluation of Lixisenatide in Acute Coronary Syndrome (ELIXA) trial studied the once-daily GLP-1 receptor agonist lixisenatide on cardiovascular outcomes in patients with type 2 diabetes who had had a recent acute coronary event (161). A total of 6,068 patients with type 2 diabetes with a recent hospitalization for MI or unstable angina within the previous 180 days were randomized to receive lixisenatide or placebo in addition to standard care and were followed for a median of approximately 2.1 years. The primary outcome of cardiovascular death, MI, stroke, or hospitalization for unstable angina occurred in 406 patients (13.4%) in the lixisenatide group vs. 399 (13.2%) in the placebo group (HR 1.2 [95% CI 0.89–1.17]), which demonstrated the noninferiority of lixisenatide to placebo (P <0.001) but did not show superiority (P = 0.81).
The Exenatide Study of Cardiovascular Event Lowering (EXSCEL) trial also reported results with the once-weekly GLP-1 receptor agonist extended-release exenatide and found that major adverse cardiovascular events were numerically lower with use of extended-release exenatide compared with placebo, although this difference was not statistically significant(162). A total of 14,752 patients with type 2 diabetes (of whom 10,782 [73.1%] had previous cardiovascular disease) were randomized to receive extended-release exenatide 2 mg or placebo and followed for a median of 3.2 years. The primary end point of cardiovascular death, MI, or stroke occurred in 839 patients (11.4%; 3.7 events per 100 person-years) in the exenatide group and in 905 patients (12.2%; 4.0 events per 100 person-years) in the placebo group (HR 0.91 [95% CI 0.83– 1.00]; P <0.001 for noninferiority) but was not superior to placebo with respect to the primary end point (P = 0.06 for superiority). However, all-cause mortality was lower in the exenatide group (HR 0.86 [95% CI 0.77–0.97]. The incidence of acute pancreatitis, pancreatic cancer, medullary thyroid carcinoma, and serious adverse events did not differ significantly between the two groups.
The Harmony Outcomes trial randomized 9,463 patients with type 2 diabetes and cardiovascular disease to once-weekly subcutaneous albiglutide or matching placebo, in addition to their standard care. Over a median duration of 1.6 years, the GLP-1 receptor agonist reduced the risk of cardiovascular death, MI, or stroke to an incidence rate of 4.6 events per 100 person-years in the albiglutide group vs. 5.9 events in the placebo group (HR ratio 0.78, P = 0.0006 for superiority) (163). This agent is not currently available for clinical use.
In summary, there are now several large randomized controlled trials reporting statistically significant reductions in cardiovascular events for two of the FDA- approved SGLT2 inhibitors (empagliflozin and canagliflozin) and three FDA-approved GLP-1 receptor agonists (liraglutide, albiglutide [although that agent was removed from the market for business reasons], and semaglutide [lower risk of cardiovascular events in a moderate-sized clinical trial but one not powered as a cardiovascular outcomes trial]). In these trials, the majority, if not all, patients in the trial had ASCVD. The empagliflozin and liraglutide trials further demonstrated significant reductions in cardiovascular death. Once-weekly exenatide did not have statistically significant reductions in major adverse cardiovascular events or cardiovascular mortality but did have a significant reduction in all-cause mortality. In contrast, other GLP-1 receptor agonists have not shown similar reductions in cardiovascular events (Table 10.4). Additional large randomized trials of other agents in these classes are ongoing.
Of note, these studies examined the drugs in combination with metformin (Table 10.4) in the great majority of patients for whom metformin was not contraindicated or was tolerated. For patients with type 2 diabetes who have ASCVD, on lifestyle and metformin therapy, it is recommended to incorporate an agent with strong evidence for cardiovascular risk reduction, especially those with proven reduction of cardiovascular death, after consideration of drug-specific patient factors (Table 9.1). See Fig. 9.1 for additional recommendations on antihyperglycemic treatment in adults with type 2 diabetes.
