
4.4.0.0 Setting and Modifying A1C Goals
Numerous factors must be considered when setting glycemic targets. The ADA proposes general targets appropriate for many patients but emphasizes the importance of individualization based on key patient characteristics. Glycemic targets must be individualized in the context of shared decision making to address the needs and preferences of each patient and the individual characteristics that influence risks and benefits of therapy for each patient.
The factors to consider in individualizing goals are depicted in Figure 6.1 is not designed to be applied rigidly but to be used as a broad construct to guide clinical decision making (40) in both type 1 and type 2 diabetes. More stringent control (such as an A1C of 6.5% [48 mmol/mol] or <7% [53 mmol/mol]) may be recommended if it can be achieved safely and with acceptable burden of therapy and if life expectancy is sufficient to reap benefits of tight control. Less stringent control (A1C up to 8% [64 mmol/mol]) may be recommended if the life expectancy of the patient is such that the benefits of an intensive goal may not be realized, or if the risks and burdens outweigh the potential benefits. Severe or frequent hypoglycemia is an absolute indication for the modification of treatment regimens, including setting higher glycemic goals.
Diabetes is a chronic disease that progresses over decades. Thus, a goal that might be appropriate for an individual early in the course of the disease may change over time. Newly diagnosed patients and/or those without comorbidities that limit life expectancy may benefit from intensive control proven to prevent microvascular complications. Both DCCT/EDIC and UKPDS demonstrated metabolic memory, or a legacy effect, in which a finite period of intensive control yielded benefits that extended for decades after that control ended. Thus, a finite period of intensive control to near-normal A1C may yield enduring benefits even if control is subsequently deintensified as patient characteristics change. Over time, comorbidities may emerge, decreasing life expectancy and the potential to reap benefits from intensive control. Also, with longer duration of disease, diabetes may become more difficult to control, with increasing risks and burdens of therapy. Thus, A1C targets should be reevaluated over time to balance the risks and benefits as patient factors change.
Recommended glycemic targets for many nonpregnant adults are shown in Table 6.2. The recommendations include blood glucose levels that appear to correlate with achievement of an A1C of <7% (53 mmol/mol). The issue of preprandial versus postprandial SMBG targets is complex (41). Elevated post-challenge (2-h oral glucose tolerance test) glucose values have been associated with increased cardiovascular risk independent of fasting plasma glucose in some epidemiologic studies, but intervention trials have not shown postprandial glucose to be a cardiovascular risk factor independent of A1C. In subjects with diabetes, surrogate measures of vascular pathology, such as endothelial dysfunction, are negatively affected by postprandial hyperglycemia. It is clear that postprandial hyperglycemia, like preprandial hyperglycemia, contributes to elevated A1C levels, with its relative contribution being greater at A1C levels that are closer to 7% (53 mmol/mol). However, outcome studies have clearly shown A1C to be the primary predictor of complications, and landmark trials of glycemic control such as the DCCT and UKPDS relied overwhelmingly on pre-prandial SMBG. Additionally, a randomized controlled trial in patients with known CVD found no CVD benefit of insulin regimens targeting postprandial glucose compared with those targeting preprandial glucose (42). Therefore, it is reasonable for postprandial testing to be recommended for individuals who have premeal glucose values within target but have A1C values above target. Measuring postprandial plasma glucose 1–2 h after the start of a meal and using treatments aimed at reducing postprandial plasma glucose values to <180 mg/dL (10.0 mmol/L) may help to lower A1C.
An analysis of data from 470 participants in the ADAG study (237 with type 1 diabetes and 147 with type 2 diabetes) found that actual average glucose levels associated with conventional A1C targets were higher than older DCCT and ADA targets (Table 6.1) (7,43). These findings support that premeal glucose targets may be relaxed without undermining overall glycemic control as measured by A1C. These data prompted the revision in the ADA-recommended premeal glucose target to 80–130 mg/dL (4.4–7.2 mmol/L) but did not affect the definition of hypoglycemia.
Table 6.2—Summary of glycemic recommendations for many nonpregnant adults with diabetes

*More or less stringent glycemic goals may be appropriate for individual patients. Goals should be individualized based on duration of diabetes, age/life expectancy, comorbid conditions, known CVD or advanced microvascular complications, hypoglycemia unawareness, and individual patient considerations.
