1.0.0.0 Introduction
The American Diabetes Association (ADA) “Standards of Medical Care in Diabetes” includes ADA’s current clinical practice recommendations and is intended to provide the components of diabetes care, general treatment goals and guidelines, and tools to evaluate quality of care. Members of the ADA Professional Practice Committee, a multidisciplinary expert committee, are responsible for updating the Standards of Care annually, or more frequently as warranted. For a detailed description of ADA standards, statements, and reports, as well as the evidence-grading system for ADA’s clinical practice recommendations, please refer to the Standards of Care Introduction. Readers who wish to comment on the Standards of Care are invited to do so at professional.diabetes.org/SOC.
Lifestyle management is a fundamental aspect of diabetes care and includes diabetes self-management education and support (DSMES), medical nutrition therapy (MNT), physical activity, smoking cessation counseling, and psychosocial care. Patients and care providers should focus together on how to optimize lifestyle from the time of the initial comprehensive medical evaluation, throughout all subsequent evaluations and follow-up, and during the assessment of complications and management of comorbid conditions in order to enhance diabetes care.
Suggested citation: American Diabetes Association. 5. Lifestyle management: Standards of Medical Care in Diabetesd2019. Diabetes Care 2019;42(Suppl. 1):S46–S60 © 2018 by the American Diabetes Association. Readers may use this article as long as the work is properly cited, the use is educational and not for profit, and the work is not altered. More information is available at http://www.diabetesjournals .org/content/license.
2.0.0.0 DM Self-Management Education & Support
2.1.0.0 Recommendations
5.1 In accordance with the national standards for diabetes self-management education and support, all people with diabetes should participate in diabetes self-management education to facilitate the knowledge, skills, and ability necessary for diabetes self-care. Diabetes self-management support is additionally recommended to assist with implementing and sustaining skills and behaviors needed for ongoing self-management. B
5.2 There are four critical times to evaluate the need for diabetes selfmanagement education and support: at diagnosis, annually, when complicating factors arise, and when transitions in care occur. E
5.3 Clinical outcomes, health status, and quality of life are key goals of diabetes self-management education and support that should be measured as part of routine care. C
5.4 Diabetes self-management education and support should be patient centered, may be given in group or individual settings or using technology, and should be communicated with the entire diabetes care team. A
5.5 Because diabetes self-management education and support can improve outcomes and reduce costs B, adequate reimbursement by third-party payers is recommended. E
2.2.0.0 Overview
DSMES services facilitate the knowledge, skills, and abilities necessary for optimal diabetes self-care and incorporate the needs, goals, and life experiences of the person with diabetes. The overall objectives of DSMES are to support informed decision making, self-care behaviors, problem-solving, and active collaboration with the health care team to improve clinical outcomes, health status, and quality of life in a cost-effective manner (1). Providers are encouraged to consider the burden of treatment and the patient’s level of confidence/self-efficacy for management behaviors as well as the level of social and family support when providing DSMES. Patient performance of self-management behaviors, including its effect on clinical outcomes, health status, and quality of life, as well as the psychosocial factors impacting the person’s self-management should be monitored as part of routine clinical care.
In addition, in response to the growing literature that associates potentially judgmental words with increased feelings of shame and guilt, providers are encouraged to consider the impact that language has on building therapeutic relationships and to choose positive, strength-based words and phrases that put people first (2,3). Patient performance of self-management behaviors as well as psychosocial factors impacting the person’s self-management should be monitored. Please see Section 4, “Comprehensive Medical Evaluation and Assessment of Comorbidities,” for more on use of language.
DSMES and the current national standards guiding it (1,4) are based on evidence of benefit. Specifically, DSMES helps people with diabetes to identify and implement effective self-management strategies and cope with diabetes at the four critical time points (described below) (1). Ongoing DSMES helps people with diabetes to maintain effective self-management throughout a lifetime of diabetes as they face new challenges and as advances in treatment become available (5).
Four critical time points have been defined when the need for DSMES is to be evaluated by the medical care provider and/or multidisciplinary team, with referrals made as needed (1):
DSMES focuses on supporting patient empowerment by providing people with diabetes the tools to make informed self-management decisions (6). Diabetes care has shifted to an approach that places the person with diabetes and his or her family at the center of the care model, working in collaboration with health care professionals. Patient-centered care is respectful of and responsive to individual patient preferences, needs, and values. It ensures that patient values guide all decision making (7).
2.3.0.0 Evidence for the Benefits
Studies have found that DSMES is associated with improved diabetes knowledge and self-care behaviors (8), lower A1C (7,9–11), lower self-reported weight (12,13), improved quality of life (10,14), reduced all-cause mortality risk (15), healthy coping (16,17), and reduced health care costs (18-20). Better outcomes were reported for DSMES interventions that were over 10 h in total duration (11), included ongoing support (5,21), were culturally (22,23) and age appropriate (24,25), were tailored to individual needs and preferences, and addressed psychosocial issues and incorporated behavioral strategies (6,16,26,27). Individual and group approaches are effective (13,28,29), with a slight benefit realized by those who engage in both (11). Emerging evidence demonstrates the benefit of Internet-based DSMES services for diabetes prevention and the management of type 2 diabetes (30-32). Technology-enabled diabetes self-management solutions improve A1C most effectively when there is two-way communication between the patient and the health care team, individualized feedback, use of patient-generated health data, and education (32). Current research supports nurses, dietitians, and pharmacists as providers of DSMES who may also develop curriculum (33-35). Members of the DSMES team should have specialized clinical knowledge in diabetes and behavior change principles. Certification as a certified diabetes educator (CDE) or board certified-advanced diabetes management (BC-ADM) certification demonstrates specialized training and mastery of a specific body of knowledge (4). Additionally, there is growing evidence for the role of community health workers (36,37), as well as peer (36-40) and lay leaders (41), in providing ongoing support.
