4.3.0.0 Combination Therapy
Although there are numerous trials comparing dual therapy with metformin alone, few directly compare drugs as add-on therapy. A comparative effectiveness meta-analysis suggests that each new class of noninsulin agents added to initial therapy generally lowers A1C approximately 0.7–1.0% (46). If the A1C target is not achieved after approximately 3 months and the patient does not have ASCVD or CKD, consider a combination of metformin and any one of the preferred six treatment options: sulfonylurea, thiazolidinedione, dipeptidyl peptidase 4 (DPP-4) inhibitor, SGLT2 inhibitor, GLP-1 receptor agonist, or basal insulin; the choice of which agent to add is based on drug-specific effects and patient factors (Fig. 9.1 and Table 9.1). For patients in whom ASCVD, HF, or CKD predominates, the best choice for a second agent is a GLP-1 receptor agonist or SGLT2 inhibitor with demonstrated cardiovascular risk reduction, after consideration of drug-specific and patient factors (Table 9.1). For patients without established ASCVD or CKD, the choice of a second agent to add to metformin is not yet guided by empiric evidence. Rather, drug choice is based on avoidance of side effects, particularly hypoglycemia and weight gain, cost, and patient preferences (47). Similar considerations are applied in patients who require a third agent to achieve glycemic goals; there is also very little trial-based evidence to guide this choice. In all cases, treatment regimens need to be continuously reviewed for efficacy, side effects, and patient burden (Table 9.1). In some instances, patients will require medication reduction or discontinuation. Common reasons for this include ineffectiveness, intolerable side effects, expense, or a change in glycemic goals (e.g., in response to development of comorbidities or changes in treatment goals). See Section 12 "Older Adults” for a full discussion of treatment considerations in older adults.
Even though most patients prefer oral medications to drugs that need to be injected, the eventual need for the greater potency of injectable medications is common, particularly in people with a longer duration of diabetes. The addition of basal insulin, either human NPH or one of the long-acting insulin analogs, to oral agent regimens is a well-established approach that is effective