1.0.0.0 Patient-Centered Collaborative Care
The American Diabetes Association (ADA) “Standards of Medical Care in Diabetes” includes ADA’s current clinical practice recommendations and is intended to provide the components of diabetes care, general treatment goals and guidelines, and tools to evaluate quality of care. Members of the ADA Professional Practice Committee, a multidisciplinary expert committee, are responsible for updating the Standards of Care annually, or more frequently as warranted. For a detailed description of ADA standards, statements, and reports, as well as the evidence-grading system for ADA’s clinical practice recommendations, please refer to the Standards of Care Introduction. Readers who wish to comment on the Standards of Care are invited to do so at professional.diabetes.org/SOC.
PATIENT-CENTERED COLLABORATIVE CARE
Recommendations
4.1 A patient-centered communication style that uses person-centered and strength-based language and active listening, elicits patient preferences and beliefs, and assesses literacy, numeracy, and potential barriers to care should be used to optimize patient health outcomes and health-related quality of life. B
4.2 Diabetes care should be managed by a multidisciplinary team that may draw from primary care physicians, subspecialty physicians, nurse practitioners, physician assistants, nurses, dietitians, exercise specialists, pharmacists, dentists, podiatrists, and mental health professionals. E
A successful medical evaluation depends on beneficial interactions between the patient and the care team. The Chronic Care Model (1–3) (see Section 1 “Improving Care and Promoting Health in Populations”) is a patient-centered approach to care that requires a close working relationship between the patient and clinicians involved in treatment planning. People with diabetes should receive health care from an interdisciplinary team that may include physicians, nurse practitioners, physician assistants, nurses, dietitians, exercise specialists, pharmacists, dentists, podiatrists, and mental health professionals. Individuals with diabetes must assume an active role in their care. The patient, family or support people, physicians, and health care team should together formulate the management plan, which includes lifestyle management (see Section 5 “Lifestyle Management”).
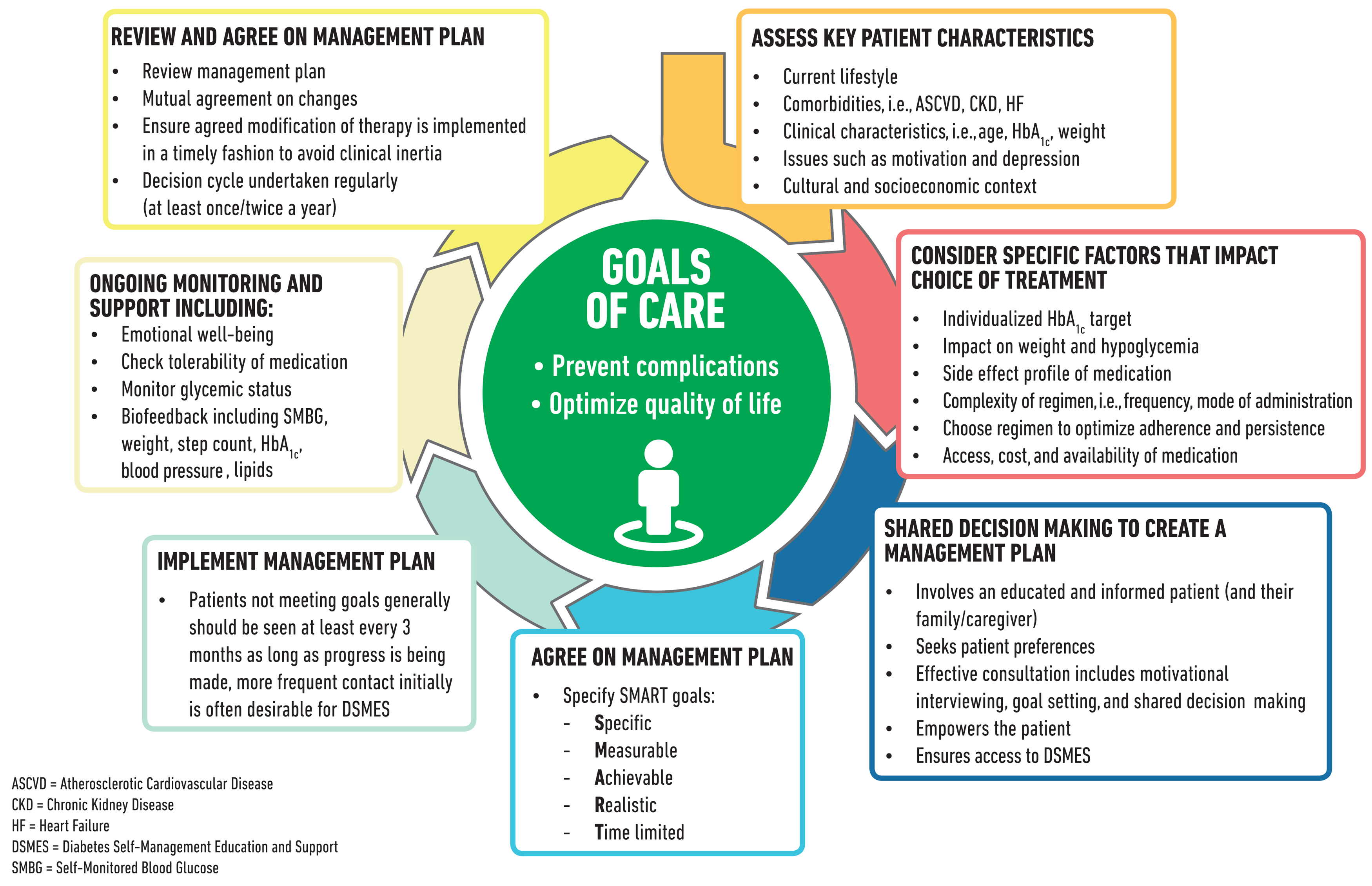
The goals of treatment for diabetes are to prevent or delay complications and maintain quality of life (Fig. 4.1). Treatment goals and plans should be created with the patients based on their individual preferences, values, and goals. The management plan should take into account the patient’s age, cognitive abilities, school/work schedule and conditions, health beliefs, support systems, eating patterns, physical activity, social situation, financial concerns, cultural factors, literacy and numeracy (mathematical literacy), diabetes complications and duration of disease, comorbidities, health priorities, other medical conditions, preferences for care, and life expectancy. Various strategies and techniques should be used to support patients’ self-management efforts, including providing education on problem-solving skills for all aspects of diabetes management.
