4.0.0.0 PHARMACOLOGIC THERAPY FOR TYPE 2 DIABETES
4.1.0.0 Recommendations
Recommendations
9.5 Metformin is the preferred initial pharmacologic agent for the treatment of type 2 diabetes. A
9.6 Once initiated, metformin should be continued as long as it is tolerated and not contraindicated; other agents, including insulin, should be added to metformin. A
9.7 Long-term use of metformin may be associated with biochemical vitamin B12 deficiency, and periodic measurement of vitamin B12 levels should be considered in metformin-treated patients, especially in those with anemia or peripheral neuropathy. B
9.8 The early introduction of insulin should be considered if there is evidence of ongoing catabolism (weight loss), if symptoms of hyperglycemia are present, or when A1C levels (>10% [86 mmol/mol]) or blood glucose levels (≥300 mg/dL [16.7 mmol/L]) are very high. E
9.9 Consider initiating dual therapy in patients with newly diagnosed type 2 diabetes who have A1C ≥1.5% (12.5 mmol/mol) above their glycemic target. E
9.10 A patient-centered approach should be used to guide the choice of pharmacologic agents. Considerations include comorbidities (atherosclerotic cardiovascular disease, heart failure, chronic kidney disease), hypoglycemia risk, impact on weight, cost, risk for side effects, and patient preferences. E
9.11 Among patients with type 2 diabetes who have established atherosclerotic cardiovascular disease, sodium–glucose cotransporter 2 inhibitors, or glucagon-like peptide 1 receptor agonists with demonstrated cardiovascular disease benefit (Table 9.1) are recommended as part of the antihyperglycemic regimen. A
9.12 Among patients with atherosclerotic cardiovascular disease at high risk of heart failure or in whom heart failure coexists, sodium–glucose cotransporter 2 inhibitors are preferred. C
9.13 For patients with type 2 diabetes and chronic kidney disease, consider use of a sodium–glucose cotransporter 2 inhibitor or glucagon-like peptide 1 receptor agonist shown to reduce risk of chronic kidney disease progression, cardiovascular events, or both. C
9.14 In most patients who need the greater glucose-lowering effect of an injectable medication, glucagon-like peptide 1 receptor agonists are preferred to insulin. B
9.15 Intensification of treatment for patients with type 2 diabetes not meeting treatment goals should not be delayed. B
9.16 The medication regimen should be reevaluated at regular intervals (every 3–6 months) and adjusted as needed to incorporate new patient factors (Table 9.1). E
The American Diabetes Association/European Association for the Study of Diabetes consensus report “Management of Hyperglycemia in Type 2 Diabetes, 2018” (39) recommends a patient-centered approach to choosing appropriate pharmacologic treatment of blood glucose (Fig. 9.1). This includes consideration of efficacy and key patient factors: 1) important comorbidities such as atherosclerotic cardiovascular disease (ASCVD), chronic kidney disease (CKD), and heart failure (HF), 2) hypoglycemia risk, 3) effects on body weight, 4) side effects, 5) cost, and 6) patient preferences. Lifestyle modifications that improve health (see Section 5 “Lifestyle Management”) should be emphasized along with any pharmacologic therapy. See Sections 12 and 13 for recommendations specific for older adults and for children and adolescents with type 2 diabetes, respectively.
