1.0.0.0 Overview
The American Diabetes Association (ADA) “Standards of Medical Care in Diabetes” includes ADA’s current clinical practice recommendations and is intended to provide the components of diabetes care, general treatment goals and guidelines, and tools to evaluate quality of care. Members of the ADA Professional Practice Committee, a multidisciplinary expert committee, are responsible for updating the Standards of Care annually, or more frequently as warranted. For a detailed description of ADA standards, statements, and reports, as well as the evidence-grading system for ADA’s clinical practice recommendations, please refer to the Standards of Care Introduction. Readers who wish to comment on the Standards of Care are invited to do so at professional.diabetes.org/SOC.
Suggested citation: American Diabetes Association.
9. Pharmacologic approaches to glycemic
treatment: Standards of Medical Care in
Diabetesd2019. Diabetes Care 2019;42(Suppl.
1):S90–S102
© 2018 by the American Diabetes Association.
Readers may use this article as long as the work is
properly cited, the use is educational and not for
profit, and the work is not altered. More information
is available at http://www.diabetesjournals
.org/content/license.
2.0.0.0 PHARMACOLOGIC THERAPY FOR TYPE 1 DIABETES
2.1.0.0 Recommendations
Recommendations
9.1 Most people with type 1 diabetes should be treated with multiple daily injections of prandial and basal insulin, or continuous subcutaneous insulin infusion. A
9.2 Most individuals with type 1 diabetes should use rapid-acting insulin analogs to reduce hypoglycemia risk. A
9.3 Consider educating individuals with type 1 diabetes on matching prandial insulin doses to carbohydrate intake, premeal blood glucose levels, and anticipated physical activity. E
9.4 Individuals with type 1 diabetes who have been successfully using continuous subcutaneous insulin infusion should have continued access to this therapy after they turn 65 years of age. E
2.2.0.0 Insulin Therapy
Because the hallmark of type 1 diabetes is absent or near-absent β-cell function, insulin treatment is essential for individuals with type 1 diabetes. Insufficient provision of insulin causes not only hyperglycemia but also systematic metabolic disturbances like hypertriglyceridemia and ketoacidosis, as well as tissue catabolism. Over the past three decades, evidence has accumulated supporting multiple daily injections of insulin or continuous subcutaneous administration through an insulin pump as providing the best combination of effectiveness and safety for people with type 1 diabetes.
Generally, insulin requirements can be estimated based on weight, with typical doses ranging from 0.4 to 1.0 units/kg/day. Higher amounts are required during puberty, pregnancy, and medical illness. The American Diabetes Association/JDRF Type 1 Diabetes Sourcebook notes 0.5 units/kg/day as a typical starting dose in patients with type 1 diabetes who are metabolically stable, with half administered as prandial insulin given to control blood glucose after meals and the other half as basal insulin to control glycemia in the periods between meal absorption (1); this guideline provides detailed information on intensification of therapy to meet individualized needs. In addition, the American Diabetes Association position statement “Type 1 Diabetes Management Through the Life Span” provides a thorough overview of type 1 diabetes treatment (2).
Physiologic insulin secretion varies with glycemia, meal size, and tissue demands for glucose. To approach this variability in people using insulin treatment, strategies have evolved to adjust prandial doses based on predicted needs. Thus, education of patients on how to adjust prandial insulin to account for carbohydrate intake, premeal glucose levels, and anticipated activity can be effective and should be considered. Newly available information suggests that individuals in whom carbohydrate counting is effective can incorporate estimates of meal fat and protein content into their prandial dosing for added benefit (3-5).
Most studies comparing multiple daily injections with continuous subcutaneous insulin infusion (CSII) have been relatively small and of short duration. However, a recent systematic review and meta-analysis concluded that pump therapy has modest advantages for lowering A1C (–0.30% [95% CI –0.58 to –0.02]) and for reducing severe hypoglycemia rates in children and adults (6). There is no consensus to guide choosing which form of insulin administration is best for a given patient, and research to guide this decision making is needed (7). The arrival of continuous glucose monitors to clinical practice has proven beneficial in specific circumstances. Reduction of nocturnal hypoglycemia in people with type 1 diabetes using insulin pumps with glucose sensors is improved by automatic suspension of insulin delivery at a preset glucose level (7-9). The U.S. Food and Drug Administration (FDA) has also approved the first hybrid closed-loop pump system. The safety and efficacy of hybrid closed-loop systems has been supported in the literature in adolescents and adults with type 1 diabetes (10,11). Intensive diabetes management using CSII and continuous glucose monitoring should be considered in selected patients. See Section 7 “Diabetes Technology”for a full discussion of insulin delivery devices.
The Diabetes Control and Complications Trial (DCCT) demonstrated that intensive therapy with multiple daily injections or CSII reduced A1C and was associated with improved long-term outcomes (12-14). The study was carried out with short-acting and intermediate-acting human insulins. Despite better microvascular, macrovascular, and all-cause mortality outcomes, intensive therapy was associated with a higher rate of severe hypoglycemia (61 episodes per 100 patient-years of therapy). Since the DCCT, rapid-acting and long-acting insulin analogs have been developed. These analogs are associated with less hypoglycemia, less weight gain, and lower A1C than human insulins in people with type 1 diabetes (15-17). Longer-acting basal analogs (U-300 glargine or degludec) may convey a lower hypoglycemia risk compared with U-100 glargine in patients with type 1 diabetes (18,19). Rapid-acting inhaled insulin to be used before meals is now available and may reduce rates of hypoglycemia in patients with type 1 diabetes (20).
Postprandial glucose excursions may be better controlled by adjusting the timing of prandial insulin dose administration. The optimal time to administer prandial insulin varies, based on the type of insulin used (regular, rapid-acting analog, inhaled, etc.), measured blood glucose level, timing of meals, and carbohydrate consumption. Recommendations for prandial insulin dose administration should therefore be individualized.