DSMES is associated with an increased use of primary care and preventive services (18,42,43) and less frequent use of acute care and inpatient hospital services (12). Patients who participate in DSMES are more likely to follow best practice treatment recommendations, particularly among the Medicare population, and have lower Medicare and insurance claim costs (19,42). Despite these benefits, reports indicate that only 5–7% of individuals eligible for DSMES through Medicare or a private insurance plan actually receive it (44,45). This low participation may be due to lack of referral or other identified barriers such as logistical issues (timing, costs) and the lack of a perceived benefit (46). Thus, in addition to educating referring providers about the benefits of DSMES and the critical times to refer (1), alternative and innovative models of DSMES delivery need to be explored and evaluated.
2.4.0.0 Reimbursement
Medicare reimburses DSMES when that service meets the national standards (1,4) and is recognized by the American Diabetes Association (ADA) or other approval bodies. DSMES is also covered by most health insurance plans. Ongoing support has been shown to be instrumental for improving outcomes when it is implemented after the completion of education services. DSMES is frequently reimbursed when performed in person. However, although DSMES can also be provided via phone calls and telehealth, these remote versions may not always be reimbursed. Changes in reimbursement policies that increase DSMES access and utilization will result in a positive impact to beneficiaries’ clinical outcomes, quality of life, health care utilization, and costs (47).
3.0.0.0 NUTRITION THERAPY
3.1.0.0 Overview
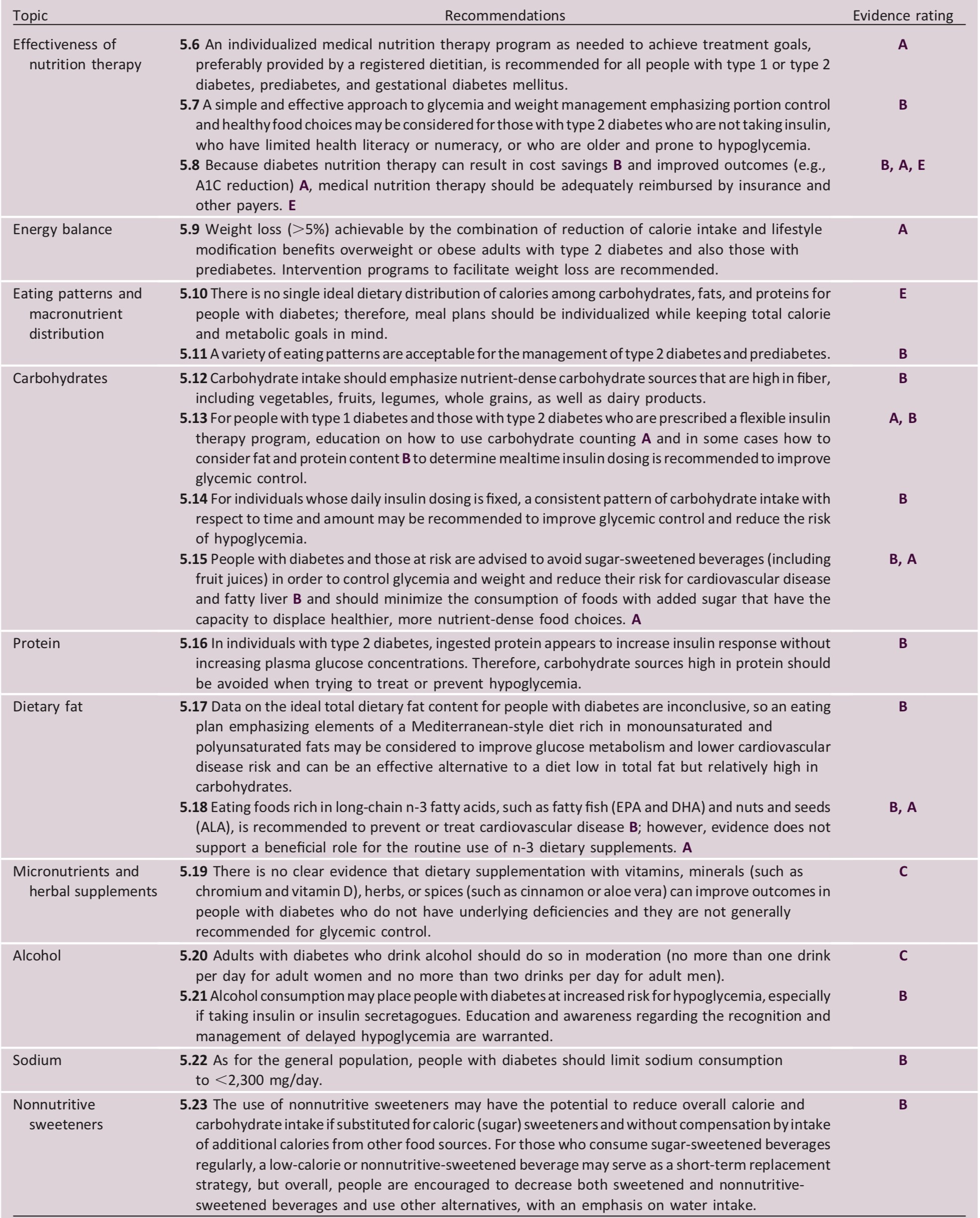
For many individuals with diabetes, the most challenging part of the treatment plan is determining what to eat and following a meal plan. There is not a one-size-fits-all eating pattern for individuals with diabetes, and meal planning should be individualized. Nutrition therapy has an integral role in overall diabetes management, and each person with diabetes should be actively engaged in education, self-management, and treatment planning with his or her health care team, including the collaborative development of an individualized eating plan (35,48). All individuals with diabetes should be offered a referral for individualized MNT provided by a registered dietitian (RD) who is knowledgeable and skilled in providing diabetes-specific MNT (49). MNT delivered by an RD is associated with A1C decreases of 1.0–1.9% for people with type 1 diabetes (50) and 0.3–2% for people with type 2 diabetes (50). See Table 5.1 for specific nutrition recommendations. Because of the progressive nature of type 2 diabetes, lifestyle changes alone may not be adequate to maintain euglycemia over time. However, after medication is initiated, nutrition therapy continues to be an important component and should be integrated with the overall treatment plan (48).
