
1.0.0.0 Introduction
The American Diabetes Association (ADA) “Standards of Medical Care in Diabetes” includes ADA’s current clinical practice recommendations and is intended to provide the components of diabetes care, general treatment goals and guidelines, and tools to evaluate quality of care. Members of the ADA Professional Practice Committee, a multidisciplinary expert committee, are responsible for updating the Standards of Care annually, or more frequently as warranted. For a detailed description of ADA standards, statements, and reports, as well as the evidence-grading system for ADA’s clinical practice recommendations, please refer to the Standards of Care Introduction. Readers who wish to comment on the Standards of Care are invited to do so at professional.diabetes.org/SOC.
There is strong and consistent evidence that obesity management can delay the progression from prediabetes to type 2 diabetes (1-5,) and is beneficial in the treatment of type 2 diabetes (6-17,). In patients with type 2 diabetes who are overweight or obese, modest and sustained weight loss has been shown to improve glycemic control and to reduce the need for glucose-lowering medications (6-8,). Small studies have demonstrated that in patients with type 2 diabetes and obesity, more extreme dietary energy restriction with very low-calorie diets can reduce A1C to <6.5% (48 mmol/mol) and fasting glucose to <126 mg/dL (7.0 mmol/L) in the absence of pharmacologic therapy or ongoing procedures (10,18,19). Weight loss– induced improvements in glycemia are most likely to occur early in the natural history of type 2 diabetes when obesity-associated insulin resistance has caused reversible β-cell dysfunction but insulin secretory capacity remains relatively preserved (8,11,19,20). The goal of this section is to provide evidence-based recommendations for weight-loss therapy, including diet, behavioral, pharmacologic, and surgical interventions, for obesity management as treatment for hyperglycemia in type 2 diabetes.
Suggested citation: American Diabetes Association.
8. Obesity management for the treatment
of type 2 diabetes: Standards of Medical Care in
Diabetesd2019. Diabetes Care 2019;42(Suppl. 1):
S81–S89
© 2018 by the American Diabetes Association.
Readers may use this article as long as the work
is properly cited, the use is educational and not
for profit, and the work is not altered. More information
is available at http://www.diabetesjournals
.org/content/license.
2.0.0.0 ASSESSMENT
Recommendation
8.1 At each patient encounter, BMI should be calculated and documented in the medical record. B
At each routine patient encounter, BMI should be calculated as weight divided by height squared (kg/m2) (21). BMI should be classified to determine the presence of overweight or obesity, discussed with the patient, and documented in the patient record. In Asian Americans, the BMI cutoff points to define overweight and obesity are lower than in other populations (Table 8.1) (22,23). Providers should advise patients who are overweight or obese that, in general, higher BMIs increase the risk of cardiovascular disease and all-cause mortality. Providers should assess each patient’s readiness to achieve weight loss and jointly determine weight-loss goals and intervention strategies. Strategies may include diet, physical activity, behavioral therapy, pharmacologic therapy, and metabolic surgery (Table 8.1). The latter two strategies may be prescribed for carefully selected patients as adjuncts to diet, physical activity, and behavioral therapy.