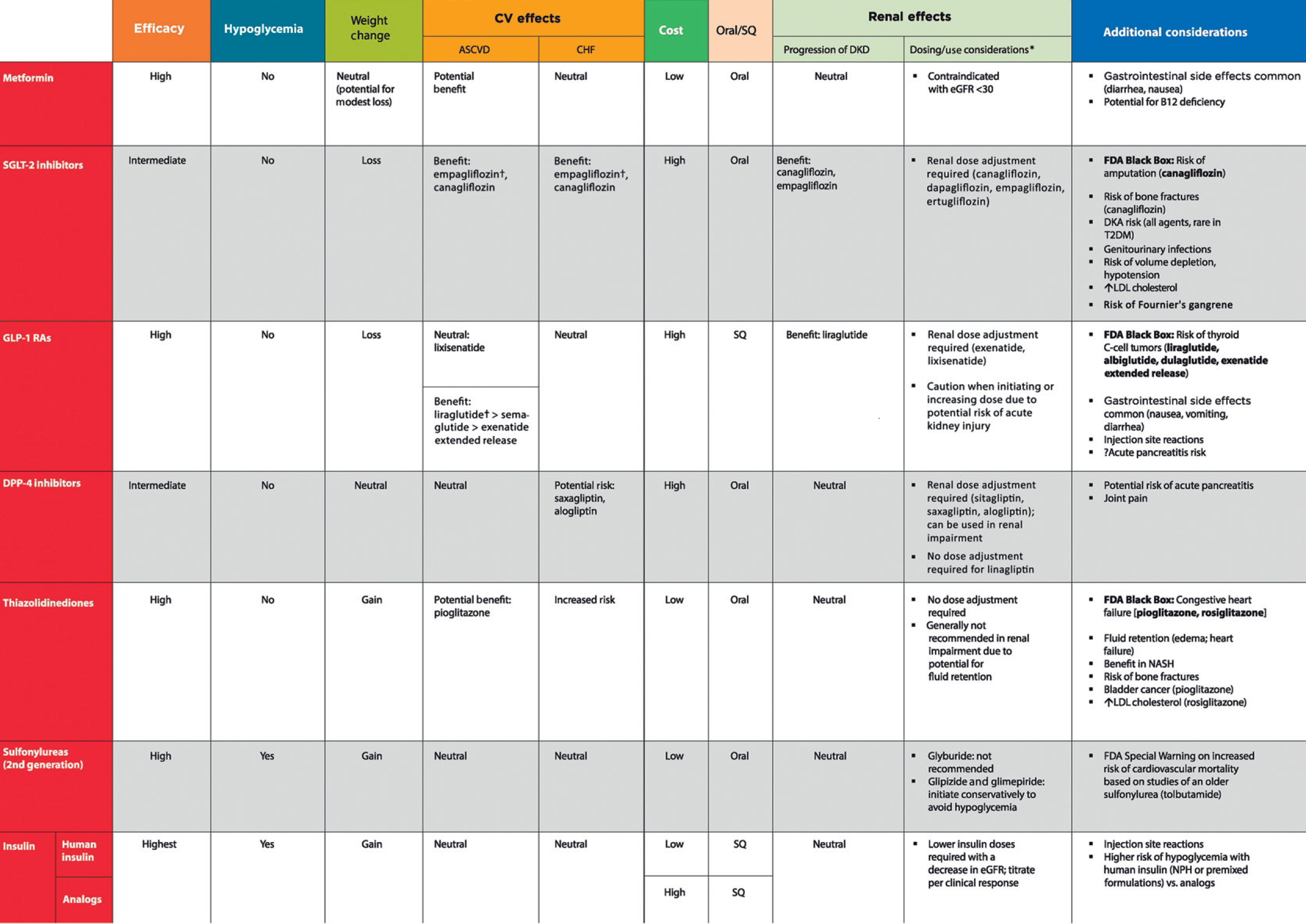
Table 9.1—Drug-specific and patient factors to consider when selecting antihyperglycemic treatment in adults with type 2 diabetes

*For agent-specific dosing recommendations, please refer to the manufacturers’ prescribing information.
†FDA approved for CVD benefit.