Table 5.1—Medical nutrition therapy recommendations

3.2.0.0 Adults With DM, Goals of Nutrition Therapy
Achieve and maintain body weight goals
Attain individualized glycemic, blood pressure, and lipid goals
Delay or prevent the complications of diabetes
3.3.0.0 Eating Patterns and Meal Planning
Evidence suggests that there is not an ideal percentage of calories from carbohydrate, protein, and fat for all people with diabetes. Therefore, macronutrient distribution should be based on an individualized assessment of current eating patterns, preferences, and metabolic goals. Consider personal preferences (e.g., tradition, culture, religion, health beliefs and goals, economics) as well as metabolic goals when working with individuals to determine the best eating pattern for them (35,51,52). It is important that each member of the health care team be knowledgeable about nutrition therapy principles for people with all types of diabetes and be supportive of their implementation. Emphasis should be on healthful eating patterns containing nutrient-dense foods, with less focus on specific nutrients (53). A variety of eating patterns are acceptable for the management of diabetes (51,54), and a referral to an RD or registered dietitian nutritionist (RDN) is essential to assess the overall nutrition status of, and to work collaboratively with, the patient to create a personalized meal plan that considers the individual’s health status, skills, resources, food preferences, and health goals to coordinate and align with the overall treatment plan including physical activity and medication. The Mediterranean (55,56), Dietary Approaches to Stop Hypertension (DASH) (57-59), and plant-based (60,61) diets are all examples of healthful eating patterns that have shown positive results in research, but individualized meal planning should focus on personal preferences, needs, and goals. In addition, research indicates that lowcarbohydrate eating plans may result in improved glycemia and have the potential to reduce antihyperglycemic medications for individuals with type 2 diabetes (62-64). As research studies on some low-carbohydrate eating plans generally indicate challenges with longterm sustainability, it is important to reassess and individualize meal plan guidance regularly for those interested in this approach. This meal plan is not recommended at this time for women who are pregnant or lactating, people with or at risk for disordered eating, or people who have renal disease, and it should be used with caution in patients taking sodium–glucose cotransporter 2 (SGLT2) inhibitors due to the potential risk of ketoacidosis (65,66). There is inadequate research in type 1 diabetes to support one eating plan over another at this time.
A simple and effective approach to glycemia and weight management emphasizing portion control and healthy food choices should be considered for those with type 2 diabetes who are not taking insulin, who have limited health literacy or numeracy, or who are older and prone to hypoglycemia (50). The diabetes plate method is commonly used for providing basic meal planning guidance (67) as it provides a visual guide showing how to control calories (by featuring a smaller plate) and carbohydrates (by limiting them to what fits in one-quarter of the plate) and puts an emphasis on low-carbohydrate (or nonstarchy) vegetables.
3.4.0.0 Weight Management
Management and reduction of weight is important for people with type 1 diabetes, type 2 diabetes, or prediabetes who have overweight or obesity. Lifestyle intervention programs should be intensive and have frequent follow-up to achieve significant reductions in excess body weight and improve clinical indicators. There is strong and consistent evidence that modest persistent weight loss can delay the progression from prediabetes to type 2 diabetes (51,68,69) (see Section 3 “Prevention or Delay of Type 2 Diabetes”) and is beneficial to the management of type 2 diabetes (see Section 8 “Obesity Management for the Treatment of Type 2 Diabetes”).
Studies of reduced calorie interventions show reductions in A1C of 0.3% to 2.0% in adults with type 2 diabetes, as well as improvements in medication doses and quality of life (50,51). Sustaining weight loss can be challenging (70,71) but has long-term benefits; maintaining weight loss for 5 years is associated with sustained improvements in A1C and lipid levels (72). Weight loss can be attained with lifestyle programs that achieve a 500–750 kcal/day energy deficit or provide ˜1,200–1,500 kcal/day for women and 1,500–1,800 kcal/day for men, adjusted for the individual’s baseline body weight. For many obese individuals with type 2 diabetes, weight loss of at least 5% is needed to produce beneficial outcomes in glycemic control, lipids, and blood pressure (70). It should be noted, however, that the clinical benefits of weight loss are progressive and more intensive weight loss goals (i.e., 15%) may be appropriate to maximize benefit depending on need, feasibility, and safety (73). MNT guidance from an RD/RDN with expertise in diabetes and weight management, throughout the course of a structured weight loss plan, is strongly recommended.