Table 8.1—Treatment options for overweight and obesity in type 2 diabetes

3.0.0.0 DIET, PHYSICAL ACTIVITY, AND BEHAVIORAL THERAPY
3.1.0.0 Recommendations
Recommendations
8.2 Diet, physical activity, and behavioral therapy designed to achieve and maintain >5% weight loss should be prescribed for patients with type 2 diabetes who are overweight or obese and ready to achieve weight loss. A
8.3 Such interventions should be high intensity (≥16 sessions in 6 months) and focus on diet, physical activity, and behavioral strategies to achieve a 500–750 kcal/day energy deficit. A
8.4 Diets should be individualized, as those that provide the same caloric restriction but differ in protein, carbohydrate, and fat content are equally effective in achieving weight loss. A
8.5 For patients who achieve short-term weight-loss goals, long-term (≥1 year) comprehensive weight-maintenance programs should be prescribed. Such programs should provide at least monthly contact and encourage ongoing monitoring of body weight (weekly or more frequently) and/or other self-monitoring strategies, such as tracking intake, steps, etc.; continued consumption of a reduced-calorie diet; and participation in high levels of physical activity (200– 300 min/week). A
8.6 To achieve weight loss of >5%, short-term (3-month) interventions that use very low-calorie diets (#800 kcal/day) and total meal replacements may be prescribed for carefully selected patients by trained practitioners in medical care settings with close medical monitoring. To maintain weight loss, such programs must incorporate long-term comprehensive weight-maintenance counseling. B
Among patients with type 2 diabetes who are overweight or obese and have inadequate glycemic, blood pressure, and lipid control and/or other obesity-related medical conditions, lifestyle changes that result in modest and sustained weight loss produce clinically meaningful reductions in blood glucose, A1C, and triglycerides (6-8,). Greater weight loss produces even greater benefits, including reductions in blood pressure, improvements in LDL and HDL cholesterol, and reductions in the need for medications to control blood glucose, blood pressure, and lipids (6–8,24), and may result in achievement of glycemic goals in the absence of antihyperglycemia agent use in some patients (25).
3.2.0.0 Look AHEAD Trial
Although the Action for Health in Diabetes (Look AHEAD) trial did not show that an intensive lifestyle intervention reduced cardiovascular events in adults with type 2 diabetes who were overweight or obese (26), it did show the feasibility of achieving and maintaining long-term weight loss in patients with type 2 diabetes. In the Look AHEAD intensive lifestyle intervention group, mean weight loss was 4.7% at 8 years (27). Approximately 50% of intensive lifestyle intervention participants lost and maintained ≥5% and 27% lost and maintained ≥10% of their initial body weight at 8 years (27). Participants randomly assigned to the intensive lifestyle group achieved equivalent risk factor control but required fewer glucose-, blood pressure–, and lipid-lowering medications than those randomly assigned to standard care. Secondary analyses of the Look AHEAD trial and other large cardiovascular outcome studies document other benefits of weight loss in patients with type 2 diabetes, including improvements in mobility, physical and sexual function, and health-related quality of life (28). A post hoc analysis of the Look AHEAD study suggests that heterogeneous treatment effects may have been present. Participants who had moderately or poorly controlled diabetes (A1C ≥6.8% [51 mmol/mol]) as well as both those with well-controlled diabetes (A1C <6.8% [51 mmol/mol]) and good selfreported health were found to have significantly reduced cardiovascular events with intensive lifestyle intervention during follow-up (29).
3.3.0.0 Lifestyle Interventions
Significant weight loss can be attained with lifestyle programs that achieve a 500–750 kcal/day energy deficit, which in most cases is approximately 1,200– 1,500 kcal/day for women and 1,500– 1,800 kcal/day for men, adjusted for the individual’s baseline body weight. Weight loss of 3–5% is the minimum necessary for any clinical benefit (21,30). However, weight-loss benefits are progressive; more intensive weight-loss goals (>5%, >7%, >15%, etc.) may be pursued if needed to achieve a healthy weight and if they can be feasibly and safely attained.
These diets may differ in the types of foods they restrict (such as high-fat or high-carbohydrate foods) but are effective if they create the necessary energy deficit (21,31–33). Use of meal replacement plans prescribed by trained practitioners, with close patient monitoring, can be beneficial. Within the intensive lifestyle intervention group of the Look AHEAD trial, for example, use of a partial meal replacement plan was associated with improvements in diet quality (34). The diet choice should be based on the patient’s health status and preferences.
Intensive behavioral lifestyle interventions should include ≥16 sessions in 6 months and focus on diet, physical activity, and behavioral strategies to achieve an ˜500–750 kcal/day energy deficit. Interventions should be provided by trained interventionists in either individual or group sessions (30).