Table 10.4—Cardiovascular outcomes trials of available antihyperglycemic medications completed after the issuance of the FDA 2008 guidelines
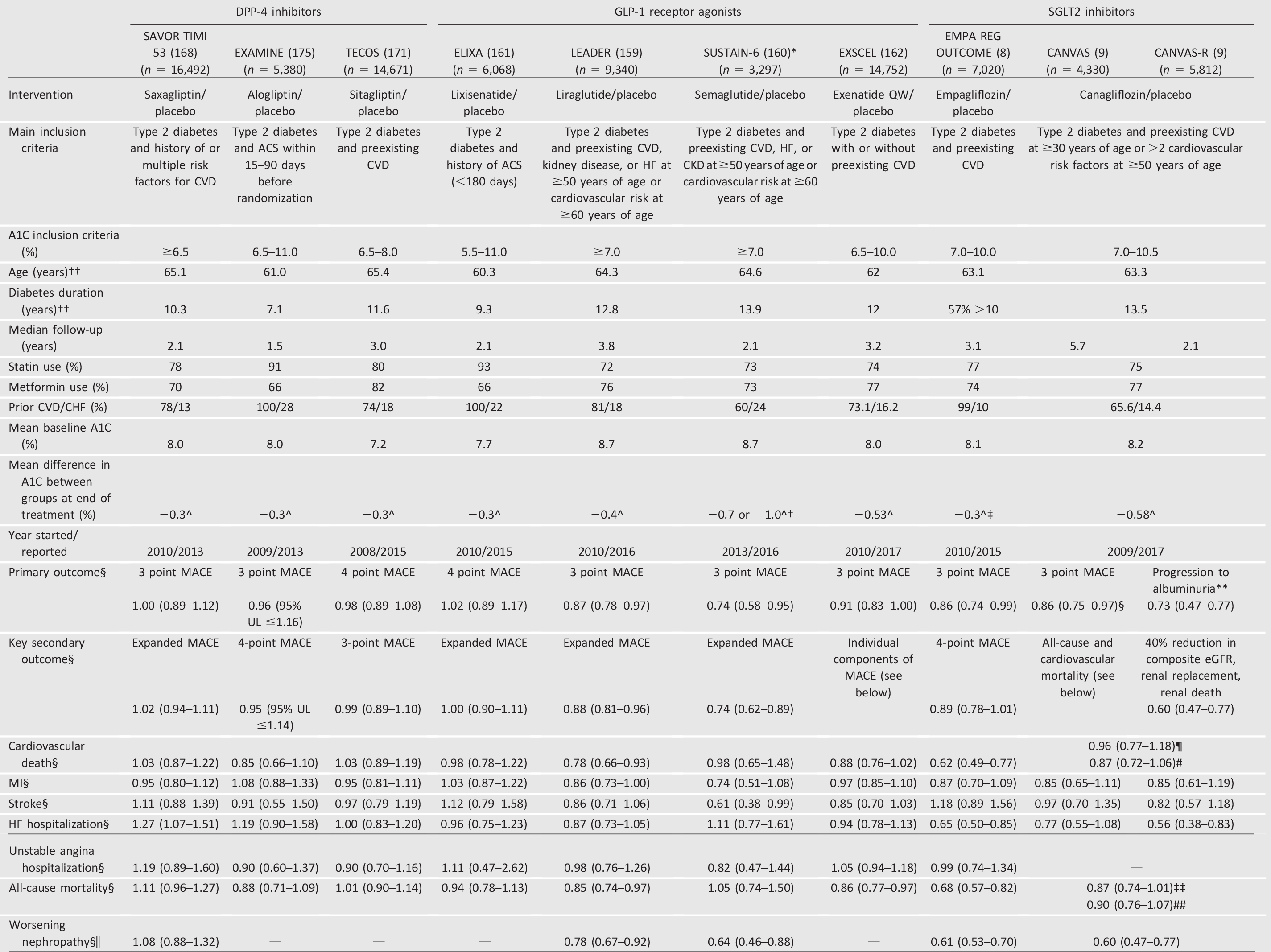
—, not assessed/reported; ACS, acute coronary syndrome; CHF, congestive heart failure; CKD, chronic kidney disease; CVD, cardiovascular disease; DPP-4, dipeptidyl peptidase 4; eGFR, estimated glomerular filtration rate; GLP-1, glucagon-like peptide 1; HF, heart failure; MACE, major adverse cardiac event; MI, myocardial infarction; SGLT2, sodium–glucose cotransporter 2; UL, upper limit. Data from this table was adapted from Cefalu et al. (176) in the January 2018 issue of Diabetes Care.
*Powered to rule out a hazard ratio of 1.8; superiority hypothesis not prespecified.
**On the basis of prespecified outcomes, the renal outcomes are not viewed as statistically significant.
††Age was reported as means in all trials except EXAMINE, which reported medians; diabetes duration was reported as means in all but four trials, with SAVOR- TIMI 58, EXAMINE, and EXSCEL reporting medians and EMPA-REG OUTCOME reporting as percentage of population with diabetes duration >10 years. †A1C change of 0.66% with 0.5 mg and 1.05% with 1 mg dose of semaglutide.
‡AlC change of 0.30 in EMPA-REG OUTCOME is based on pooled results for both doses (i.e., 0.24% for 10 mg and 0.36% for 25 mg of empagliflozin). §Outcomes reported as hazard ratio (95% CI).
||Worsening nephropathy is defined as the new onset of urine albumin-to-creatinine ratio >300 mg/g creatinine or a doubling of the serum creatinine level and an estimated glomerular filtration rate of <45 mL/min/1.73 m2, the need for continuous renal-replacement therapy, or death from renal disease in EMPA-REG OUTCOME, LEADER, and SUSTAIN-6 and as doubling of creatinine level, initiation of dialysis, renal transplantation, or creatinine >6.0 mg/dL (530 mmol/L) in SAVOR-TIMI 53. Worsening nephropathy was a prespecified exploratory adjudicated outcome in SAVOR-TIMI 53, LEADER, and SUSTAIN-6 but not in EMPA-REG OUTCOME.