Provider communications withpatients and families should acknowledge that multiple factors impact glycemic management but also emphasize that collaboratively developed treatment plans and a healthy lifestyle can significantly improve disease outcomes and wellbeing (4–7). Thus, the goal of provider-patient communication is to establish a collaborative relationship and to assess and address self-management barriers without blaming patients for “noncompliance” or “nonadherence” when the outcomes of self-management are not optimal (8). The familiar terms “noncompliance” and “nonadherence” denote a passive, obedient role for a person with diabetes in “following doctor’s orders” that is at odds with the active role people with diabetes take in directing the day-to-day decision making, planning, monitoring, evaluation, and problem-solving involved in diabetes self-management. Using a nonjudgmental approach that normalizes periodic lapses in self-management may help minimize patients’ resistance to reporting problems with self-management. Empathizing and using active listening techniques, such as open-ended questions, reflective statements, and summarizing what the patient said, can help facilitate communication. Patients’ perceptions about their own ability, or selfefficacy, to self-manage diabetes are one important psychosocial factor related to improved diabetes self-management and treatment outcomes in diabetes (9–13) and should be a target of ongoing assessment, patient education, and treatment planning.
Language has a strong impact on perceptions and behavior. The use of empowering language in diabetes care and education can help to inform and motivate people, yet language that shames and judges may undermine this effort. The American Diabetes Association (ADA) and American Association of Diabetes Educators consensus report, “The Use of Language in Diabetes Care and Education,” provides the authors’ expert opinion regarding the use of language by health care professionals when speaking or writing aboutdiabetesforpeople with diabetesor for professional audiences (14). Although further research is needed to address the impact of language on diabetes outcomes, the report includes five key consensus recommendations for language use:
Use language that is neutral, nonjudgmental, and based on facts, actions, or physiology/biology.
Use language that is free from stigma.
Use language that is strength based, respectful, and inclusive and that imparts hope.
Use language that fosters collaboration between patients and providers.
Use language that is person centered (e.g., “person with diabetes” is preferred over “diabetic”).
Figure 4.1—Decision cycle for patient-centered glycemic management in type 2 diabetes. Adapted from Davies et al. (119).

Suggested citation: American Diabetes Association.
4. Comprehensive medical evaluation and
assessment of comorbidities: Standards of
Medical Care in Diabetesd2019. Diabetes Care
2019;42(Suppl. 1):S34–S45
© 2018 by the American Diabetes Association.
Readers may use this article as long as the work
is properly cited, the use is educational and not
for profit, and the work is not altered. More information
is available at http://www.diabetesjournals
.org/content/license.
