4.4.0.0 Approved Weight-Loss Medications
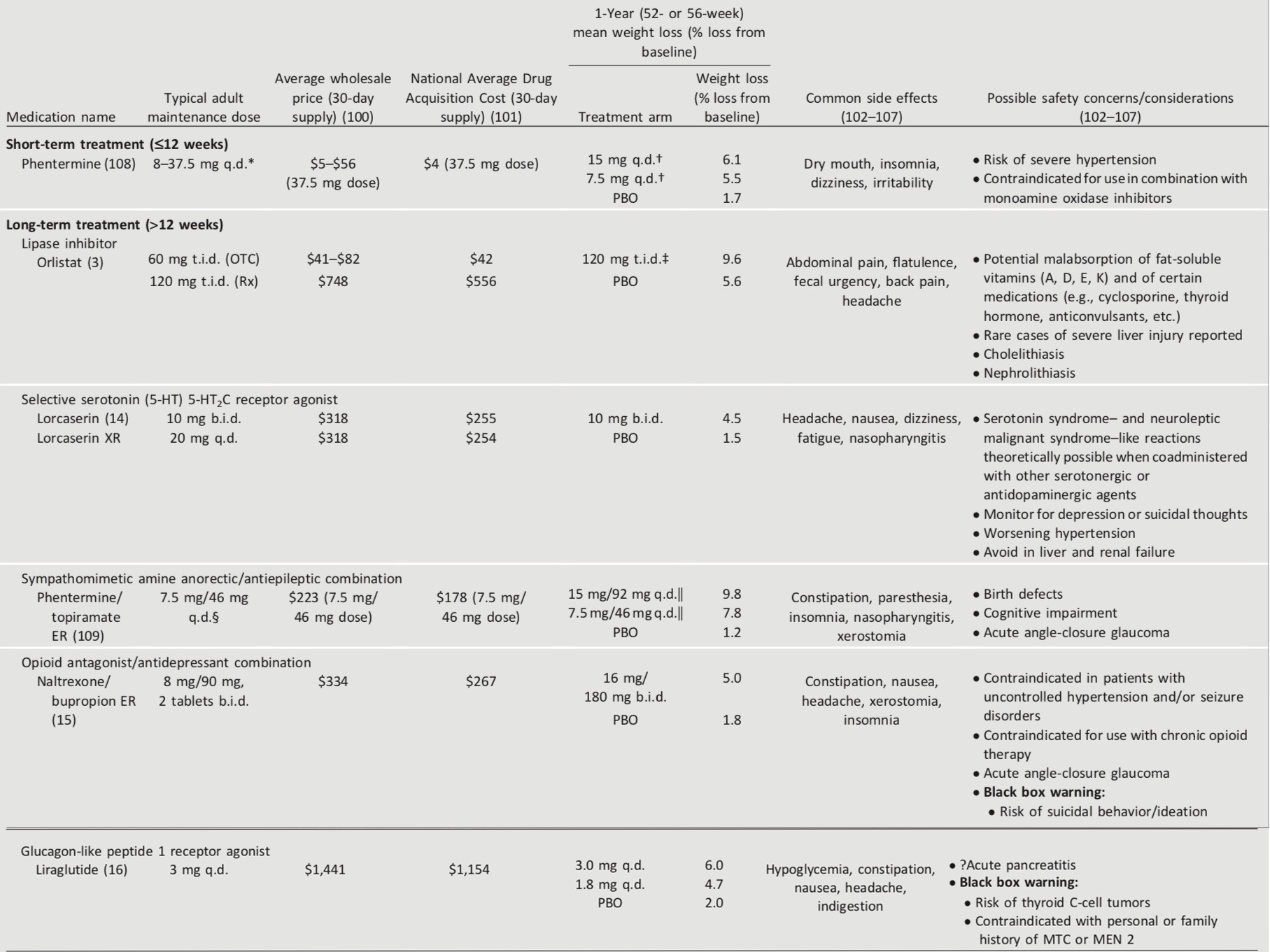
The U.S. Food and Drug Administration (FDA) has approved medications for both short-term and long-term weight management as adjuncts to diet, exercise, and behavioral therapy. Nearly all FDA- approved medications for weight loss have been shown to improve glycemic control in patients with type 2 diabetes and delay progression to type 2 diabetes in patients at risk (41). Phentermine is indicated as short-term (≤12 weeks) treatment (42). Five weight-loss medications (or combination medications) are FDA-approved for long-term use (more than a few weeks) by patients with BMI ≥27 kg/m2 with one or more obesity-associated comorbid conditions (e.g., type 2 diabetes, hypertension, and dyslipidemia) who are motivated to lose weight (41). Medications approved by the FDA for the treatment of obesity and their advantages and disadvantages are summarized in Table 8.2. The rationale for weight-loss medications is to help patients to more consistently adhere to low-calorie diets and to reinforce lifestyle changes. Providers should be knowledgeable about the product label and should balance the potential benefits of successful weight loss against the potential risks of the medication for each patient. These medications are contraindicated in women who are pregnant or actively trying to conceive. Women of reproductive potential must be counseled regarding the use of reliable methods of contraception.
Table 8.2—Medications approved by the FDA for the treatment of obesity

All medications are contraindicated in women who are or may become pregnant. Women of reproductive potential must be counseled regarding the use of reliable methods of contraception. Select safety and side effect information is provided; for a comprehensive discussion of safety considerations, please refer to the prescribing information for each agent. b.i.d., twice daily; ER, extended release; MEN 2, multiple endocrine neoplasia syndrome type 2; MTC, medullary thyroid carcinoma; OTC, over the counter; PBO, placebo; q.d., daily; Rx, prescription; t.i.d, three times daily; XR, extended release.
*Use lowest effective dose; maximum appropriate dose is 37.5 mg.
†Duration of treatment was 28 weeks in a general obese adult population.
‡Enrolled participants had normal (79%) or impaired (21%) glucose tolerance.
§Maximum dose, depending on response, is 15 mg/92 mg q.d.
||Approximately 68% of enrolled participants had type 2 diabetes or impaired glucose tolerance.