for many patients. In addition, recent evidence supports the utility of GLP-1 receptor agonists in patients not reaching glycemic targets with oral agent regimens. In trials comparing the addition of GLP-1 receptor agonists or insulin in patients needing further glucose lowering, the efficacy of the two treatments was similar (48-50). However, GLP-1 receptor agonists had a lower risk of hypoglycemia and beneficial effects on body weight compared with insulin, albeit with greater gastrointestinal side effects. Thus, trial results support a GLP-1 receptor agonist as the preferred option for patients requiring the potency of an injectable therapy for glucose control (Fig. 9.2). However, high costs and tolerability issues are important barriers to the use of GLP-1 receptor agonists.
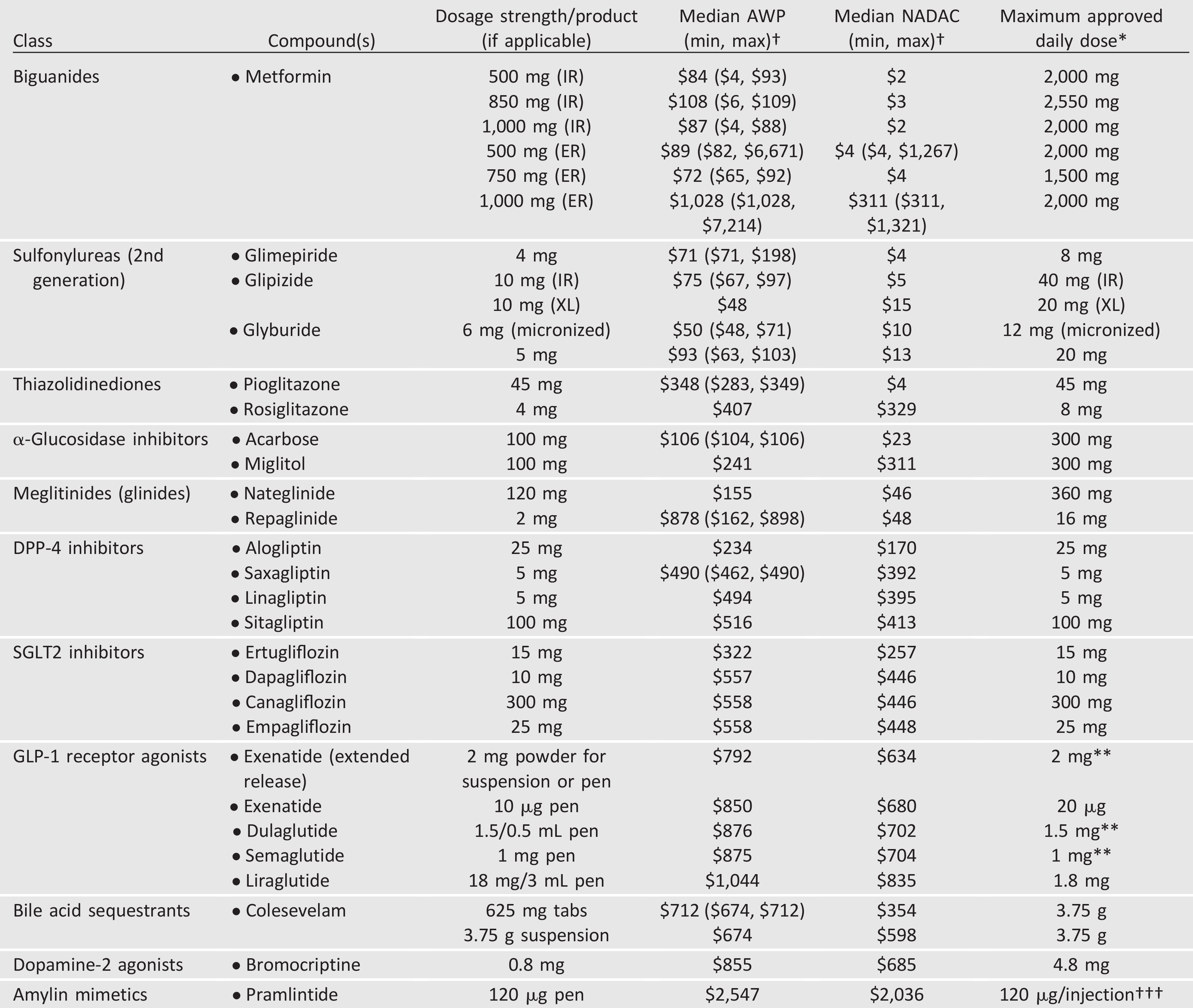
Cost-effectiveness models of the newer agents based on clinical utility and glycemic effect have been reported (51). Table 9.2 provides cost information for currently approved noninsulin therapies. Of note, prices listed are average wholesale prices (AWP) (52) and National Average Drug Acquisition Costs (NADAC) (53) and do not account for discounts, rebates, or other price adjustments often involved in prescription sales that affect the actual cost incurred by the patient. While there are alternative means to estimate medication prices, AWP and NADAC were utilized to provide two separate measures to allow for a comparison of drug prices with the primary goal of highlighting the importance of cost considerations when prescribing antihyperglycemic treatments.
Table 9.2—Median monthly cost of maximum approved daily dose of noninsulin glucose-lowering agents in the U.S.

AWP, average wholesale price; DPP-4, dipeptidyl peptidase 4; ER and XL, extended release; GLP-1, glucagon-like peptide 1; IR, immediate release; NADAC, National Average Drug Acquisition Cost; SGLT2, sodium–glucose cotransporter 2.
†Calculated for 30-day supply (AWP [44] or NADAC [45] unit price x number of doses required to provide maximum approved daily dose x 30 days); median AWP or NADAC listed alone when only one product and/or price.
*Utilized to calculate median AWP and NADAC (min, max); generic prices used, if available commercially.
**Administered once weekly.
†††AWP and NADAC calculated based on 120 µg three times daily.