2.0.0.0 COMPREHENSIVE MEDICAL EVALUATION
Recommendations
4.3 A complete medical evaluation should be performed at the initial visit to:
Confirm the diagnosis and classify diabetes. B
Evaluate for diabetes complications and potential comorbid conditions. B
Review previous treatment and risk factor control in patients with established diabetes. B
Begin patient engagement in the formulation of a care management plan.B
Develop a plan for continuing care. B
4.4 A follow-up visit should include most components of the initial comprehensive medical evaluation including: interval medical history, assessment of medication-taking behavior and intolerance/ side effects, physical examination, laboratory evaluation as appropriate to assess attainment of A1C and metabolic targets, and assessment of risk for complications, diabetes self-management behaviors, nutrition, psychosocial health, and the need for referrals, immunizations, or other routine health maintenance screening. B
4.5 Ongoing management should be guided by the assessment of diabetes complications and shared decision making to set therapeutic goals. B
4.6 The 10-year risk of a first atherosclerotic cardiovascular disease event should be assessed using the race- and sex-specific Pooled Cohort Equations to better stratify atherosclerotic cardiovascular disease risk. B
The comprehensive medical evaluation includes the initial and follow-up evaluations, assessment of complications, psychosocial assessment, management of comorbid conditions, and engagement of the patient throughout the process. While a comprehensive list is provided in Table 4.1, in clinical practice, the provider may need to prioritize the components of the medical evaluation given the available resources and time. The goal is to provide the health care team information to optimally support a patient. In addition to the medical history, physical examination, and laboratory tests, providers should assess diabetes self-management behaviors, nutrition, and psychosocial health (see Section 5 “Lifestyle Management”) and give guidance on routine immunizations. The assessment of sleep pattern and duration should be considered; a recent metaanalysis found that poor sleep quality, short sleep, and long sleep were associated with higher A1C in people with type 2 diabetes (15). Interval follow-up visits should occur at least every 3–6 months, individualized to the patient, and then annually.
Lifestyle management and psychosocial care are the cornerstones of diabetes management. Patients should be referred for diabetes self-management education and support, medical nutrition therapy, and assessment of psychosocial/emotional health concerns if indicated. Patients should receive recommended preventive care services (e.g., immunizations, cancer screening, etc.), smoking cessation counseling, and ophthalmological, dental, and podiatric referrals.
The assessment of risk of acute and chronic diabetes complications and treatment planning are key components of initial and follow-up visits (Table 4.2). The risk of atherosclerotic cardiovascular disease and heart failure (Section 10 “Cardiovascular Disease and Risk Management”), chronic kidney disease staging (Section 11 “Microvascular Complications and Foot Care”), and risk of treatment-associated hypoglycemia (Table 4.3) should be used to individualize targets for glycemia (Section 6 “Glycemic Targets”), blood pressure, and lipids and to select specific glucose-lowering medication (Section 9 “Pharmacologic Approaches to Glycemic Treatment”), antihypertension medication, or statin treatment intensity.
Additional referrals should be arranged as necessary (Table 4.4). Clinicians should ensure that individuals with diabetes are appropriately screened for complications and comorbidities. Discussing and implementing an approach to glycemic control with the patient is a part, not the sole goal, of the patient encounter.