Insulin Injection Technique
Ensuring that patients and/or caregivers understand correct insulin injection technique is important to optimize glucose control and insulin use safety. Thus, it is important that insulin be delivered into the proper tissue in the right way. Recommendations have been published elsewhere outlining best practices for insulin injection (21). Proper insulin injection technique includes injecting into appropriate body areas, injection site rotation, appropriate care of injection sites to avoid infection or other complications, and avoidance of intramuscular (IM) insulin delivery.
Exogenous-delivered insulin should be injected into subcutaneous tissue, not intramuscularly. Recommended sites for insulin injection include the abdomen, thigh, buttock, and upper arm (21). Because insulin absorption from IM sites differs according to the activity of the muscle, inadvertent IM injection can lead to unpredictable insulin absorption and variable effects on glucose, with IM injection being associated with frequent and unexplained hypoglycemia in several reports (21-23). Risk for IM insulin delivery is increased in younger and lean patients when injecting into the limbs rather than truncal sites (abdomen and buttocks) and when using longer needles (24). Recent evidence supports the use of short needles (e.g., 4-mm pen needles) as effective and well tolerated when compared to longer needles (25,26), including a study performed in obese adults (27). Injection site rotation is additionally necessary to avoid lipohypertrophy and lipoatrophy (21). Lipohypertrophy can contribute to erratic insulin absorption, increased glycemic variability, and unexplained hypoglycemic episodes (28). Patients and/or caregivers should receive education about proper injection site rotation and to recognize and avoid areas of lipohypertrophy (21). As noted in Table 4.1, examination of insulin injection sites for the presence of lipohypertrophy, as well as assessment of injection device use and injection technique, are key components of a comprehensive diabetes medical evaluation and treatment plan. As referenced above, there are now numerous evidence-based insulin delivery recommendations that have been published. Proper insulin injection technique may lead to more effective use of this therapy and, as such, holds the potential for improved clinical outcomes.
2.3.0 0 Noninsulin Treatments for Type 1 Diabetes
Injectable and oral glucose-lowering drugs
have been studied for their efficacy as adjuncts to insulin treatment of type 1 diabetes. Pramlintide is based on the naturally occurring β-cell peptide amylin and is approved for use in adults with type 1 diabetes. Results from randomized controlled studies show variable reductions of A1C (0–0.3%) and body weight (1–2 kg) with addition of pramlintide to insulin (29,30). Similarly, results have been reported for several agents currently approved only for the treatment of type 2 diabetes. The addition of metformin to adults with type 1 diabetes caused small reductions in body weight and lipid levels but did not improve A1C (31,32). The addition of the glucagon-like peptide 1 (GLP-1) receptor agonists liraglutide and exenatide to insulin therapy caused small (0.2%) reductions in A1C compared with insulin alone in people with type 1 diabetes and also reduced body weight by ˜3 kg (33). Similarly, the addition of a sodium–glucose cotransporter 2 (SGLT2) inhibitor to insulin therapy has been associated with improvements in A1C and body weight when compared with insulin alone (34-36); however, SGLT2 inhibitor use is also associated with more adverse events including ketoacidosis. The dual SGLT1/2 inhibitor sotagliflozin is currently under consideration by the FDA and, if approved, would be the first adjunctive oral therapy in type 1 diabetes.
The risks and benefits of adjunctive agents beyond pramlintide in type 1 diabetes continue to be evaluated through the regulatory process; however, at this time, these adjunctive agents are not approved in the context of type 1 diabetes (37).
3.0.0.0 SURGICAL TREATMENT FOR TYPE 1 DIABETES
Pancreas and Islet Transplantation
Pancreas and islet transplantation normalizes glucose levels but requires life-long immunosuppression to prevent graft rejection and recurrence of autoimmune islet destruction. Given the potential adverse effects of immunosuppressive therapy, pancreas transplantation should be reserved for patients with type 1 diabetes undergoing simultaneous renal transplantation, following renal transplantation, or for those with recurrent ketoacidosis or severe hypoglycemia despite intensive glycemic management (38).