Studies have demonstrated that a variety of eating plans, varying in macronutrient composition, can be used effectively and safely in the short term (1–2 years) to achieve weight loss in people with diabetes. This includes structured low-calorie meal plans that include meal replacements (72-74) and the Mediterranean eating pattern (75) as well as low-carbohydrate meal plans (62). However, no single approach has been proven to be consistently superior (76,77), and more data are needed to identify and validate those meal plans that are optimal with respect to longterm outcomes as well as patient acceptability. The importance of providing guidance on an individualized meal plan containing nutrient-dense foods, such as vegetables, fruits, legumes, dairy, lean sources of protein (including plant-based sources as well as lean meats, fish, and poultry), nuts, seeds, and whole grains, cannot be overemphasized (77), as well as guidance on achieving the desired energy deficit (78-81). Any approach to meal planning should be individualized considering the health status, personal preferences, and ability of the person with diabetes to sustain the recommendations in the plan.
3.5.0.0 Carbohydrates
Studies examining the ideal amount of carbohydrate intake for people with diabetes are inconclusive, although monitoring carbohydrate intake and considering the blood glucose response to dietary carbohydrate are key for improving postprandial glucose control (82,83). The literature concerning glycemic index and glycemic load in individuals with diabetes is complex, often yielding mixed results, though in some studies lowering the glycemic load of consumed carbohydrates has demonstrated A1C reductions of 0.2% to 0.5% (84,85). Studies longer than 12 weeks report no significant influence of glycemic index or glycemic load independent of weight loss on A1C; however, mixed results have been reported for fasting glucose levels and endogenous insulin levels.
For people with type 2 diabetes or prediabetes, low-carbohydrate eating plans show potential to improve glycemia and lipid outcomes for up to 1 year (62–64,86–89). Part of the challenge in interpreting low-carbohydrate research has been due to the wide range of definitions for a low-carbohydrate eating plan (85,86). As research studies on low-carbohydrate eating plans generally indicate challenges with long-term sustainability, it is important to reassess and individualize meal plan guidance regularly for those interested in this approach. Providers should maintain consistent medical oversight and recognize that certain groups are not appropriate for low-carbohydrate eating plans, including women who are pregnant or lactating, children, and people who have renal disease or disordered eating behavior, and these plans should be used with caution for those taking SGLT2 inhibitors due to potential risk of ketoacidosis (65,66). There is inadequate research about dietary patterns for type 1 diabetes to support one eating plan over another at this time.
Most individuals with diabetes report a moderate intake of carbohydrate (44– 46% of total calories) (51). Efforts to modify habitual eating patterns are often unsuccessful in the long term; people generally go back to their usual macronutrient distribution (51). Thus, the recommended approach is to individualize meal plans to meet caloric goals with a macronutrient distribution that is more consistent with the individual’s usual intake to increase the likelihood for long-term maintenance.
As for all individuals in developed countries, both children and adults with diabetes are encouraged to minimize intake of refined carbohydrates and added sugars and instead focus on carbohydrates from vegetables, legumes, fruits, dairy (milk and yogurt), and whole grains. The consumption of sugar-sweetened beverages (including fruit juices) and processed “low-fat” or “nonfat” food products with high amounts of refined grains and added sugars is strongly discouraged (90-92). Individuals with type 1 or type 2 diabetes taking insulin at mealtime should be offered intensive and ongoing education on the need to couple insulin administration with carbohydrate intake. For people whose meal schedule or carbohydrate consumption is variable, regular counseling to help them understand the complex relationship between carbohydrate intake and insulin needs is important. In addition, education on using the insulin-to-carbohydrate ratios for meal planning can assist them with effectively modifying insulin dosing from meal to meal and improving glycemic control (51,82,93–96). Individuals who consume meals containing more protein and fat than usual may also need to make mealtime insulin dose adjustments to compensate for delayed postprandial glycemic excursions (97-99). For individuals on a fixed daily insulin schedule, meal planning should emphasize a relatively fixed carbohydrate consumption pattern with respect to both time and amount (35).
3.6.0.0 Protein
There is no evidence that adjusting the daily level of protein intake (typically 1–1.5 g/kg body weight/day or 15–20% total calories) will improve health in individuals without diabetic kidney disease, and research is inconclusive regarding the ideal amount of dietary protein to optimize either glycemic control or cardiovascular disease (CVD) risk (84,100). Therefore, protein intake goals should be individualized based on current eating patterns. Some research has found successful management of type 2 diabetes with meal plans including slightly higher levels of protein (20–30%), which may contribute to increased satiety (58).
Those with diabetic kidney disease (with albuminuria and/or reduced estimated glomerular filtration rate) should aim to maintain dietary protein at the recommended daily allowance of 0.8 g/kg body weight/day. Reducing the amount of dietary protein below the recommended daily allowance is not recommended because it does not alter glycemic measures, cardiovascular risk measures, or the rate at which glomerular filtration rate declines (101,102).
In individuals with type 2 diabetes, protein intake may enhance or increase the insulin response to dietary carbohydrates (103). Therefore, use of carbohydrate sources high in protein (such as milk and nuts) to treat or prevent hypoglycemia should be avoided due to the potential concurrent rise in endogenous insulin.
3.7.0.0 Fats
The ideal amount of dietary fat for individuals with diabetes is controversial. The National Academy of Medicine has defined an acceptable macronutrient distribution for total fat for all adults to be 20–35% of total calorie intake (104). The type of fats consumed is more important than total amount of fat when looking at metabolic goals and CVD risk, and it is recommended that the percentage of total calories from saturated fats should be limited (75,90,105–107). Multiple randomized controlled trials including patients with type 2 diabetes have reported that a Mediterranean-style eating pattern (75,108–113), rich in polyunsaturated and monounsaturated fats, can improve both glycemic control and blood lipids. However, supplements do not seem to have the same effects as their whole-food counterparts. A systematic review concluded that dietary supplements with n-3 fatty acids did not improve glycemic control in individuals with type 2 diabetes (84). Randomized controlled trials also do not support recommending n-3 supplements for primary or secondary prevention of CVD (114-118). People with diabetes should be advised to follow the guidelines for the general population for the recommended intakes of saturated fat, dietary cholesterol, and trans fat (90). In general, trans fats should be avoided. In addition, as saturated fats are progressively decreased in the diet, they should be replaced with unsaturated fats and not with refined carbohydrates (112).