Patients with type 2 diabetes who are overweight or obese and have lost weight during the 6-month intensive behavioral lifestyle intervention should be enrolled in long-term (≥1 year) comprehensive weight-loss maintenance programs that provide at least monthly contact with a trained interventionist and focus on ongoing monitoring of body weight (weekly or more frequently) and/or other self-monitoring strategies such as tracking intake, steps, etc.; continued consumption of a reduced-calorie diet; and participation in high levels of physical activity (200–300 min/ week (35). Some commercial and proprietary weight-loss programs have shown promising weight-loss results (36).
When provided by trained practitioners in medical care settings with close medical monitoring, short-term (3-month) interventions that use very low-calorie diets (defined as ≤800 kcal/day) and total meal replacements may achieve greater short-term weight loss (10%–15%) than intensive behavioral lifestyle interventions that typically achieve 5% weight loss. However, weight regain following the cessation of very low-calorie diets is greater than following intensive behavioral lifestyle interventions unless a long-term comprehensive weight-loss maintenance program is provided (37,38).
4.0.0.0 PHARMACOTHERAPY
4.1.0.0 Recommendations
Recommendations
8.7 When choosing glucose-lowering medications for overweight or obese patients with type 2 diabetes, consider their effect on weight. E
8.8 Whenever possible, minimize medications for comorbid conditions that are associated with weight gain. E
8.9 Weight-loss medications are effective as adjuncts to diet, physical activity, and behavioral counseling for selected patients with type 2 diabetes and BMI ≥27 kg/m2. Potential benefits must be weighed against the potential risks of the medications. A
8.10 If a patient’s response to weight-loss medications is <5% weight loss after 3 months or if there are significant safety or tolerability issues at any time, the medication should be discontinued and alternative medications or treatment approaches should be considered. A
4.2.0.0 Antihyperglycemia Therapy
Agents associated with varying degrees of weight loss include metformin, α-glucosidase inhibitors, sodium–glucose cotransporter 2 inhibitors, glucagon-like peptide 1 receptor agonists, and amylin mimetics. Dipeptidyl peptidase 4 inhibitors are weight neutral. Unlike these agents, insulin secretagogues, thiazolidinediones, and insulin often cause weight gain (see Section 9 “Pharmacologic Approaches to Glycemic Treatment”).
A recent meta-analysis of 227 randomized controlled trials of antihyperglycemia treatments in type 2 diabetes found that A1C changes were not associated with baseline BMI, indicating that patients with obesity can benefit from the same types of treatments for diabetes as normal-weight patients (39).
4.3.0.0 Concomitant Medications
Providers should carefully review the patient’s concomitant medications and, whenever possible, minimize or provide alternatives for medications that promote weight gain. Medications associated with weight gain include antipsychotics (e.g., clozapine, olanzapine, risperidone, etc.) and antidepressants (e.g., tricyclic antidepressants, selective serotonin reuptake inhibitors, and monoamine oxidase inhibitors), glucocorticoids, injectable progestins, anticonvulsants including gabapentin, and possibly sedating antihistamines and anticholinergics (40).
4.4.0.0 Approved Weight-Loss Medications
The U.S. Food and Drug Administration (FDA) has approved medications for both short-term and long-term weight management as adjuncts to diet, exercise, and behavioral therapy. Nearly all FDA- approved medications for weight loss have been shown to improve glycemic control in patients with type 2 diabetes and delay progression to type 2 diabetes in patients at risk (41). Phentermine is indicated as short-term (≤12 weeks) treatment (42). Five weight-loss medications (or combination medications) are FDA-approved for long-term use (more than a few weeks) by patients with BMI ≥27 kg/m2 with one or more obesity-associated comorbid conditions (e.g., type 2 diabetes, hypertension, and dyslipidemia) who are motivated to lose weight (41). Medications approved by the FDA for the treatment of obesity and their advantages and disadvantages are summarized in Table 8.2. The rationale for weight-loss medications is to help patients to more consistently adhere to low-calorie diets and to reinforce lifestyle changes. Providers should be knowledgeable about the product label and should balance the potential benefits of successful weight loss against the potential risks of the medication for each patient. These medications are contraindicated in women who are pregnant or actively trying to conceive. Women of reproductive potential must be counseled regarding the use of reliable methods of contraception.