¶Truncated data set (prespecified in treating hierarchy as the principal data set for analysis for superiority of all-cause mortality and cardiovascular death in the CANVAS Program).
^Significant difference in A1C between groups (P <0.05).
#Nontruncated data set.
‡‡Truncated integrated data set (refers to pooled data from CANVAS after 20 November 2012 plus CANVAS-R; prespecified in treating hierarchy as the principal data set for analysis for superiority of all-cause mortality and cardiovascular death in the CANVAS Program).
##Nontruncated integrated data (refers to pooled data from CANVAS, including before 20 November 2012 plus CANVAS-R).
5.6.0.0 Antihyperglycemic Therapies and Heart Failure
As many as 50% of patients with type 2 diabetes may develop heart failure (164). Data on the effects of glucose-lowering agents on heart failure outcomes have demonstrated that thiazolidinediones have a strong and consistent relationship with increased risk of heart failure (165-167). Therefore, thiazolidinedione use should be avoided in patients with symptomatic heart failure.
Recent studies have also examined the relationship between DPP-4 inhibitors and heart failure and have had mixed results. The Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus2Thrombolysis in Myocardial Infarction 53 (SAVOR-TIMI 53) study showed that patients treated with saxagliptin (a DPP-4 inhibitor) were more likely to be hospitalized for heart failure than those given placebo (3.5% vs. 2.8%, respectively) (168). Two other recent multicenter, randomized, double-blind, noninferiority trials, Examination of Cardiovascular Outcomes with Alogliptin versus Standard of Care (EXAMINE) and Trial Evaluating Cardiovascular Out-comes with Sitagliptin (TECOS), did not show associations between DPP-4 inhibitor use and heart failure. The FDA reported that the hospital admission rate for heart failure in EXAMINE was 3.9% for patients randomly assigned to alogliptin compared with 3.3% for those randomly assigned to placebo (169). Alogliptin had no effect on the composite end point of cardiovascular death and hospital admission for heart failure in the post hoc analysis (HR 1.0 [95% CI 0.82–1.21]) (170). TECOS showed no difference in the rate of heart failure hospitalization for the sitagliptin group (3.1%; 1.07 per 100 person-years) compared with the placebo group (3.1%; 1.09 per 100 person-years) (171).
In four cardiovascular outcome trials of GLP-1 receptor agonists, no evidence for an increased risk of heart failure was found and the agents had a neutral effect on hospitalization for heart failure (159– 162).
A benefit on the incidence of heart failure has been observed with the use of some SGLT2 inhibitors. In EMPA- REG OUTCOME, the addition of empagliflozin to standard care led to a significant 35% reduction in hospitalization for heart failure compared with placebo (8). Although the majority of patients in the study did not have heart failure at baseline, this benefit was consistent in patients with and without a prior history of heart failure (172). Similarly, in CANVAS, there was a 33% reduction in hospitalization for heart failure with canagliflozin versus placebo (9). Although heart failure hospitalizations were prospectively adjudicated in both trials, the type(s) of heart failure events prevented were not characterized. These preliminary findings, which strongly suggest heart failure–related benefits of SGLT2 inhibitors (particularly the prevention of heart failure), are being followed up with new outcomes trials in patients with established heart failure, both with and without diabetes, to determine their efficacy in treatment of heart failure.
6.0.0.0 References
costs of diabetes in the U.S. in 2017. Diabetes Care 2018;41:917–928
ejection fraction: from mechanisms to therapies. Eur Heart J 2018;39:2780–2792
home” hypertension as opposed to “isolated office” hypertension a sign of greater cardiovascular risk? Arch Intern Med 2001;161:2205–2211
analysis of randomized controlled studies. J Hypertens 2013;31:455–467; discussion 467–468
pertensive treatment at different blood pressure levels in patients with diabetes mellitus: systematic review and meta-analyses. BMJ 2016;352: i717
diabetes and hypertension: comparing the ADA and the ACC/AHA recommendations. JAMA 2018; 319:1319–1320
A randomized controlled study of finerenone vs. eplerenone in patients with worsening chronic heart failure and diabetes mellitus and/or chronic kidney disease. Eur Heart J 2016; 37:2105–2114
Pyorala K, Pedersen TR, Kjekshus J,Faergeman O, Olsson AG, Thorgeirsson G. Cholesterol lowering with simvastatin improves prognosis of diabetic patients with coronary heart disease. A subgroup analysis of the Scandinavian Simvastatin Survival Study (4S). Diabetes Care 1997;20:614–620
therapy with or without PCI for stable coronary disease. N Engl J Med 2007;356:1503–1516
Disease (TRANSCEND) Investigators, Yusuf S, Teo K, et al. Effects of the angiotensin-receptor blocker telmisartan on cardiovascular events in high-risk patients intolerant to angiotensin-converting enzyme inhibitors: a randomised controlled trial. Lancet 2008;372:1174–1183