3.8.0.0 Sodium
As for the general population, people with diabetes are advised to limit their sodium consumption to <2,300 mg/day (35). Restriction below 1,500 mg, even for those with hypertension, is generally not recommended (119-121). Sodium intake recommendations should take into account palatability, availability, affordability, and the difficulty of achieving low-sodium recommendations in a nutritionally adequate diet (122).
3.9.0.0 Micronutrients and Supplements
There continues to be no clear evidence of benefit from herbal or nonherbal (i.e., vitamin or mineral) supplementation for people with diabetes without underlying deficiencies (35). Metformin is associated with vitamin B12 deficiency, with a recent report from the Diabetes Prevention Program Outcomes Study (DPPOS) suggesting that periodic testing of vitamin B12 levels should be considered in patients taking metformin, particularly in those with anemia or peripheral neuropathy (123). Routine supplementation with antioxidants, such as vitamins E and C and carotene, is not advised due to lack of evidence of efficacy and concern related to long-term safety. In addition, there is insufficient evidence to support the routine use of herbals and micronutrients, such as cinnamon (124), curcumin, vitamin D (125), or chromium, to improve glycemia in people with diabetes (35,126). However, for special populations, including pregnant or lactating women, older adults, vegetarians, and people following very low-calorie or low-carbohydrate diets, a multivitamin may be necessary.
3.10.0.0 Alcohol
Moderate alcohol intake does not have major detrimental effects on long-term blood glucose control in people with diabetes. Risks associated with alcohol consumption include hypoglycemia (particularly for those using insulin or insulin secretagogue therapies), weight gain, and hyperglycemia (for those consuming excessive amounts) (35,126). People with diabetes can follow the same guidelines as those without diabetes if they choose to drink. For women, no more than one drink per day, and for men, no more than two drinks per day is recommended (one drink is equal to a 12-oz beer, a 5-oz glass of wine, or 1.5 oz of distilled spirits).
3.11.0.0 Nonnutritive Sweeteners
For some people with diabetes who are accustomed to sugar-sweetened products, nonnutritive sweeteners (containing few or no calories) may be an acceptable substitute for nutritive sweeteners (those containing calories such as sugar, honey, agave syrup) when consumed in moderation. While use of nonnutritive sweeteners does not appear to have a significant effect on glycemic control (127), they can reduce overall calorie and carbohydrate intake (51). Most systematic reviews and metaanalyses show benefits for nonnutritive sweetener use in weight loss (128,129); however, some research suggests an association with weight gain (130). Regulatory agencies set acceptable daily intake levels for each nonnutritive sweetener, defined as the amount that can be safely consumed over a person’s lifetime (35,131). For those who consume sugar-sweetened beverages regularly, a low-calorie or nonnutritive-sweetened beverage may serve as a short-term replacement strategy, but overall, people are encouraged to decrease both sweetened and nonnutritive-sweetened beverages and use other alternatives, with an emphasis on water intake (132).
4.0.0.0 Physical Activity
4.1.0.0 Overview
Physical activity is a general term that includes all movement that increases energy use and is an important part of the diabetes management plan. Exercise is a more specific form of physical activity that is structured and designed to improve physical fitness. Both physical activity and exercise are important.
Exercise has been shown to improve blood glucose control, reduce cardiovascular risk factors, contribute to weight loss, and improve well-being (133). Physical activity is as important for those with type 1 diabetes as it is for the general population, but its specific role in the prevention of diabetes complications and the management of blood glucose is not as clear as it is for those with type
2 diabetes. A recent study suggested that the percentage of people with diabetes who achieved the recommended exercise level per week (150 min) varied by race. Objective measurement by accelerometer showed that 44.2%, 42.6%, and 65.1% of whites, African Americans, and Hispanics, respectively, met the threshold (134). It is important for diabetes care management teams to understand the difficulty that many patients have reaching recommended treatment targets and to identify individualized approaches to improve goal achievement.
Moderate to high volumes of aerobic activity are associated with substantially lower cardiovascular and overall mortality risks in both type 1 and type 2 diabetes (135). A recent prospective observational study of adults with type 1 diabetes suggested that higher amounts of physical activity led to reduced cardiovascular mortality after a mean follow-up time of 11.4 years for patients with and without chronic kidney disease (136). Additionally, structured exercise interventions of at least 8 weeks’ duration have been shown to lower A1C by an average of 0.66% inpeoplewithtype 2 diabetes, even without a significant change in BMI (137). There are also considerable data for the health benefits (e.g., increased cardiovascular fitness, greater muscle strength, improved insulin sensitivity, etc.) of regular exercise for those with type 1 diabetes (138). A recent study suggested that exercise training in type 1 diabetes may also improve several important markers such as triglyceride level, LDL, waist circumference, and body mass (139). Higher levels of exercise intensity are associated with greater improvements in A1C and in fitness (140). Other benefits include slowing the decline in mobility among overweight patients with diabetes (141). The ADA position statement “Physical Activity/Exercise and Diabetes” reviews the evidence for the benefits of exercise in people with type 1 and type 2 diabetes and offers specific recommendation (142).