Table 4.1 Components of the comprehensive diabetes medical evaluation at initial, follow-up , and annual visits
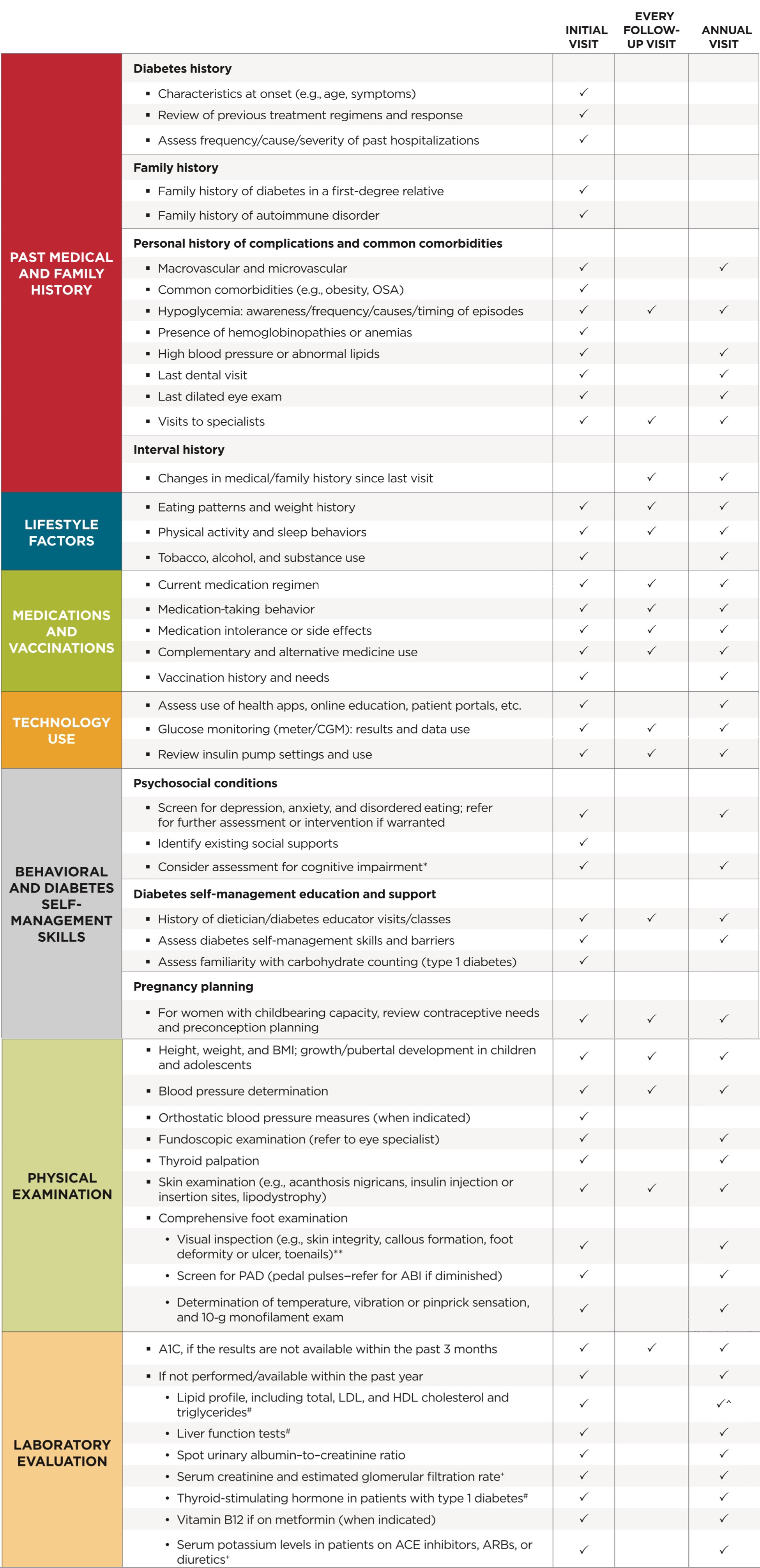
Table 4.2—Assessment and treatment plan*

Table 4.3—Assessment of hypoglycemia risk

Table 4.4—Referrals for initial care management

3.0.0.0 Immunizations
Recommendations
4.7 Provide routinely recommended vaccinations for children and adults with diabetes by age. C
4.8 Annual vaccination against influenza is recommended for all people ≥6 months of age, especially those with diabetes. C
4.9 Vaccination against pneumococcal disease, including pneumococcal pneumonia, with 13-valent pneumococcal conjugate vaccine (PCV13) is recommended for children before age 2 years. People with diabetes ages 2 through 64 years should also receive 23-valent pneumococcal polysaccharide vaccine (PPSV23). At age ≥65 years, regardless of vaccination history, additional PPSV23 vaccination is necessary. C
4.10 Administer a 2- or 3-dose series of hepatitis B vaccine, depending on the vaccine, to unvaccinated adults with diabetes ages 18 through 59 years. C
4.11 Consider administering 3-dose series of hepatitis B vaccine to unvaccinated adults with diabetes ages ≥60 years. C
Children and adults with diabetes should receive vaccinations according to age-appropriate recommendations (16,17). The child and adolescent (≤18 years of age) vaccination schedule is available at www.cdc.gov/vaccines/ schedules/hcp/imz/child-adolescent.html, and the adult (≥19 years of age) vaccination schedule is available at www.cdc.gov/vaccines/schedules/hcp/imz/adult.html. These immunization schedules include vaccination schedules specifically for children, adolescents, and adults with diabetes.
People with diabetes are at higher risk for hepatitis B infection and are more likely to develop complications from influenza and pneumococcal disease. The Centers for Disease Control and Prevention (CDC) Advisory Committee on Immunization Practices (ACIP) recommends influenza, pneumococcal, and hepatitis B vaccinations specifically for people with diabetes. Vaccinations against tetanus-diphtheria-pertussis, measles-mumps-rubella, human papillomavirus, and shingles are also important for adults with diabetes, as they are for the general population.
Influenza
Influenza is a common, preventable infectious disease associated with high mortality and morbidity in vulnerable populations including the young and the elderly and people with chronic diseases. Influenza vaccination in people with diabetes has been found to significantly reduce influenza and diabetes-related hospital admissions (18).
Pneumococcal Pneumonia
Like influenza, pneumococcal pneumonia is a common, preventable disease. People with diabetes are at increased risk for the bacteremic form of pneumococcal infection and have been reported to have a high risk of nosocomial bacteremia, with a mortality rate as high as 50% (19). The ADA endorses recommendations from the CDC ACIP that adults age ≥65 years, who are at higher risk for pneumococcal disease, receive an additional 23-valent pneumococcal polysaccharide vaccine (PPSV23), regardless of prior pneumococcal vaccination history. See detailed recommendations at www.cdc.gov/vaccines/hcp/acip-recs/vacc-specific/pneumo.html.
Hepatitis B
Compared with the general population, people with type 1 or type 2 diabetes have higher rates of hepatitis B. This may be due to contact with infected blood or through improper equipment use (glucose monitoring devices or infected needles). Because of the higher likelihood of transmission, hepatitis B vaccine is recommended for adults with diabetes age <60 years. For adults age ≥60 years, hepatitis B vaccine may be administered at the discretion of the treating clinician based on the patient’s likelihood of acquiring hepatitis B infection.