4.0.0.0 PHARMACOLOGIC THERAPY FOR TYPE 2 DIABETES
4.1.0.0 Recommendations
9.5 Metformin is the preferred initial pharmacologic agent for the treatment of type 2 diabetes. A
9.6 Once initiated, metformin should be continued as long as it is tolerated and not contraindicated; other agents, including insulin, should be added to metformin. A
9.7 Long-term use of metformin may be associated with biochemical vitamin B12 deficiency, and periodic measurement of vitamin B12 levels should be considered in metformin-treated patients, especially in those with anemia or peripheral neuropathy. B
9.8 The early introduction of insulin should be considered if there is evidence of ongoing catabolism (weight loss), if symptoms of hyperglycemia are present, or when A1C levels (.10% [86 mmol/mol]) or blood glucose levels (≥300 mg/dL [16.7 mmol/L]) are very high. E
9.9 Consider initiating dual therapy in patients with newly diagnosed type 2 diabetes who have A1C ≥1.5% (12.5 mmol/ mol) above their glycemic target. E
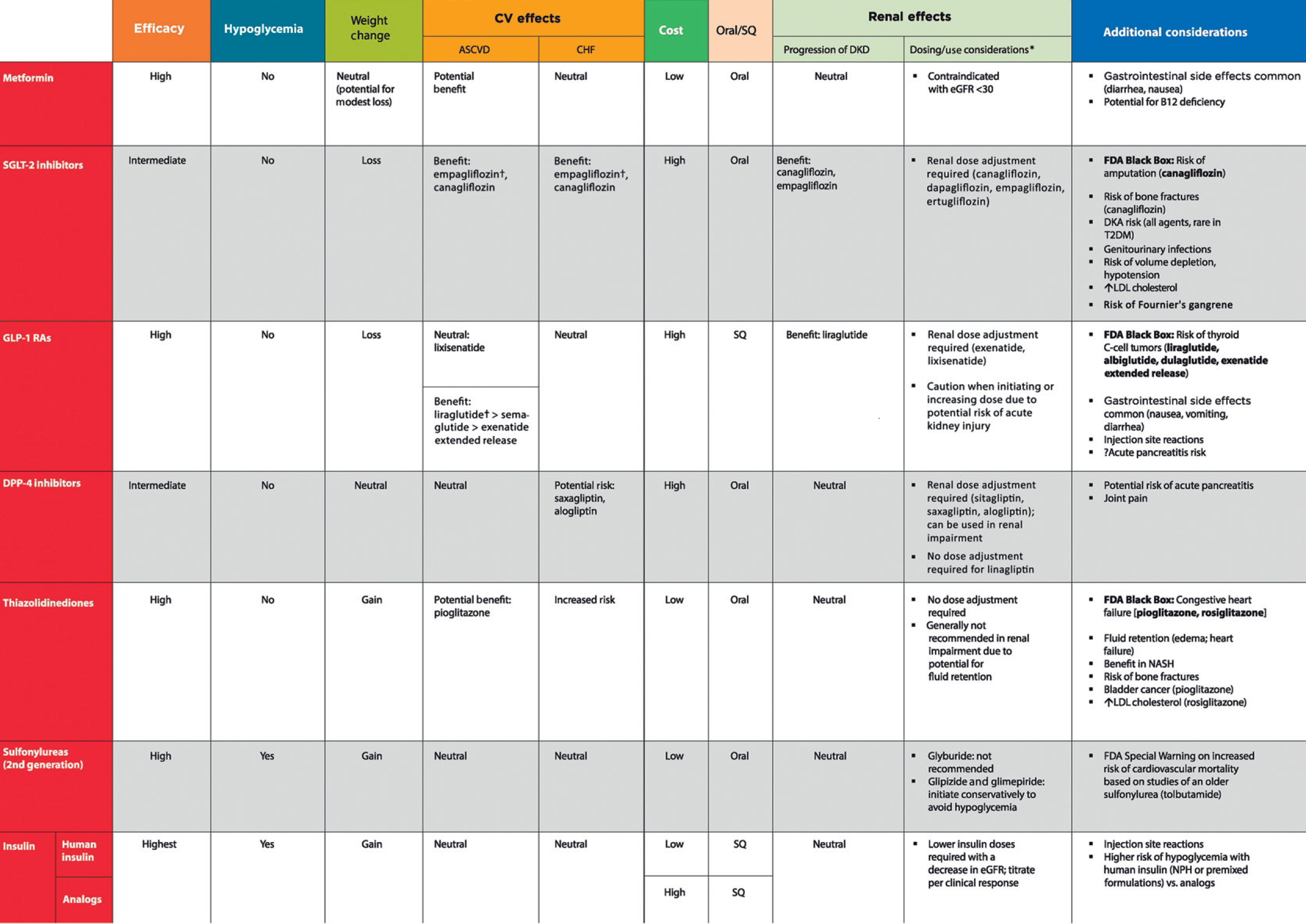
9.10 A patient-centered approach should be used to guide the choice of pharmacologic agents. Considerations include comorbidities (atherosclerotic cardiovascular disease, heart failure, chronic kidney disease), hypoglycemia risk, impact on weight, cost, risk for side effects, and patient preferences. E
9.11 Among patients with type 2 diabetes who have established atherosclerotic cardiovascular disease, sodium–glucose cotransporter 2 inhibitors, or glucagon-like peptide 1 receptor agonists with demonstrated cardiovascular disease benefit (Table 9.1) are recommended as part of the antihyperglycemic regimen. A
9.12 Among patients with atherosclerotic cardiovascular disease at high risk of heart failure or in whom heart failure coexists, sodium–glucose cotransporter 2 inhibitors are preferred. C
9.13 For patients with type 2 diabetes and chronic kidney disease, consider use of a sodium– glucose cotransporter 2 inhibitor or glucagon-like peptide 1 receptor agonist shown to reduce risk of chronic kidney disease progression, cardiovascular events, or both. C
9.14 In most patients who need the greater glucose-lowering effect of an injectable medication, glucagon-like peptide 1 receptor agonists are preferred to insulin. B
9.15 Intensification of treatment for patients with type 2 diabetes not meeting treatment goals should not be delayed. B
9.16 The medication regimen should be reevaluated at regular intervals (every 3–6 months) and adjusted as needed to incorporate new patient factors (Table 9.1). E
The American Diabetes Association/ European Association for the Study of Diabetes consensus report “Management of Hyperglycemia in Type 2 Diabetes, 2018” (39) recommends a patient-centered approach to choosing appropriate pharmacologic treatment of blood glucose (Fig. 9.1). This includes consideration of efficacy and key patient factors: 1) important comorbidities such as atherosclerotic cardiovascular disease (ASCVD), chronic kidney disease (CKD), and heart failure (HF), 2) hypoglycemia risk, 3) effects on body weight, 4) side effects, 5) cost, and 6) patient preferences. Lifestyle modifications that improve health (see Section 5 “Lifestyle Management”) should be emphasized along with any pharmacologic therapy. See Sections 12 and 13 for recommendations specific for older adults and for children and adolescents with type 2 diabetes, respectively.
Table 9.1—Drug-specific and patient factors to consider when selecting antihyperglycemic treatment in adults with type 2 diabetes

Figure 9.1 Glucose-lowering medication in type 2 diabetes:
Figure 9.1—Glucose-lowering medication in type 2 diabetes: overall approach. For appropriate context, see Fig. 4.1. ASCVD, atherosclerotic cardiovascular disease; CKD, chronic kidney disease; CV, cardiovascular; CVD, cardiovascular disease; CVOTs, cardiovascular outcomes trials; DPP-4i, dipeptidyl peptidase 4 inhibitor; eGFR, estimated glomerular filtration rate; GLP-1 RA, glucagon-like peptide 1 receptor agonist; HF, heart failure; SGLT2i, sodium–glucose cotransporter 2 inhibitor; SU, sulfonylurea; TZD, thiazolidinedione. Adapted from Davies et al. (39).