†Postprandial glucose may be targeted if A1C goals are not met despite reaching preprandial glucose goals. Postprandial glucose measurements should be made 1–2 h after the beginning of the meal, generally peak levels in patients with diabetes.
Figure 6.1 Approach to Individualization of Glycemic Targets
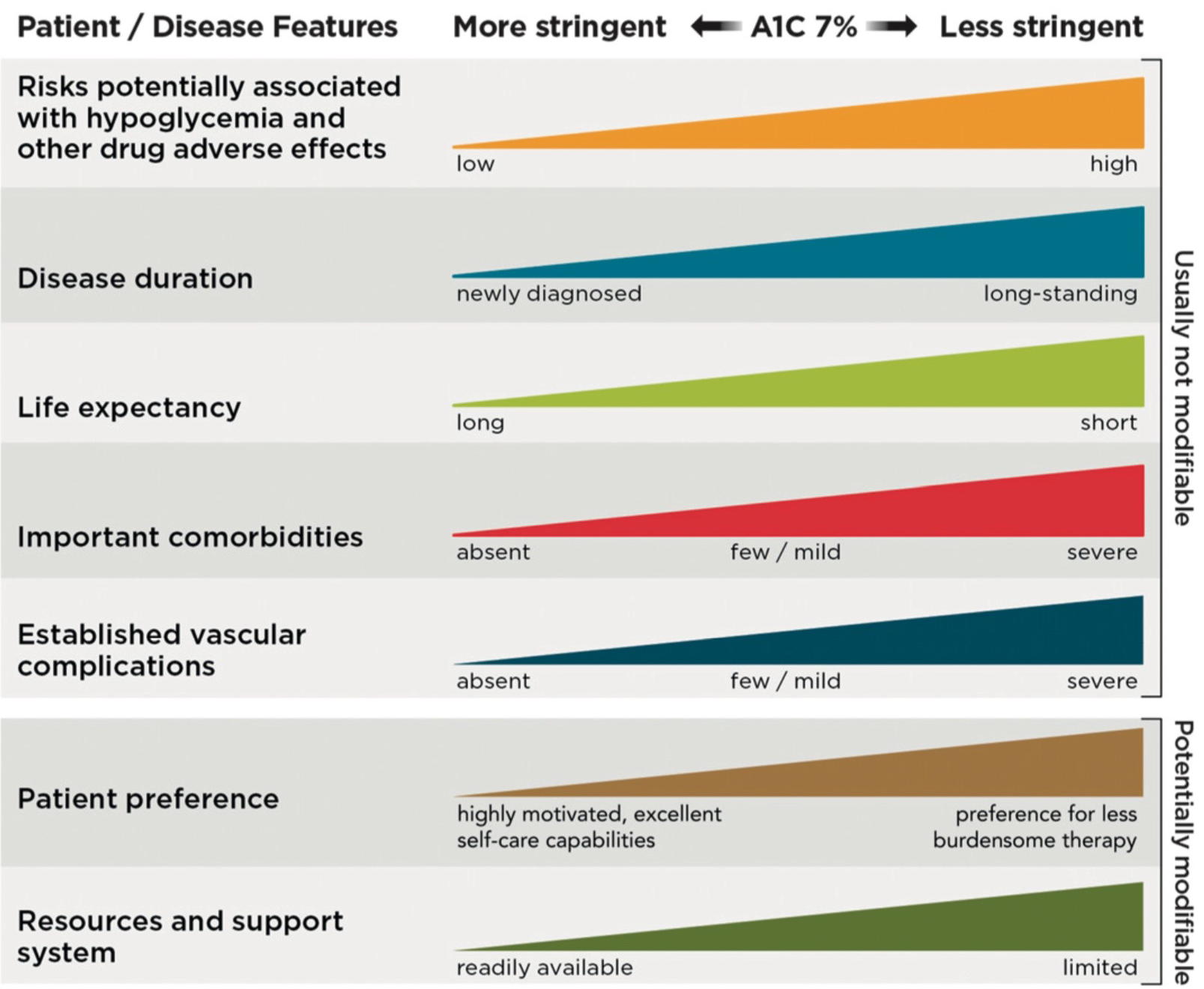
Figure 6.1—Depicted are patient and disease factors used to determine optimal A1C targets. Characteristics and predicaments toward the left justify more stringent efforts to lower A1C; those toward the right suggest less stringent efforts. A1C 7% = 53 mmol/mol. Adapted with permission from Inzucchi et al. (40).
For interactive tool, see here