4.0.0.0 ASSESSMENT OF COMORBIDITIES
4.1.0.0 Introduction
Besides assessing diabetes-related complications, clinicians and their patients need to be aware of common comorbidities that affect people with diabetes and may complicate management (20–24). Diabetes comorbidities are conditions that affect people with diabetes more often than age-matched people without diabetes. This section includes many of the common comorbidities observed in patients with diabetes but is not necessarily inclusive of all the conditions that have been reported.
4.2.0.0 Autoimmune Diseases
Recommendation
4.12 Consider screening patients with type 1 diabetes for autoimmune thyroid disease and celiac disease soon after diagnosis. B
People with type 1 diabetes are at increased risk for other autoimmune diseases including thyroid disease, primary adrenal insufficiency, celiac disease, autoimmune gastritis, autoimmune hepatitis, dermatomyositis, and myasthenia gravis (25–27). Type 1 diabetes may also occur with other autoimmune diseases in the context of specific genetic disorders or polyglandular autoimmune syndromes (28). In autoimmune diseases, the immune system fails to maintain self-tolerance to specific peptides within target organs. It is likely that many factors trigger autoimmune disease; however, common triggering factors are known for only some autoimmune conditions (i.e., gliadin peptides in celiac disease) (see Section 13 “Children and Adolescents”).
4.3.0.0 Cancer
Diabetes is associated with increased risk of cancers of the liver, pancreas, endometrium, colon/rectum, breast, and bladder (29). The association may result from shared risk factors between type 2 diabetes and cancer (older age, obesity, and physical inactivity) but may also be due to diabetes-related factors (30), such as underlying disease physiology or diabetes treatments, although evidence for these links is scarce. Patients with diabetes should be encouraged to undergo recommended age- and sex- appropriate cancer screenings and to reduce their modifiable cancer risk factors (obesity, physical inactivity, and smoking). New onset of atypical diabetes (lean body habitus, negative family history) in a middle-aged or older patient may precede the diagnosis of pancreatic adenocarcinoma (31). However, in the absence of other symptoms (e.g., weight loss, abdominal pain), routine screening of all such patients is not currently recommended.
4.4.0.0 Cognitive Impairment/Dementia
Recommendation
4.13 In people with a history of cognitive impairment/dementia, intensive glucose control cannot be expected to remediate deficits. Treatment should be tailored to avoid significant hypoglycemia. B
Diabetes is associated with a significantly increased risk and rate of cognitive decline and an increased risk of dementia (32,33). A recent meta-analysis of prospective observational studies in people with diabetes showed 73% increased risk of all types of dementia, 56% increased risk of Alzheimer dementia, and 127% increased risk of vascular dementia compared with individuals without diabetes (34). The reverse is also true: people with Alzheimer dementia are more likely to develop diabetes than people without Alzheimer dementia. In a 15-year prospective study of community-dwelling people >60 years of age, the presence of diabetes at baseline significantly increased the age- and sex-adjusted incidence of all-cause dementia, Alzheimer dementia, and vascular dementia compared with rates in those with normal glucose tolerance (35).
Hyperglycemia
In those with type 2 diabetes, the degree and duration of hyperglycemia are related todementia. More rapid cognitive decline is associated with both increased A1C and longer duration ofdiabetes (34). The Action to Control Cardiovascular Risk in Diabetes (ACCORD) study found that each 1% higher A1C level was associated with lower cognitive function in individuals with type 2 diabetes (36). However, the ACCORD study found no difference in cognitive outcomes in participants randomly assigned to intensive and standard glycemic control, supporting the recommendation that intensive glucose control should not be advised for the improvement of cognitive function in individuals with type 2 diabetes (37).
Hypoglycemia
In type 2 diabetes, severe hypoglycemia is associated with reduced cognitive function, and those with poor cognitive function have more severe hypoglycemia. In a long-term study of older patients with type 2 diabetes, individuals with one or more recorded episode of severe hypoglycemia had a stepwise increase in risk of dementia (38). Likewise, the ACCORD trial found that as cognitive function decreased, the risk of severe hypoglycemia increased (39). Tailoring glycemic therapy may help to prevent hypoglycemia in individuals with cognitive dysfunction.
Nutrition
In one study, adherence to the Mediterranean diet correlated with improved cognitive function (40). However, a recent Cochrane review found insufficient evidence to recommend any dietary change for the prevention or treatment of cognitive dysfunction (41).
Statins
A systematic review has reported that data do not support an adverse effect of statins on cognition (42). The U.S. Food and Drug Administration postmarketing surveillance databases have also revealed a low reporting rate for cognitive-related adverse events, including cognitive dysfunction or dementia, with statin therapy, similar to rates seen with other commonly prescribed cardiovascular medications (42). Therefore, fear of cognitive decline should not be a barrier to statin use in individuals with diabetes and a high risk for cardiovascular disease.