CHF, congestive heart failure; CV, cardiovascular; DPP-4, dipeptidyl peptidase 4; DKA, diabetic ketoacidosis; DKD, diabetic kidney disease; GLP-1 RAs, glucagon-like peptide 1 receptor agonists; NASH, nonalcoholic steatohepatitis; SGLT2, sodium–glucose cotransporter 2; SQ, subcutaneous; T2DM, type 2 diabetes
For interactive tool, See here
Figure 9.1 Glucose-lowering medication in type 2 diabetes:
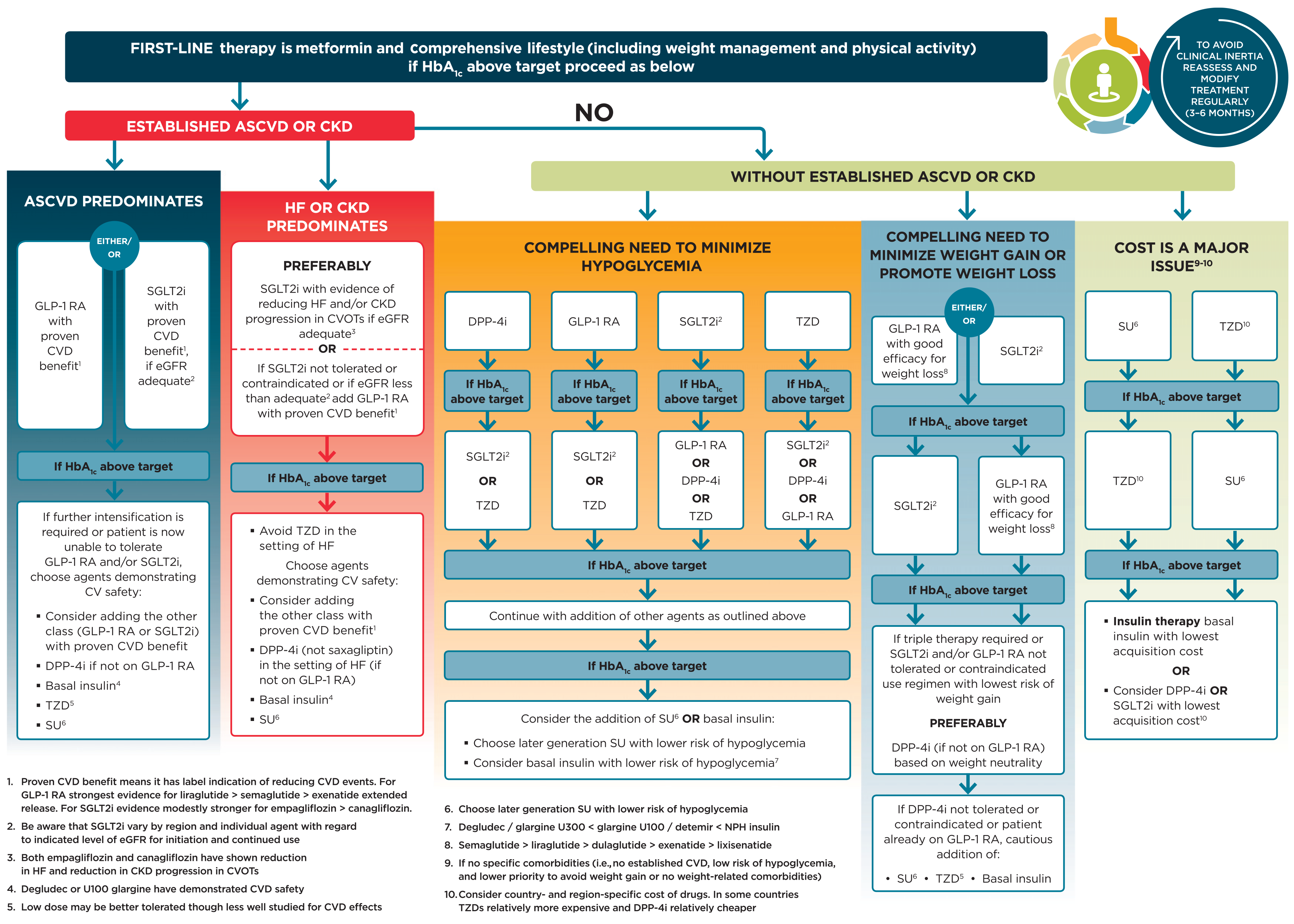
Figure 9.1—Glucose-lowering medication in type 2 diabetes: overall approach. For appropriate context, see Fig. 4.1.
ASCVD, atherosclerotic cardiovascular disease; CKD, chronic kidney disease; CV, cardiovascular; CVD, cardiovascular disease; CVOTs, cardiovascular outcomes trials; DPP-4i, dipeptidyl peptidase 4 inhibitor; eGFR, estimated glomerular filtration rate; GLP-1 RA, glucagon-like peptide 1 receptor agonist; HF, heart failure; SGLT2i, sodium–glucose cotransporter 2 inhibitor; SU, sulfonylurea; TZD, thiazolidinedione. Adapted from Davies et al. (39).
For interactive tool, See here