4.2.0.0 Initial Therapy
Metformin should be started at the time type 2 diabetes is diagnosed unless there are contraindications; for most patients this will be monotherapy in combination with lifestyle modifications. Metformin is effective and safe, is inexpensive, and may reduce risk of cardiovascular events and death (40). Metformin is available in an immediate-release form for twicedaily dosing or as an extended-release form that can be given once daily. Compared with sulfonylureas, metformin as first-line therapy has beneficial effects on A1C, weight, and cardiovascular mortality (41); there is little systematic data available for other oral agents as initial therapy of type 2 diabetes. The principal side effects of metformin are gastrointestinal intolerance due to bloating, abdominal discomfort, and diarrhea. The drug is cleared by renal filtration, and very high circulating levels (e.g., as a result of overdose or acute renal failure) have been associated with lactic acidosis. However, the occurrence of this complication is now known to be very rare, and metformin may be safely used in patients with reduced estimated glomerular filtration rates (eGFR); the FDA has revised the label for metformin to reflect its safety in patients with eGFR ≥30 mL/min/1.73 m2 (42). A recent randomized trial confirmed previous observations that metformin use is associated with vitamin B12 deficiency and worsening of symptoms of neuropathy (43). This is compatible with a recent report from the Diabetes Prevention Program Outcomes Study (DPPOS) suggesting periodic testing of vitamin B12 (44).
In patients with contraindications or intolerance of metformin, initial therapy should be based on patient factors; consider a drug from another class depicted in Fig. 9.1. When A1C is ≥1.5% (12.5 mmol/mol) above glycemic target (see Section 6 “Glycemic Targets” for more information on selecting appropriate targets), many patients will require dual combination therapy to achieve their target A1C level (45). Insulin has the advantage of being effective where other agents are not and should be considered as part of any combination regimen when hyperglycemia is severe, especially if catabolic features (weight loss, hypertriglyceridemia, ketosis) are present. Consider initiating insulin therapy when blood glucose is ≥300 mg/dL (16.7 mmol/L) or A1C is ≥10% (86 mmol/mol) or if the patient has symptoms of hyperglycemia (i.e., polyuria or polydipsia), even at diagnosis or early in the course of treatment (Fig. 9.2). As glucose toxicity resolves, simplifying the regimen and/or changing to oral agents is often possible.
Figure 9.2 Intensifying to injectable therapies
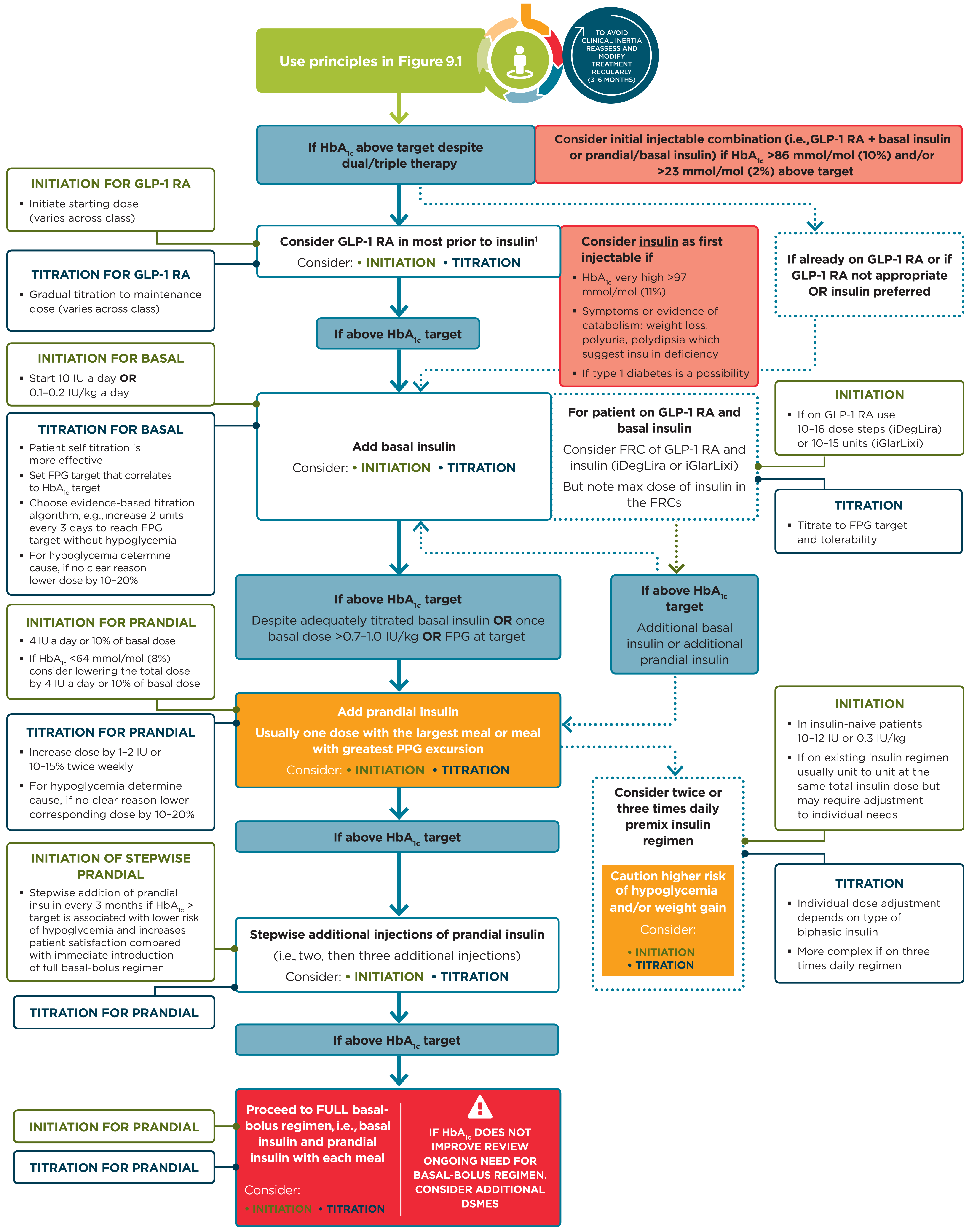
Figure 9.2—Intensifying to injectable therapies. For appropriate context, see Fig. 4.1. DSMES, diabetes self-management education and support; FPG, fasting plasma glucose; FRC, fixed-ratio combination; GLP-1 RA, glucagon-like peptide 1 receptor agonist; max, maximum; PPG, postprandial glucose. Adapted from Davies et al. (39).