Table 8.2—Medications approved by the FDA for the treatment of obesity
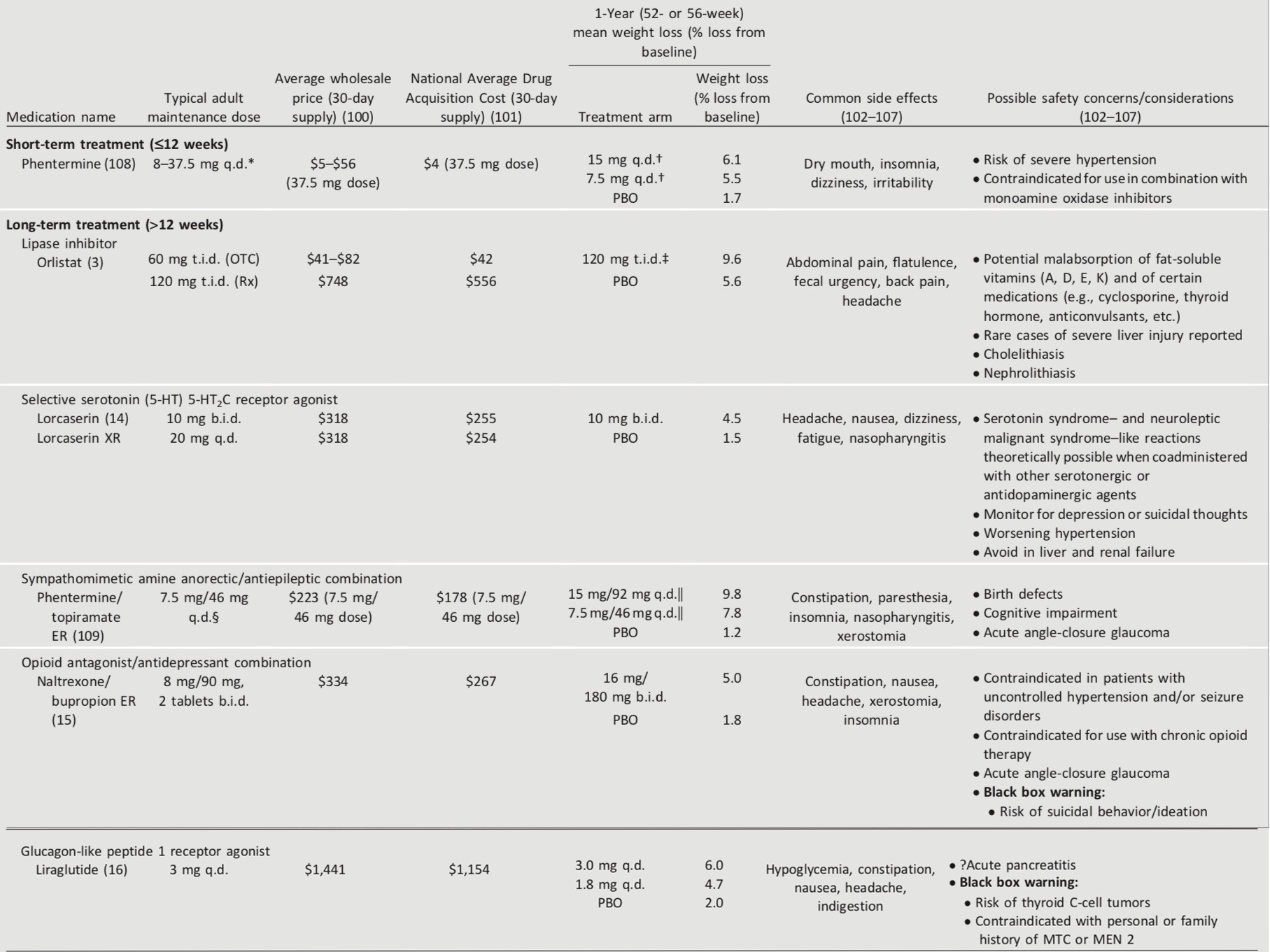
4.5.0.0 Assessing Efficacy and Safety
Efficacy and safety should be assessed at least monthly for the first 3 months of treatment. If a patient’s response is deemed insufficient (weight loss <5%) after 3 months or if there are significant safety or tolerability issues at any time, the medication should be discontinued and alternative medications or treatment approaches should be considered.