4.2.0.0 Recommendations
5.24 Children and adolescents with type 1 or type 2 diabetes or prediabetes should engage in 60 min/day or more of moderate- or vigorous-intensity aerobic activity, with vigorous muscle-strengthening and bone-strengthening activities at least 3 days/week. C
5.25 Most adults with type 1 C and type 2 B diabetes should engage in 150 min or more of moderate-to-vigorous intensity aerobic activity per week, spread over at least 3 days/week, with no more than 2 consecutive days without activity. Shorter durations (minimum 75 min/week) of vigorous-intensity or interval training may be sufficient for younger and more physically fit individuals.
5.26 Adults with type 1 C and type 2 B diabetes should engage in 2–3 sessions/week of resistance exercise on nonconsecutive days.
5.27 All adults, and particularly those with type 2 diabetes, should decrease the amount of time spent in daily sedentary behavior. B Prolonged sitting should be interrupted every 30 min for blood glucose benefits, particularly in adults with type 2 diabetes. C
5.28 Flexibility training and balance training are recommended 2–3 times/week for older adults with diabetes. Yoga and tai chi may be included based on individual preferences to increase flexibility, muscular strength, and balance. C
4.3.0.0 Exercise and Children
All children, including children with diabetes or prediabetes, should be encouraged to engage in regular physical activity. Children should engage in at least 60 min of moderate-to-vigorous aerobic activity every day with muscle- and bone-strengthening activities at least 3 days per week (143). In general, youth with type 1 diabetes benefit from being physically active, and an active lifestyle should be recommended to all (144). Youth with type 1 diabetes who engage in more physical activity may have better health-related quality of life (145).
4.4.0.0 Frequency and Type of Physical Activity
People with diabetes should perform aerobic and resistance exercise regularly (142). Aerobic activity bouts should ideally last at least 10 min, with the goal of ˜30 min/day or more, most days of the week for adults with type 2 diabetes. Daily exercise, or at least not allowing more than 2 days to elapse between exercise sessions, is recommended to decrease insulin resistance, regardless of diabetes type (146,147). Over time, activities should progress in intensity, frequency, and/or duration to at least 150 min/week of moderate-intensity exercise. Adults able to run at 6 miles/h (9.7 km/h) for at least 25 min can benefit sufficiently from shorter-intensity activity (75 min/week) (142). Many adults, including most with type 2 diabetes, would be unable or unwilling to participate in such intense exercise and should engage in moderate exercise for the recommended duration. Adults with diabetes should engage in 2–3 sessions/ week of resistance exercise on nonconsecutive days (148). Although heavier resistance training with free weights and weight machines may improve glycemic control and strength (149), resistance training of any intensity is recommended to improve strength, balance, and the ability to engage in activities of daily living throughout the life span. Providers and staff should help patients set stepwise goals toward meeting the recommended exercise targets. Recent evidence supports that all individuals, including those with diabetes, should be encouraged to reduce the amount of time spent being sedentary (e.g., working at a computer, watching TV) by breaking up bouts of sedentary activity (>30 min) by briefly standing, walking, or performing other light physical activities (150,151). Avoiding extended sedentary periods may help prevent type 2 diabetes for those at risk and may also aid in glycemic control for those with diabetes.
A wide range of activities, including yoga, tai chi, and other types, can have significant impacts on A1C, flexibility, muscle strength, and balance (133,152,153). Flexibility and balance exercises may be particularly important in older adults with diabetes to maintain range of motion, strength, and balance (142).
4.5.0.0 Physical Activity and Glycemic Control
Clinical trials have provided strong evidence for the A1C-lowering value of resistance training in older adults with type 2 diabetes (154) and for an additive benefit of combined aerobic and resistance exercise in adults with type 2 diabetes (155). If not contraindicated, patients with type 2 diabetes should be encouraged to do at least two weekly sessions of resistance exercise (exercise with free weights or weight machines), with each session consisting of at least one set (group of consecutive repetitive exercise motions) of five or more different resistance exercises involving the large muscle groups (154).
For type 1 diabetes, although exercise in general is associated with improvement in disease status, care needs to be taken in titrating exercise with respect to glycemic management. Each individual with type 1 diabetes has a variable glycemic response to exercise. This variability should be taken into consideration when recommending the type and duration of exercise for a given individual (138).
Women with preexisting diabetes, particularly type 2 diabetes, and those at risk for or presenting with gestational diabetes mellitus should be advised to engage in regular moderate physical activity prior to and during their pregnancies as tolerated (142).
4.6.0.0 Pre-exercise Evaluation
As discussed more fully in Section 10 “Cardiovascular Disease and Risk Management,” the best protocol for assessing asymptomatic patients with diabetes for coronary artery disease remains unclear. The ADA consensus report “Screening for Coronary Artery Disease in Patients With Diabetes” (156) concluded that routine testing is not recommended. However, providers should perform a careful history, assess cardiovascular risk factors, and be aware of the atypical presentation of coronary artery disease in patients with diabetes. Certainly, high-risk patients should be encouraged to start with short periods of low-intensity exercise and slowly increase the intensity and duration as tolerated. Providers should assess patients for conditions that might contraindicate certain types of exercise or predispose to injury, such as uncontrolled hypertension, untreated proliferative retinopathy, autonomic neuropathy, peripheral neuropathy, and a history of foot ulcers or Charcot foot. The patient’s age and previous physical activity level should be considered. The provider should customize the exercise regimen to the individual’s needs. Those with complications may require a more thorough evaluation prior to beginning an exercise program (138).