4.3.0.0 Combination Therapy
Although there are numerous trials comparing dual therapy with metformin alone, few directly compare drugs as add-on therapy. A comparative effectiveness meta-analysis suggests that each new class of noninsulin agents added to initial therapy generally lowers A1C approximately 0.7–1.0% (46). If the A1C target is not achieved after approximately 3 months and the patient does not have ASCVD or CKD, consider a combination of metformin and any one of the preferred six treatment options: sulfonylurea, thiazolidinedione, dipeptidyl peptidase 4 (DPP-4) inhibitor, SGLT2 inhibitor, GLP-1 receptor agonist, or basal insulin; the choice of which agent to add is based on drug-specific effects and patient factors (Fig. 9.1 and Table 9.1). For patients in whom ASCVD, HF, or CKD predominates, the best choice for a second agent is a GLP-1 receptor agonist or SGLT2 inhibitor with demonstrated cardiovascular risk reduction, after consideration of drug-specific and patient factors (Table 9.1). For patients without established ASCVD or CKD, the choice of a second agent to add to metformin is not yet guided by empiric evidence. Rather, drug choice is based on avoidance of side effects, particularly hypoglycemia and weight gain, cost, and patient preferences (47). Similar considerations are applied in patients who require a third agent to achieve glycemic goals; there is also very little trial-based evidence to guide this choice. In all cases, treatment regimens need to be continuously reviewed for effi- cacy, side effects, and patient burden (Table 9.1). In some instances, patients will require medication reduction or discontinuation. Common reasons for this include ineffectiveness, intolerable side effects, expense, or a change in glycemic goals (e.g., in response to development of comorbidities or changes in treatment goals). See Section 12 "Older Adults” for a full discussion of treatment considerations in older adults.
Even though most patients prefer oral medications to drugs that need to be injected, the eventual need for the greater potency of injectable medications is common, particularly in people with a longer duration of diabetes. The addition of basal insulin, either human NPH or one of the long-acting insulin analogs, to oral agent regimens is a well-established approach that is effective for many patients. In addition, recent evidence supports the utility of GLP-1 receptor agonists in patients not reaching glycemic targets with oral agent regimens. In trials comparing the addition of GLP-1 receptor agonists or insulin in patients needing further glucose lowering, the efficacy of the two treatments was similar (48-50). However, GLP-1 receptor agonists had a lower risk ofhypoglycemiaandbeneficialeffects on body weight compared with insulin, albeit with greater gastrointestinal side effects. Thus, trial results support a GLP-1 receptor agonist as the preferred option for patients requiring the potency of an injectable therapy for glucose control (Fig. 9.2). However, high costs and tolerability issues are important barriers to the use of GLP-1 receptor agonists.
Cost-effectiveness models of the newer agents based on clinical utility and glycemic effect have been reported (51). Table 9.2 provides cost information for currently approved noninsulin therapies. Of note, prices listed are average wholesale prices (AWP) (52) and National Average Drug Acquisition Costs (NADAC) (53) and do not account for discounts, rebates, or other price adjustments often involved in prescription sales that affect the actual cost incurred by the patient. While there are alternative means to estimate medication prices, AWP and NADAC were utilized to provide two separate measures to allow for a comparison of drug prices with the primary goal of highlighting the importance of cost considerations when prescribing antihyperglycemic treatments.
Table 9.2—Median monthly cost of maximum approved daily dose of noninsulin glucose-lowering agents in the U.S.