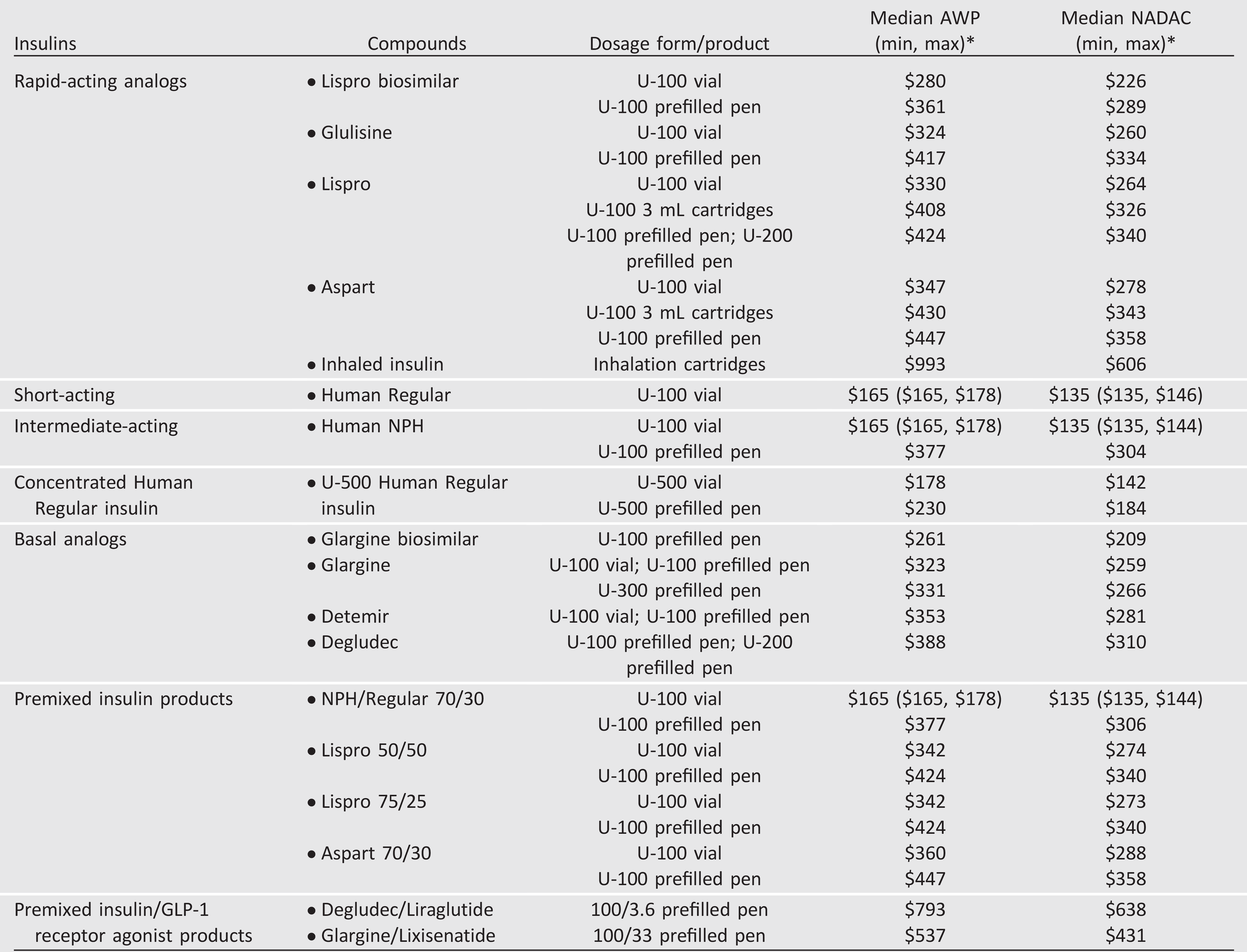
Table 9.3—Median cost of insulin products in the U.S. calculated as AWP (44) and NADAC (45) per 1,000 units of specified dosage form/product