4.7.0.0 Hypoglycemia
In individuals taking insulin and/or insulin secretagogues, physical activity may cause hypoglycemia if the medication dose or carbohydrate consumption is not altered. Individuals on these therapies may need to ingest some added carbohydrate if pre-exercise glucose levels are <90 mg/dL (5.0 mmol/L), depending on whether they are able to lower insulin doses during the workout (such as with an insulin pump or reduced pre-exercise insulin dosage), the time of day exercise is done, and the intensity and duration of the activity (138,142). In some patients, hypoglycemia after exercise may occur and last for several hours due to increased insulin sensitivity. Hypoglycemia is less common in patients with diabetes who are not treated with insulin or insulin secretagogues, and no routine preventive measures for hypoglycemia are usually advised in these cases. Intense activities may actually raise blood glucose levels instead of lowering them, especially if pre-exercise glucose levels are elevated (157).
4.8.0.0 Exercise in the Presence of Specific Long-term Compli. of DM
4.8.1.0 Retinopathy
See Section 11 “Microvascular Complications and Foot Care” for more information on these long-term complications.
Retinopathy
If proliferative diabetic retinopathy or severe nonproliferative diabetic retinopathy is present, then vigorous-intensity aerobic or resistance exercise may be contraindicated because of the risk of triggering vitreous hemorrhage or retinal detachment (158). Consultation with an ophthalmologist prior to engaging in an intense exercise regimen may be appropriate.
4.8.2 Peripheral Neuropathy
Decreased pain sensation and a higher pain threshold in the extremities result in an increased risk of skin breakdown, infection, and Charcot joint destruction with some forms of exercise. Therefore, a thorough assessment should be done to ensure that neuropathy does not alter kinesthetic or proprioceptive sensation during physical activity, particularly in those with more severe neuropathy. Studies have shown that moderate-intensity walking may not lead to an increased risk of foot ulcers or reulceration in those with peripheral neuropathy who use proper footwear (159). In addition, 150 min/week of moderate exercise was reported to improve outcomes in patients with prediabetic neuropathy (160). All individuals with peripheral neuropathy should wear proper footwear and examine their feet daily to detect lesions early. Anyone with a foot injury or open sore should be restricted to non–weight-bearing activities.
4.8.3.0 Autonomic Neuropathy
Autonomic neuropathy can increase the risk of exercise-induced injury or adverse events through decreased cardiac responsiveness to exercise, postural hypotension, impaired thermoregulation, impaired night vision due to impaired papillary reaction, and greater susceptibility to hypoglycemia (161). Cardiovascular autonomic neuropathy is also an independent risk factor for cardiovascular death and silent myocardial ischemia (162). Therefore, individuals with diabetic autonomic neuropathy should undergo cardiac investigation before beginning physical activity more intense than that to which they are accustomed.
4.8.4.0 Diabetic Kidney Disease
Physical activity can acutely increase urinary albumin excretion. However, there is no evidence that vigorous-intensity exercise increases the rate of progression of diabetic kidney disease, and there appears to be no need for specific exercise restrictions for people with diabetic kidney disease in general (158).
5.0.0.0 Smoking Cessation: Tobacco and e-Cigarettes
5.1.0.0 Overview
Results from epidemiological, case-control, and cohort studies provide convincing evidence to support the causal link between cigarette smoking and health risks (163). Recent data show tobacco use is higher among adults with chronic conditions (164) as well as in adolescents and young adults with diabetes (165). Smokers with diabetes (and people with diabetes exposed to second-hand smoke) have a heightened risk of CVD, premature death, microvascular complications, and worse glycemic control when compared with nonsmokers (166,167). Smoking may have a role in the development of type 2 diabetes (168-171).
The routine and thorough assessment of tobacco use is essential to prevent smoking or encourage cessation. Numerous large randomized clinical trials have demonstrated the efficacy and cost-effectiveness of brief counseling in smoking cessation, including the use of telephone quit lines, in reducing tobacco use. Pharmacologic therapy to assist with smoking cessation in people with diabetes has been shown to be effective (172), and for the patient motivated to quit, the addition of pharmacologic therapy to counseling is more effective than either treatment alone (173). Special considerations should include assessment of level of nicotine dependence, which is associated with difficulty in quitting and relapse (174). Although some patients may gain weight in the period shortly after smoking cessation (175), recent research has demonstrated that this weight gain does not diminish the substantial CVD benefit realized from smoking cessation (176). One study in smokers with newly diagnosed type 2 diabetes found that smoking cessation was associated with amelioration of metabolic parameters and reduced blood pressure and albuminuria at 1 year (177).
In recent years e-cigarettes have gained public awareness and popularity because of perceptions that e-cigarette use is less harmful than regular cigarette smoking (178,179). Nonsmokers should be advised not to use e-cigarettes (180,181). There are no rigorous studies that have demonstrated that e-cigarettes are a healthier alternative to smoking or that e-cigarettes can facilitate smoking cessation (182). On the contrary, a recently published pragmatic trial found that use of e-cigarettes for smoking cessation was not more effective than “usual care,” which included access to educational information on the health benefits of smoking cessation, strategies to promote cessation, and access to a free text-messaging service that provided encouragement, advice, and tips to facilitate smoking cessation (183). Several organizations have called for more research on the short- and long-term safety and health effects of e-cigarettes (184-186).
5.2.0.0 Recommendations
5.29 Advise all patients not to use cigarettes and other tobacco products A or e-cigarettes. B
5.30 Include smoking cessation counseling and other forms of treatment as a routine component of diabetes care. A
6.0.0.0 Psychosocial Issues
6.1.0.0 Overview
Please refer to the ADA position statement “Psychosocial Care for People With Diabetes” for a list of assessment tools and additional details (187).
Complex environmental, social, behavioral, and emotional factors, known as psychosocial factors, influence living with diabetes, both type 1 and type 2, and achieving satisfactory medical outcomes and psychological well-being. Thus, individuals with diabetes and their families are challenged with complex, multifaceted issues when integrating diabetes care into daily life.