5.0.0.0 MEDICAL DEVICES FOR WEIGHT LOSS
Several minimally invasive medical devices have been recently approved by the FDA for short-term weight loss (43). It remains to be seen how these are used for obesity treatment. Given the high cost, extremely limited insurance coverage, and paucity of data in people with diabetes at this time, these are not considered to be the standard of care for obesity management in people with type 2 diabetes.
6.0.0.0 METABOLIC SURGERY
6.1.0.0 Recommendations
Recommendations
8.11 Metabolic surgery should be recommended as an option to treat type 2 diabetes in appropriate surgical candidates with BMI ≥40 kg/m2 (BMI ≥37.5 kg/m2 in Asian Americans) and in adults with BMI 35.0–39.9 kg/m2 (32.5–37.4 kg/m2 in Asian Americans) who do not achieve durable weight loss and improvement in comorbidities (including hyperglycemia) with reasonable nonsurgical methods. A
8.12 Metabolic surgery may be considered as an option for adults with type 2 diabetes and BMI 30.0– 34.9 kg/m2 (27.5–32.4 kg/m2 in Asian Americans) who do not achieve durable weight loss and improvement in comorbidities (including hyperglycemia) with reasonable nonsurgical methods. A
8.13 Metabolic surgery should be performed in high-volume centers with multidisciplinary teams that understand and are experienced in the management of diabetes and gastrointestinal surgery. C
8.14 Long-term lifestyle support and routine monitoring of micronutrient and nutritional status must be provided to patients after surgery, according to guidelines for postoperative management of metabolic surgery by national and international professional societies. C
8.15 People presenting for metabolic surgery should receive a comprehensive readiness and mental health assessment. B
8.16 People who undergo metabolic surgery should be evaluated to assess the need for ongoing mental health services to help them adjust to medical and psychosocial changes after surgery. C
Several gastrointestinal (GI) operations including partial gastrectomies and bariatric procedures (35) promote dramatic and durable weight loss and improvement of type 2 diabetes in many patients. Given the magnitude and rapidity of the effect of GI surgery on hyperglycemia and experimental evidence that rearrangements of GI anatomy similar to those in some metabolic procedures directly affect glucose homeostasis (36), GI interventions have been suggested as treatments for type 2 diabetes, and in that context they are termed “metabolic surgery.”
A substantial body of evidence has now been accumulated, including data from numerous randomized controlled (nonblinded) clinical trials, demonstrating that metabolic surgery achieves superior glycemic control and reduction of cardiovascular risk factors in patients with type 2 diabetes and obesity compared with various lifestyle/medical interventions (17). Improvements in microvascular complications of diabetes, cardiovascular disease, and cancer have been observed only in nonrandomized observational studies (44-53,). Cohort studies attempting to match surgical and nonsurgical subjects suggest that the procedure may reduce longer-term mortality (45).
On the basis of this mounting evidence, several organizations and government agencies have recommended expanding the indications for metabolic surgery to include patients with type 2 diabetes who do not achieve durable weight loss and improvement in comorbidities (including hyperglycemia) with reasonable nonsurgical methods at BMIs as low as 30 kg/m2 (27.5 kg/m2 for Asian Americans) (54-61,). Please refer to “Metabolic Surgery in the Treatment Algorithm for Type 2 Diabetes: A Joint Statement by International Diabetes Organizations” for a thorough review (17).
Randomized controlled trials have documented diabetes remission during postoperative follow-up ranging from 1 to 5 years in 30%–63% of patients with Roux-en-Y gastric bypass (RYGB), which generally leads to greater degrees and lengths of remission compared with other bariatric surgeries (17,62). Available data suggest an erosion of diabetes remission over time (63): 35%–50% or more of patients who initially achieve remission of diabetes eventually experience recurrence. However, the median disease-free period among such individuals following RYGB is 8.3 years (64,65). With or without diabetes relapse, the majority of patients who undergo surgery maintain substantial improvement of glycemic control from baseline for at least 5 (66,67) to 15 (45,46,65,68–70) years.