4.5.0.0 Nonalcoholic Fatty Liver Disease
Recommendation
4.14 Patients with type 2 diabetes or prediabetes and elevated liver enzymes (alanine aminotransferase) or fatty liver on ultrasound should be evaluated for presence of nonalcoholic steatohepatitis and liver fibrosis. C
Diabetes is associated with the development of nonalcoholic fatty liver disease, including its more severe manifestations of nonalcoholic steatohepatitis, liver fibrosis, cirrhosis, and hepatocellular carcinoma (43). Elevations of hepatic transaminase concentrations are associated with higher BMI, waist circumference, and triglyceride levels and lower HDL cholesterol levels. Noninvasive tests, such as elastography or fibrosis biomarkers, may be used to assess risk of fibrosis, but referral to a liver specialist and liver biopsy may be required for definitive diagnosis (43a). Interventions that improve metabolic abnormalities in patients with diabetes (weight loss, glycemic control, and treatment with specific drugs for hyperglycemia or dyslipidemia) are also beneficial for fatty liver disease (44,45). Pioglitazone and vitamin E treatment of biopsy-proven nonalcoholic steatohepatitis have been shown to improve liver histology, but effects on longerterm clinical outcomes are not known (46,47). Treatment with liraglutide and with sodium–glucose cotransporter 2 inhibitors (dapagliflozin and empagliflozin) has also shown some promise in preliminary studies, although benefits may be mediated, at least in part, by weight loss (48–50).
4.6.0.0 Pancreatitis
Recommendation
4.15 Islet autotransplantation should be considered for patients requiring total pancreatectomy for medically refractory chronic pancreatitis to prevent postsurgical diabetes. C
Diabetes is linked to diseases of the exocrine pancreas such as pancreatitis, which may disrupt the global architecture or physiology of the pancreas, often resulting in both exocrine and endocrine dysfunction. Up to half of patients with diabetes may have impaired exocrine pancreas function (51). People with diabetes are at an approximately twofold higher risk of developing acute pancreatitis (52).
Conversely, prediabetes and/or diabetes has been found to develop in approximately one-third of patients after an episode of acute pancreatitis (53), thus the relationship is likely bidirectional. Postpancreatitis diabetes may include either new-onset disease or previously unrecognized diabetes (54). Studies of patients treated with incretin-based therapies for diabetes have also reported that pancreatitis may occur more frequently with these medications, but results have been mixed (55,56).
Islet autotransplantation should be considered for patients requiring total pancreatectomy for medically refractory chronic pancreatitis to prevent postsurgical diabetes. Approximately one-third of patients undergoing total pancreatectomy with islet autotransplantation are insulin free 1 year postoperatively, and observational studies from different centers have demonstrated islet graft function up to a decade after the surgery in some patients (57–61). Both patient and disease factors should be carefully considered when deciding the indications and timing of this surgery. Surgeries should be performed in skilled facilities that have demonstrated expertise in islet autotransplantation.
4.7.0.0 Fractures
Age-specific hip fracture risk is significantly increased in people with both type 1 (relative risk 6.3) and type 2 (relative risk 1.7) diabetes in both sexes (62). Type 1 diabetes is associated with osteoporosis, but in type 2 diabetes, an increased risk of hip fracture is seen despite higher bone mineral density (BMD) (63). In three large observational studies of older adults, femoral neck BMD T score and the World Health Organization Fracture Risk Assessment Tool (FRAX) score were associated with hip and nonspine fractures. Fracture risk was higher in participants with diabetes compared with those without diabetes for a given T score and age or for a given FRAX score (64). Providers should assess fracture history and risk factors in older patients with diabetes and recommend measurement of BMD if appropriate for the patient’s age and sex. Fracture prevention strategies for people with diabetes are the same as for the general population and include vitamin D supplementation. For patients with type 2 diabetes with fracture risk factors, thiazolidinediones (65) and sodium– glucose cotransporter 2 inhibitors (66) should be used with caution.
4.8.0.0 Hearing Impairment
Hearing impairment, both in high frequency and low/midfrequency ranges, is more common in people with diabetes than in those without, perhaps due to neuropathy and/or vascular disease. In a National Health and Nutrition Examination Survey (NHANES) analysis, hearing impairment was about twice as prevalent in people with diabetes compared with those without, after adjusting for age and other risk factors for hearing impairment (67).
4.9.0.0 HIV
Recommendation
4.16 Patients with HIV should be screened for diabetes and pre-diabetes with a fasting glucose test before starting antiretroviral therapy, at the time ofswitching antiretroviral therapy, and 3–6 months after starting or switching antiretroviral therapy. If initial screening results are normal, checking fasting glucose every year is advised. E
Diabetes risk is increased with certain protease inhibitors (PIs) and nucleoside reverse transcriptase inhibitors (NRTIs). New-onset diabetes is estimated to occur in more than 5% of patients infected with HIV on PIs, whereas more than 15% may have prediabetes (68). PIs are associated with insulin resistance and may also lead to apoptosis of pancreatic β-cells. NRTIs also affect fat distribution (both lipohypertrophy and lipoatrophy), which is associated with insulin resistance.