Emotional well-being is an important part of diabetes care and self-management. Psychological and social problems can impair the individual’s (188-190) or family’s (191) ability to carry outdiabetes care tasks and therefore potentially compromise health status. There are opportunities for the clinician to routinely assess psychosocial status in a timely and efficient manner for referral to appropriate services. A systematic review and meta-analysis showed that psychosocial interventions modestly but significantly improved A1C (standardized mean difference –0.29%) and mental health outcomes (192). However, there was a limited association between the effects on A1C and mental health, and no intervention characteristics predicted benefit on both outcomes.
6.2.0.0 Recommendations
Recommendations
5.31 Psychosocial care should be integrated with a collaborative, patient-centered approach and provided to all people with diabetes, with the goals of optimizing health outcomes and health-related quality of life. A
5.32 Psychosocial screening and follow-up may include, but are not limited to, attitudes about diabetes, expectations for medical management and outcomes, affect or mood, general and diabetes-related quality of life, available resources (financial, social, and emotional), and psychiatric history. E
5.33 Providers should consider assessment for symptoms of diabetes distress, depression, anxiety, disordered eating, and cognitive capacities using patient-appropriate standardized and validated tools at the initial visit, at periodic intervals, and when there is a change in disease, treatment, or life circumstance. Including caregivers and family members in this assessment is recommended. B
5.34 Consider screening older adults (aged ≥65 years) with diabetes for cognitive impairment and depression. B
6.3.0.0 Screening
Key opportunities for psychosocial screening occur at diabetes diagnosis, during regularly scheduled management visits, during hospitalizations, with new onset of complications, or when problems with glucose control, quality of life, or self-management are identified (1). Patients are likely to exhibit psychological vulnerability at diagnosis, when their medical status changes (e.g., end of the honeymoon period), when the need for intensified treatment is evident, and when complications are discovered.
Providers can start with informal verbal inquires, for example, by asking if there have been changes in mood during the past 2 weeks or since the patient’s last visit. Providers should consider asking if there are new or different barriers to treatment and self-management, such as feeling overwhelmed or stressed by diabetes or other life stressors. Standardized and validated tools for psychosocial monitoring and assessment can also be used by providers (187), with positive findings leading to referral to a mental health provider specializing in diabetes for comprehensive evaluation, diagnosis, and treatment.
6.4.0.0 Diabetes Distress
6.4.1.0 Overview
Diabetes distress (DD) is very common and is distinct from other psychological disorders (193-195). DD refers to significant negative psychological reactions related to emotional burdens and worries specific to an individual’s experience in having to manage a severe, complicated, and demanding chronic disease such as diabetes (194-196). The constant behavioral demands (medication dosing, frequency, and titration; monitoring blood glucose, food intake, eating patterns, and physical activity) of diabetes self-management and the potential or actuality of disease progression are directly associated with reports of DD (194). The prevalence of DD is reported to be 18–45% with an incidence of 38–48% over 18 months (196). In the second Diabetes Attitudes, Wishes and Needs (DAWN2) study, significant DD was reported by 45% of the participants, but only 24% reported that their health care teams asked them how diabetes affected their lives (193). High levels of DD significantly impact medicationtaking behaviors and are linked to higher A1C, lower self-efficacy, and poorer dietary and exercise behaviors (17,194, 196). DSMES has been shown to reduce DD (17). It may be helpful to provide counseling regarding expected diabetes-related versus generalized psychological distress at diagnosis and when disease state or treatment changes (197).
DD should be routinely monitored (198) using patient-appropriate validated measures (187). If DD is identified, the person should be referred for specific diabetes education to address areas of diabetes self-care that are most relevant to the patient and impact clinical management. People whose self-care remains impaired after tailored diabetes education should be referred by their care team to a behavioral health provider for evaluation and treatment.
Other psychosocial issues known to affect self-management and health outcomes include attitudes about the illness, expectations for medical management and outcomes, available resources (financial, social, and emotional) (199), and psychiatric history. For additional information on psychiatric comorbidities (depression, anxiety, disordered eating, and serious mental illness), please refer to Section 4 “Comprehensive Medical Evaluation and Assessment of Comorbidities.”
6.4.2.0 Recommendations
5.35 Routinely monitor people with diabetes for diabetes distress, particularly when treatment targets are not met and/or at the onset of diabetes complications. B
6.5.0.0 Referral to a Mental Health Specialist
Indications for referral to a mental health specialist familiar with diabetes management may include positive screening for overall stress related to work-life balance, DD, diabetes management difficulties, depression, anxiety, disordered eating, and cognitive dysfunction (see Table 5.2 for a complete list). It is preferable to incorporate psychosocial assessment and treatment into routine care rather than waiting for a specific problem or deterioration in metabolic or psychological status to occur (26,193). Providers should identify behavioral and mental health providers, ideally those who are knowledgeable about diabetes treatment and the psychosocial aspects of diabetes, to whom they can refer patients. The ADA provides a list of mental health providers who have received additional education in diabetes at the ADA Mental Health Provider Directory (professional. diabetes.org/ada-mental-health-provider-directory). Ideally, psychosocial care providers should be embedded in diabetes care settings. Although the clinician may not feel qualified to treat psychological problems (200), optimizing the patient-provider relationship as a foundation may increase the likelihood of the patient accepting referral for other services. Collaborative care interventions and a team approach have demonstrated efficacy in diabetes self-management, outcomes of depression, and psychosocial functioning (17,201).
Table 5.2—Situations that warrant referral of a person with diabetes to a mental health provider for evaluation and treatment

7.0.0.0 References