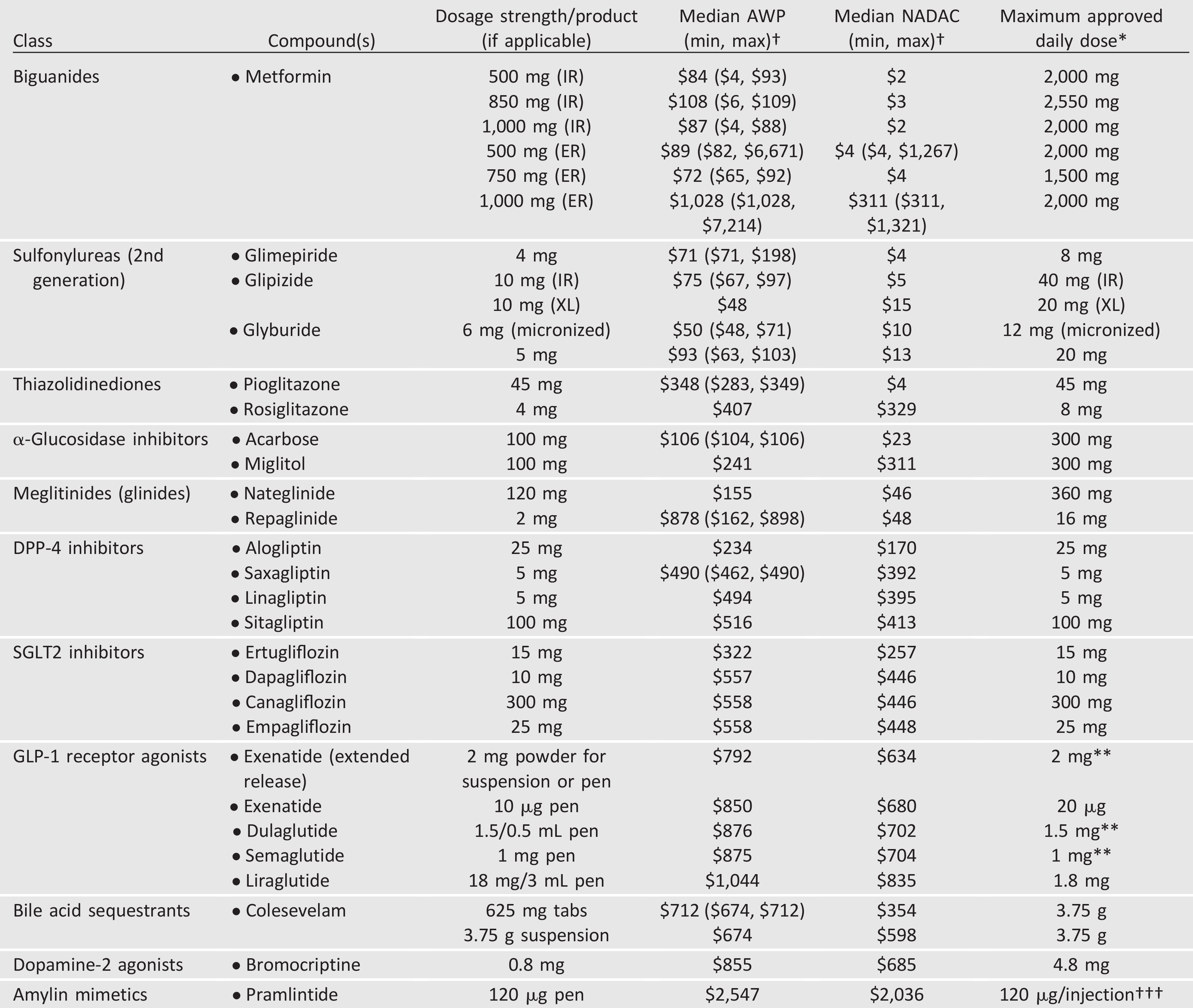
AWP, average wholesale price; DPP-4, dipeptidyl peptidase 4; ER and XL, extended release; GLP-1, glucagon-like peptide 1; IR, immediate release; NADAC, National Average Drug Acquisition Cost; SGLT2, sodium–glucose cotransporter 2. †Calculated for 30-day supply (AWP [44] or NADAC [45] unit price 3 number of doses required to provide maximum approved daily dose 3 30 days); median AWP or NADAC listed alone when only one product and/or price. *Utilized to calculate median AWP and NADAC (min, max); generic prices used, if available commercially. **Administered once weekly. †††AWP and NADAC calculated based on 120 mg three times daily.
Table 9.3—Median cost of insulin products in the U.S. calculated as AWP (44) and NADAC (45) per 1,000 units of specified dosage form/product
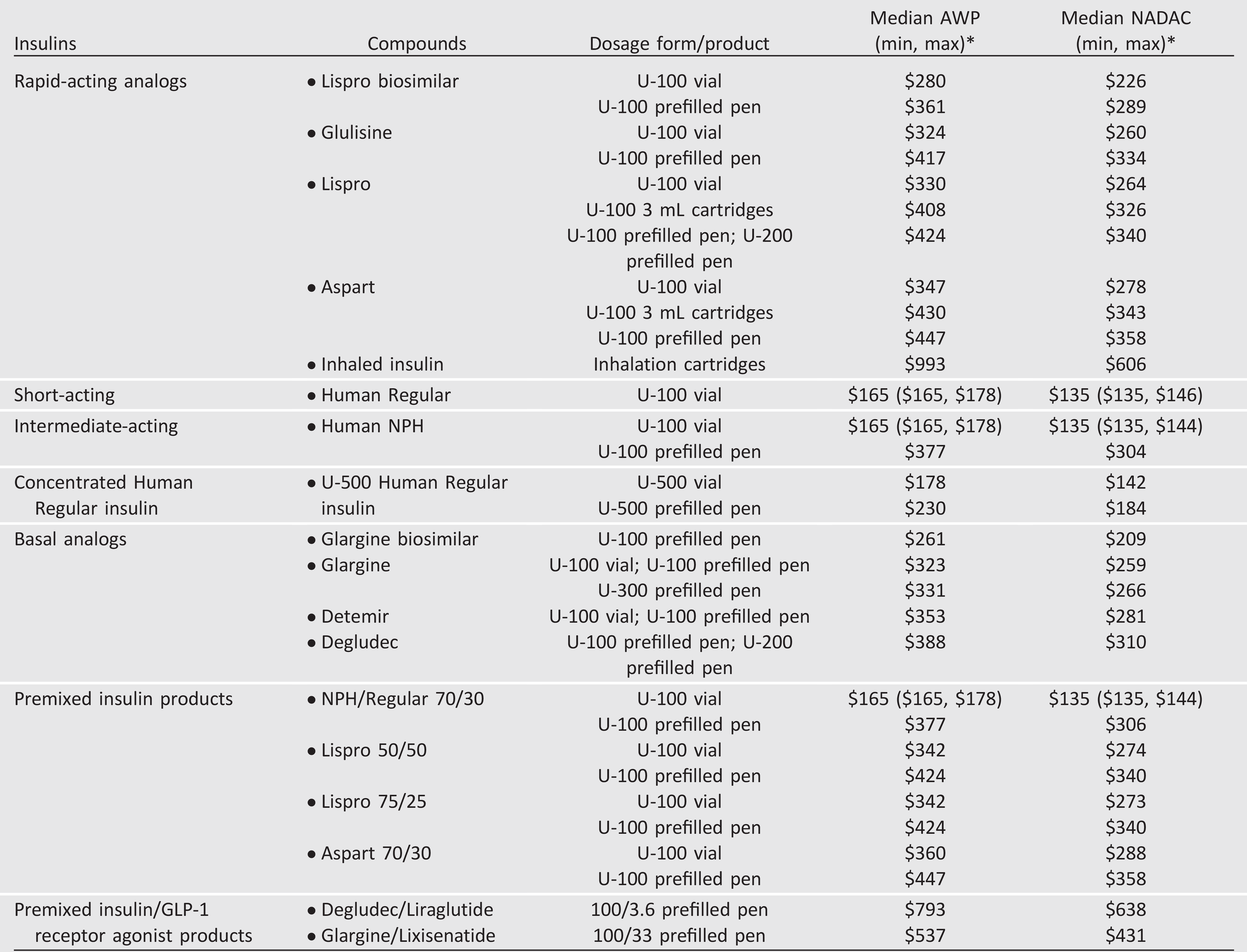
AWP, average wholesale price; GLP-1, glucagon-like peptide 1; NADAC, National Average Drug Acquisition Cost. *AWP or NADAC calculated as in
Table 9.2; median listed alone when only one product and/or price.