Individuals with HIV are at higher risk for developing prediabetes and diabetes on antiretroviral (ARV) therapies, so a screening protocol is recommended (69). The A1C test may underestimate glycemia in people with HIV and is not recommended for diagnosis and may present challenges for monitoring (70). In those with prediabetes, weight loss through healthy nutrition and physical activity may reduce the progression toward diabetes. Among patients with HIV and diabetes, preventive health care using an approach similar to that used in patients without HIV is critical to reduce the risks of microvascular and macrovascular complications.
For patients with HIV and ARV-associated hyperglycemia, it may be appropriate to consider discontinuing the problematic ARV agents if safe and effective alternatives are available (71). Before making ARV substitutions, carefully consider the possible effect on HIV virological control and the potential adverse effects of new ARV agents. In some cases, antihyperglycemia agents may still be necessary.
4.10.0.0 Low Testosterone_ in Men
Recommendation
4.17 In men with diabetes who have symptoms or signs of hypogonadism, such as decreased sexual desire (libido) or activity, or erectile dysfunction, consider screening with a morning serum testosterone level. B
Mean levels of testosterone are lower in men with diabetes compared with age-matched men without diabetes, but obesity is a major confounder (72,73). Treatment in asymptomatic men is controversial. Testosterone_ replacement in men with symptomatic hypogonadism may have benefits including improved sexual function, well-being, muscle mass and strength, and bone density (74). In men with diabetes who have symptoms or signs of low testosterone (hypogonadism), a morning total testosterone should be measured using an accurate and reliable assay. Free or bioavailable testosterone levels should also be measured in men with diabetes who have total testosterone levels close to the lower limit, given expected decreases in sex hormone– binding globulin with diabetes. Further testing (such as luteinizing hormone and follicle-stimulating hormone levels) may be needed to distinguish between primary and secondary hypogonadism.
4.11.0.0 Obstructive Sleep Apnea
Age-adjusted rates of obstructive sleep apnea, a risk factor for cardiovascular disease, are significantly higher (4- to 10-fold) with obesity, especially with central obesity (75). The prevalence of obstructive sleep apnea in the population with type 2 diabetes may be as high as 23%, and the prevalence of any sleepdisordered breathing may be as high as 58% (76,77). In obese participants enrolled in the Action for Health in Diabetes (Look AHEAD) trial, it exceeded 80% (78). Patients with symptoms suggestive of obstructive sleep apnea (e.g., excessive daytime sleepiness, snoring, witnessed apnea) should be considered for screening (79). Sleep apnea treatment (lifestyle modification, continuous positive airway pressure, oral appliances, and surgery) significantly improves quality of life and blood pressure control. The evidence for a treatment effect on glycemic control is mixed (80).
4.12.0.0 Periodontal Disease
Periodontal disease is more severe, and may be more prevalent, in patients with diabetes than in those without (81,82). Current evidence suggests that periodontal disease adversely affects diabetes outcomes, although evidence for treatment benefits remains controversial (24).
4.13.0.0 Psychosocial/Emotional Disorders
4.13.1.0 Introduction
Prevalence of clinically significant psychopathology diagnoses are considerably more common in people with diabetes than in those without the disease (83). Symptoms, both clinical and subclinical, that interfere with the person’s ability to carry out daily diabetes self-management tasks must be addressed. Providers should consider an assessment of symptoms of depression, anxiety, and disordered eating and of cognitive capacities using patient-appropriate standardized/validated tools at the initial visit, at periodic intervals, and when there is a change in disease, treatment, or life circumstance. Including caregivers and family members in this assessment is recommended. Diabetes distress is addressed in Section 5 “Lifestyle Management,” as this state is very common and distinct from the psychological disorders discussed below (84).
4.13.2.0 Anxiety Disorders
Recommendations
4.18 Consider screening for anxiety in people exhibiting anxiety or worries regarding diabetes complications, insulin injections or infusion, taking medications, and/or hypoglycemia that interfere with self-management behaviors and those who express fear, dread, or irrational thoughts and/or show anxiety symptoms such as avoidance behaviors, excessive repetitive behaviors, or social withdrawal. Refer for treatment if anxiety is present. B
4.19 People with hypoglycemia unawareness, which can co-occur with fear of hypoglycemia, should be treated using blood glucose awareness training (or other evidence-based intervention) to help reestablish awareness of hypoglycemia and reduce fear of hypoglycemia. A
Anxiety symptoms and diagnosable disorders (e.g., generalized anxiety disorder, body dysmorphic disorder, obsessive-compulsive disorder, spe-cific phobias, and posttraumatic stress disorder) are common in people with diabetes (85).
The Behavioral Risk Factor Surveil-lance System (BRFSS) estimated the life-time prevalence of generalized anxiety disorder to be 19.5% in people with either type 1 or type 2 diabetes (86). Common diabetes-specific concerns in-clude fears related to hypoglycemia (87, 88), not meeting blood glucose targets (85), and insulin injections or infusion (89). Onset of complications presents another critical point when anxiety can occur (90). People with diabetes who exhibit excessive diabetes self-management behaviors well beyond what is prescribed or needed to achieve glycemic targets may be experiencing symptoms of obsessive-compulsive disorder (91).