Exceedingly few presurgical predictors of success have been identified, but younger age, shorter duration of diabetes (e.g., <8 years) (71), nonuse of insulin, maintenance of weight loss, and better glycemic control are consistently associated with higher rates of diabetes remission and/or lower risk of weight regain (45,69,71,72). Greater baseline visceral fat area may also help to predict better postoperative outcomes, especially among Asian American patients with type 2 diabetes, who typically have more visceral fat compared with Caucasians with diabetes of the same BMI (73).
Beyond improving glycemia, metabolic surgery has been shown to confer additional health benefits in randomized controlled trials, including substantial reductions in cardiovascular disease risk factors (17), reductions in incidence of microvascular disease (74), and enhancements in quality of life (66,71,75).
Although metabolic surgery has been shown to improve the metabolic profiles of patients with type 1 diabetes and morbid obesity, establishing the role of metabolic surgery in such patients will require larger and longer studies (76).
Metabolic surgery is more expensive than nonsurgical management strategies, but retrospective analyses and modeling studies suggest that metabolic surgery may be cost-effective or even cost-saving for patients with type 2 diabetes. However, results are largely dependent on assumptions about the long-term effectiveness and safety of the procedures (77,78).
6.2.0.0 Adverse Effects
The safety of metabolic surgery has improved significantly over the past two decades, with continued refinement of minimally invasive approaches (laparoscopic surgery), enhanced training and credentialing, and involvement of multidisciplinary teams. Mortality rates with metabolic operations are typically 0.1%–0.5%, similar to cholecystectomy or hysterectomy (79-83,). Morbidity has also dramatically declined with laparoscopic approaches. Major complications rates (e.g., venous thromboembolism, need for operative reintervention) are 2%–6%, with other minor complications in up to 15% (79-88,), which compare favorably with rates for other commonly performed elective operations (83). Empirical data suggest that proficiency of the operating surgeon is an important factor for determining mortality, complications, reoperations, and readmissions (89).
Longer-term concerns include dumping syndrome (nausea, colic, and diarrhea), vitamin and mineral deficiencies, anemia, osteoporosis, and, rarely (90), severe hypoglycemia. Long-term nutritional and micronutrient deficiencies and related complications occur with variable frequency depending on the type of procedure and require life-long vitamin/nutritional supplementation (91,92). Postprandial hypoglycemia is most likely to occur with RYGB (92,93). The exact prevalence of symptomatic hypoglycemia is unknown. In one study, it affected 11% of 450 patients who had undergone RYGB or vertical sleeve gastrectomy (90). Patients who undergo metabolic surgery may be at increased risk for substance use, including drug and alcohol use and cigarette smoking. Additional potential risks of metabolic surgery that have been described include worsening or new-onset depression and/or anxiety, need for additional GI surgery, and suicidal ideation (94-97,).
People with diabetes presenting for metabolic surgery also have increased rates of depression and other major psychiatric disorders (98). Candidates for metabolic surgery with histories of alcohol, tobacco, or substance abuse; significant depression; suicidal ideation; or other mental health conditions should therefore first be assessed by a mental health professional with expertise in obesity management prior to consideration for surgery (99). Surgery should be postponed in patients with alcohol or substance abuse disorders, significant depression, suicidal ideation, or other mental health conditions until these conditions have been fully addressed. Individuals with preoperative psychopathology should be assessed regularly following metabolic surgery to optimize mental health management and to ensure psychiatric symptoms do not interfere with weight loss and lifestyle changes.
7.0.0.0 References
U.S. National Library of Medicine. Phentermine - phentermine hydrochloride capsule [Internet], 2017. Available from https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid5737eef3b- 9a6b-4ab3-a25c-49d84d2a0197. Accessed 12 September 2018