4.4.0.0 Cardiovascular Outcomes Trials
There are now multiple large randomized controlled trials reporting statistically significant reductions in cardiovascular events in patients with type 2 diabetes treated with an SGLT2 inhibitor (empagliflozin, canagliflozin) or GLP-1 receptor agonist (liraglutide, semaglutide). In people with diabetes with established ASCVD, empagliflozin decreased a composite three-point major cardiovascular event (MACE) outcome and mortality compared with placebo (54). Similarly, canagliflozin reduced the occurrence of MACE in a group of subjects with, or at high risk for, ASCVD (55). In both of these trials, SGLT2 inhibitors reduced hospitalization for HF (54,55); this was a secondary outcome of these studies and will require confirmation in more defined populations. In people with type 2 diabetes with ASCVD or increased risk for ASCVD, the addition of liraglutide decreased MACE and mortality (56), and the closely related GLP-1 receptor agonist semaglutide also had favorable effects on cardiovascular end points in high-risk subjects (57). In these cardiovascular outcomes trials, empagliflozin, canagliflozin, liraglutide, and semaglutide all had beneficial effects on composite indices of CKD (54-57). See ANTIHYPERGLYCEMIC THERAPIES AND CARDIOVASCULAR OUTCOMES in Section 10 “Cardiovascular Disease and Risk Management” and Table 10.4 for a detailed description of these cardiovascular outcomes trials, as well as a discussion of how HF may impact treatment choices. See Section 11 “Microvascular Complications and Foot Care” for a detailed discussion on how CKD may impact treatment choices. Additional large randomized trials of other agents in these classes are ongoing.
The subjects enrolled in the cardiovascular outcomes trials using empagliflozin, canagliflozin, liraglutide, and semaglutide had A1C ≥7%, and more than 70% were taking metformin at baseline. Moreover, the benefit of treatment was less evident in subjects with lower risk for ASCVD. Thus, extension of these results to practice is most appropriate for people with type 2 diabetes and established ASCVD who require additional glucose-lowering treatment beyond metformin and lifestyle management. For these patients, incorporating one of the SGLT2 inhibitors or GLP-1 receptor agonists that have been demonstrated to reduce cardiovascular events is recommended (Table 9.1).
4.5.0.0 Insulin Therapy
4.5.1.0 Overview
Many patients with type 2 diabetes eventually require and benefit from insulin therapy (Fig. 9.2). See the section above, INSULIN INJECTION TECHNIQUE, for important guidance on how to administer insulin safely and effectively. The progressive nature of type 2 diabetes should be regularly and objectively explained to patients, and providers should avoid using insulin as a threat or describing it as a sign of personal failure or punishment. Rather, the utility and importance of insulin to maintain glycemic control once progression of the disease overcomes the effect of oral agents should be emphasized. Educating and involving patients in insulin management is beneficial. Instruction of patients in self-titration of insulin doses based on self-monitoring of blood glucose improves glycemic control in patients with type 2 diabetes initiating insulin(58). Comprehensive education regarding self-monitoring of blood glucose, diet, and the avoidance and appropriate treatment of hypoglycemia are critically important in any patient using insulin.
4.5.2.0 Basal Insulin
Basal insulin alone is the most convenient initial insulin regimen and can be added to metformin and other oral agents. Starting doses can be estimated based on body weight (e.g., 10 units a day or 0.1–0.2 units/kg/day) and the degree of hyperglycemia, with individualized titration over days to weeks as needed. The principal action of basal insulin is to restrain hepatic glucose production, with a goal of maintaining euglycemia overnight and between meals (59,60). Control of fasting glucose can be achieved with human NPH insulin or with the use of a long-acting insulin analog. In clinical trials, long-acting basal analogs (U-100 glargine or detemir) have been demonstrated to reduce the risk of symptomatic and nocturnal hypoglycemia compared with NPH insulin (61-66), although these advantages are generally modest and may not persist (67). Longer-acting basal analogs (U-300 glargine or degludec) may convey a lower hypoglycemia risk compared with U-100 glargine when used in combination with oral agents (68-74). Despite evidence for reduced hypoglycemia with newer, longer-acting basal insulin analogs in clinical trial settings, in practice they may not affect the development of hypoglycemia compared with NPH insulin (75).