General anxiety is a predictor of injection-related anxiety and associated with fear of hypoglycemia (88,92). Fear of hypoglycemia and hypoglycemia unawareness often co-occur, and interventions aimed at treating one often benefit both (93). Fear of hypoglycemia may explain avoidance of behaviors associated with lowering glucose such as increasing insulin doses or frequency of monitoring. If fear of hypoglycemia is identified and a person does not have symptoms of hypoglycemia, a structured program of blood glucose awareness training delivered in routine clinical practice can improve A1C, reduce the rate of severe hypoglycemia, and restore hypoglycemia awareness (94,95).
4.13.3.0 Depression
Recommendations
4.20 Providers should consider annual screening of all patients with diabetes, especially those with a self-reported history of depression, for depressivesymptoms with age-appropriate depression screening measures, recognizing that further evaluation will be necessary for individuals who have a positive screen. B
4.21 Beginning at diagnosis of complications or when there are significant changes in medical status, consider assessment for depression. B
4.22 Referrals for treatment of depression should be made to mental health providers with experience using cognitive behavioral therapy, interpersonal therapy, or other evidence-based treatment approaches in conjunction with collaborative care with the patient’s diabetes treatment team. A
History of depression, current depression, and antidepressant medication use are risk factors for the development of type 2 diabetes, especially if the individual has other risk factors such as obesity and family history of type 2 diabetes (96–98). Elevated depressive symptoms and depressive disorders affect one in four patients with type 1 or type 2 diabetes (99). Thus, routine screening for depressive symptoms is indicated in this high-risk population including people with type 1 or type 2 diabetes, gestational diabetes mellitus, and postpartum diabetes. Regardless of diabetes type, women have significantly higher rates of depression than men (100).
Routine monitoring with patient-appropriate validated measures can help to identify if referral is warranted. Adult patients with a history of depressive symptoms or disorder need ongoing monitoring of depression recurrence within the context of routine care (96). Integrating mental and physical health care can improve outcomes. When a patient is in psychological therapy (talk therapy), the mental health provider should be incorporated into the diabetes treatment team (101).
4.13.4.0 Disordered Eating Behavior
Recommendations
4.23 Providers should consider reevaluating the treatment regimen of people with diabetes who present with symptoms of disordered eating behavior, an eating disorder, or disrupted patterns of eating. B
4.24 Consider screening for disordered or disrupted eating using validated screening measures when hyperglycemia and weight loss are unexplained based on self-reported behaviors related to medication dosing, meal plan, and physical activity. In addition, a review of the medical regimen is recommended to identify potential treatment-related effects on hunger/ caloric intake. B
Estimated prevalence of disordered eating behaviors and diagnosable eating disorders in people with diabetes varies (102–104). For people with type 1 diabetes, insulin omission causing glycosuria in order to lose weight is the most commonly reported disordered eating behavior (105,106); in people with type 2 diabetes, bingeing (excessive food intake with an accompanying sense of loss of control) is most commonly reported. For people with type 2 diabetes treated with insulin, intentional omission is also frequently reported (107). People with diabetes and diagnosable eating disorders have high rates of comorbid psychiatric disorders (108). People with type 1 diabetes and eating disorders have high rates of diabetes distress and fear of hypoglycemia (109). When evaluating symptoms of disordered or disrupted eating in people with diabetes, etiology and motivation for the behavior should be considered (104,110). Adjunctive medication such as glucagon-like peptide 1 receptor agonists (111) may help individuals not only to meet glycemic targets but also to regulate hunger and food intake, thus having the potential to reduce uncontrollable hunger and bulimic symptoms.
4.13.5.0 Serious Mental Illness
Recommendations
Annually screen people who are prescribed atypical antipsychotic medications for prediabetes or diabetes. B
If a second-generation antipsychotic medication is prescribed for adolescents or adults with diabetes, changes in weight, glycemic control, and cholesterol levels should be carefully monitored and the treatment regimen should be reassessed. C
Incorporate monitoring of diabetes self-care activities into treatment goals in people with diabetes and serious mental illness. B
Studies of individuals with serious mental illness, particularly schizophrenia and other thought disorders, show significantly increased rates of type 2 diabetes (112). People with schizophrenia should bemonitored for type 2 diabetes because of the known comorbidity. Disordered thinking and judgment can be expected to make it difficult to engage in behaviors that reduce risk factors for type 2 diabetes, such as restrained eating for weight management. Coordinated management of diabetes or prediabetes and serious mental illness is recommended to achieve diabetes treatment targets. In addition, those taking second-generation (atypical) antipsychotics, such as olanzapine, require greater monitoring because of an increase in risk of type 2 diabetes associated with this medication (113).
5.0.0.0 References