The cost of insulin has been rising steadily, and at a pace several fold that of other medical expenditures, over the past decade (76). This expense contributes significant burden to the patient as insulin has become a growing “out-of-pocket” cost for people with diabetes, and direct patient costs contribute to treatment nonadherence (76). Therefore, consideration of cost is an important component of effective management. For many patients with type 2 diabetes (e.g., individuals with relaxed A1C goals, low rates of hypoglycemia, and prominent insulin resistance, as well as those with cost concerns), human insulin (NPH and Regular) may be the appropriate choice of therapy, and clinicians should be familiar with its use (77). Table 9.3 provides AWP (52) and NADAC (53) information (cost per 1,000 units) for currently available insulin and insulin combination products in the U.S. As stated for Table 9.2, AWP and NADAC prices listed do not account for discounts, rebates, or other price adjustments that may affect the actual cost to the patient. For example, human regular insulin, NPH, and 70/30 NPH/Regular products can be purchased for considerably less than the AWP and NADAC prices listed in Table 9.3 at select pharmacies.
4.5.3.0 Prandial Insulin
Individuals with type 2 diabetes may require doses of insulin before meals in addition to basal insulin. The recommended starting dose of mealtime insulin is either 4 units or 10% of the basal dose at each meal. Titration is done based on home glucose monitoring or A1C. With significant additions to the prandial insulin dose, particularly with the evening meal, consideration should be given to decreasing the basal insulin dose. Meta-analyses of trials comparing rapid-acting insulin analogs with human regular insulin in patients with type 2 diabetes have not reported important differences in A1C or hypoglycemia (78,79).
4.5.4.0 Premixed Insulin
Premixed insulin products contain both a basal and prandial component, allowing coverage of both basal and prandial needs with a single injection. The NPH/Regular premix is composed of 70% NPH insulin and 30% regular insulin. The use of premixed insulin products has its advantages and disadvantages, as discussed below in COMBINATION INJECTABLE THERAPY.
4.5.5.0 Concentrated Insulin Products
Several concentrated insulin preparations are currently available. U-500 regular insulin is, by definition, five times more concentrated than U-100 regular insulin. Regular U-500 has distinct pharmacokinetics with delayed onset and longer duration of action, characteristics more like an intermediate-acting insulin. U-300 glargine and U-200 degludec are three and two times as concentrated, respectively, as their U-100 formulations and allow higher doses of basal insulin administration per volume used. U-300 glargine has a longer duration of action than U-100 glargine but modestly lower efficacy per unit administered (80,81). The FDA has also approved a concentrated formulation of rapid-acting insulin lispro, U-200 (200 units/mL). These concentrated preparations may be more convenient and comfortable for patients to inject and may improve adherence in those with insulin resistance who require large doses of insulin. While U-500 regular insulin is available in both prefilled pens and vials (a dedicated syringe was FDA approved in July 2016), other concentrated insulins are available only in prefilled pens to minimize the risk of dosing errors.
4.5.6.0 Inhaled Insulin
Inhaled insulin is available for prandial use with a limited dosing range; studies in people with type 1 diabetes suggest rapid pharmacokinetics (20). A pilot study found evidence that compared with injectable rapid-acting insulin, supplemental doses of inhaled insulin taken based on post-prandial glucose levels may improve blood glucose management without additional hypoglycemia or weight gain, although results from a larger study are needed for confirmation (82).
Inhaled insulin is contraindicated in patients with chronic lung disease, such as asthma and chronic obstructive pulmonary disease, and is not recommended in patients who smoke or who recently stopped smoking. All patients require spirometry (FEV1) testing to identify potential lung disease prior to and after starting inhaled insulin therapy.
4.6.0.0 Combination Injectable Therapy
If basal insulin has been titrated to an acceptable fasting blood glucose level (or if the dose is >0.5 units/kg/day) and A1C remains above target, consider advancing to combination injectable therapy (Fig. 9.2). Thisapproach can use a GLP- 1 receptor agonist added to basal insulin or multiple doses of insulin. The combination of basal insulin and GLP-1 receptor agonist has potent glucose-lowering actions and less weight gain and hypoglycemia compared with intensified insulin regimens (83-85). Two different once-daily fixed-dual combination products containing basal insulin plus a GLP-1 receptor agonist are available: insulin glargine plus lixisenatide and insulin degludec plus liraglutide.
Intensification of insulin treatment can be done by adding doses of prandial to basal insulin. Starting with a single prandial dose with the largest meal of the day is simple and effective, and it can be advanced to a regimen with multiple prandial doses if necessary (86). Alternatively, in a patient on basal insulin in whom additional prandial coverage is desired, the regimen can be converted to two or three doses of a premixed insulin. Each approach has advantages and disadvantages. For example, basal/prandial regimens offer greater flexibility for patients who eat on irregular schedules. On the other hand, two doses of premixed insulin is a simple, convenient means of spreading insulin across the day. Moreover, human insulins, separately or as premixed NPH/Regular (70/30) formulations, are less costly alternatives to insulin analogs. Figure 9.2 outlines these options, as well as recommendations for further intensification, if needed, to achieve glycemic goals.
When initiating combination injectable therapy, metformin therapy should be maintained while sulfonylureas and DPP-4 inhibitors are typically discontinued. In patients with suboptimal blood glucose control, especially those requiring large insulin doses, adjunctive use of a thiazolidinedione or an SGLT2 inhibitor may help to improve control and reduce the amount of insulin needed, though potential side effects should be considered. Once a basal/bolus insulin regimen is initiated, dose titration is important, with adjustments made in both mealtime and basal insulins based on the blood glucose levels and an understanding of the pharmacodynamic profile of each formulation (pattern control). As people with type 2 diabetes get older, it may become necessary to simplify complex insulin regimens because of a decline in self-management ability (see Section 12 “Older Adults”).
5.0.0.0 References
U.S. Food and Drug Administration. FDA Drug Safety Communication: FDA revises warnings regarding use of the diabetes medicine metformin in certain patients with reduced kidney function [Internet]. Available from http://www.fda.gov/Drugs/DrugSafety/ucm493244.htm. Accessed 14 October 2016
Truven Health Analytics. Micromedex 2.0 Introduction to RED BOOK Online [Internet], 2018. Available from .com/micromedex2/4.34.0/WebHelp/RED_BOOK/ Introduction_to_REDB_BOOK_Online.htm. Accessed 5 September 2018