xxxxx
2.2.0.0 Fasting and 2-Hour Plasma Glucose
The FPG and 2-h PG may be used to diagnose diabetes (Table 2.2). The concordance between the FPG and 2-h PG tests is imperfect, as is the concordance between A1C and either glucose-based test. Compared with FPG and A1C cut points, the 2-h PG value diagnoses more people with prediabetes and diabetes (9).
xxxxx
2.3.0.0 A1C
Recommendations
2.1 To avoid misdiagnosis or missed diagnosis, the A1C test should be performed using a method that is certified by the NGSP and standardized to the Diabetes Control and Complications Trial (DCCT) assay. B
2.2 Marked discordance between measured A1C and plasma glucose levels should raise the possibility of A1C assay interference due to hemoglobin variants (i.e., hemoglobinopathies) and consideration of using an assay without interference or plasma blood glucose criteria to diagnose diabetes. B
2.3 In conditions associated with an altered relationship between A1C and glycemia, such as sickle cell disease, pregnancy (second and third trimesters and the postpartum period), glucose-6-phosphate dehydrogenase deficiency, HIV, hemodialysis, recent blood loss or transfusion, or erythropoietin therapy, only plasma blood glucose criteria should be used to diagnose diabetes. B
The A1C test should be performed using a method that is certified by the NGSP (www.ngsp.org) and standardized or traceable to the Diabetes Control and Complications Trial (DCCT) reference assay. Although point-of-care A1C assays may be NGSP certified or U.S. Food and Drug Administration approved for diagnosis, proficiency testing is not always mandated for performing the test. Therefore, point-of-care assays approved for diagnostic purposes should only be considered in settings licensed to perform moderate-to-high complexity tests. As discussed in Section 6 “Glycemic Targets,” point-of-care A1C assays may be more generally applied for glucose monitoring.
The A1C has several advantages compared with the FPG and OGTT, including greater convenience (fasting not required), greater preanalytical stability, and less day-to-day perturbations during stress and illness. However, these advantages may be offset by the lower sensitivity of A1C at the designated cut point, greater cost, limited availability of A1C testing in certain regions of the developing world, and the imperfect correlation between A1C and average glucose in certain individuals. The A1C test, with a diagnostic threshold of ≥6.5% (48 mmol/mol), diagnoses only 30% of the diabetes cases identified collectively using A1C, FPG, or 2-h PG, according to National Health and Nutrition Examination Survey (NHANES) data (10). When using A1C to diagnose diabetes, it is important to recognize that A1C is an indirect measure of average blood glucose levels and to take other factors into consideration that may impact hemoglobin glycation independently of glycemia including HIV treatment (11,12), age, race/ ethnicity, pregnancy status, genetic background, and anemia/hemoglobinopathies.
Table 2.2 - Criteria for the diagnosis of diabetes

Age
The epidemiological studies that formed the basis for recommending A1C to diagnose diabetes included only adult populations (10). However, a recent ADA clinical guidance concluded that A1C, FPG, or 2-h PG can be used to test for prediabetes or type 2 diabetes in children and adolescents. (see p. S20 SCREENING AND TESTING FOR PREDIABETES AND TYPE 2 DIABETES IN CHILDREN AND ADOLESCENTS for additional information) (13).
Race/Ethnicity/Hemoglobinopathies
Hemoglobin variants can interfere with the measurement of A1C, although most assays in use in the U.S. are unaffected by the most common variants. Marked discrepancies between measured A1C and plasma glucose levels should prompt consideration that the A1C assay may not be reliable for that individual. For patients with a hemoglobin variant but normal red blood cell turnover, such as those with the sickle cell trait, an A1C assay without interference from hemoglobin variants should be used. An updated list of A1C assays with interferences is available at www.ngsp.org/interf.asp.
African Americans heterozygous for the common hemoglobin variant HbS may have, for any given level of mean glycemia, lower A1C by about 0.3% than those without the trait (14). Another genetic variant, X-linked glucose-6-phosphate dehydrogenase G202A, carried by 11% of African Americans, was associated with a decrease in A1C of about 0.8% in homozygous men and 0.7% in homozygous women compared with those without the variant (15).
Even in the absence of hemoglobin variants, A1C levels may vary with race/ ethnicity independently of glycemia (16-18). For example, African Americans may have higher A1C levels than non-Hispanic whites with similar fasting and postglucose load glucose levels (19), and A1C levels may be higher for a given mean glucose concentration when measured with continuous glucose monitoring (20). Though conflicting data exists, African Americans may also have higher levels of fructosamine and glycated albumin and lower levels of 1,5-anhydroglucitol, suggesting that their glycemic burden (particularly postprandially) may be higher (21,22). The association of A1C with risk for complications appears to be similar in African Americans and non-Hispanic whites (23,24).
Other Conditions Altering the Relationship of A1C and Glycemia
In conditions associated with increased red blood cell turnover, such as sickle cell disease, pregnancy (second and third trimesters), glucose-6-phosphate dehydrogenase deficiency (25,26), hemodialysis, recent blood loss or transfusion, or erythropoietin therapy, only plasma blood glucose criteria should be used to diagnose diabetes (27). A1C is less reliable than blood glucose measurement in other conditions such as postpartum (28-30), HIV treated with certain drugs (11), and irondeficient anemia (31).
xxxxx
2.4.0.0 Confirming the Diagnosis
Unless there is a clear clinical diagnosis (e.g., patient in a hyperglycemic crisis or with classic symptoms of hyperglycemia and a random plasma glucose ≥200 mg/dL [11.1 mmol/L]), diagnosis requires two abnormal test results from the same sample (32) or in two separate test samples. If using two separate test samples, it is recommended that the second test, which may either be a repeat of the initial test or a different test, be performed without delay. For example, if the A1C is 7.0% (53 mmol/mol) and a repeat result is 6.8% (51 mmol/mol), the diagnosis of diabetes is confirmed. If two different tests (such as A1C and FPG) are both above the diagnostic threshold when analyzed from the same sample or in two different test samples, this also confirms the diagnosis. On the other hand, if a patient has discordant results from two different tests, then the test result that is above the diagnostic cut point should be repeated, with consideration of the possibility of A1C assay interference. The diagnosis is made on the basis of the confirmed test. For example, if a patient meets the diabetes criterion of the A1C (two results ≥6.5% [48 mmol/mol]) but not FPG (,126 mg/dL [7.0 mmol/L]), that person should nevertheless be considered to have diabetes. Since all the tests have preanalytic and analytic variability, it is possible that an abnormal result (i.e., above the diagnostic threshold), when repeated, will produce a value below the diagnostic cut point. This scenario is likely for FPG and 2-h PG if the glucose samples remain at room temperatureand are notcentrifuged promptly. Because of the potential for preanalytic variability, it is critical that samples for plasma glucose be spun and separated immediately after they are drawn. If patients have test results nearthemarginsof the diagnostic threshold, the health care professional should follow the patient closely and repeat the test in 3–6 months.
xxxxx
3.0.0.0 TYPE 1 DIABETES
3.1.0.0 Recommendations
Recommendations
2.4 Plasma blood glucose rather than A1C should be used to diagnose the acute onset of type 1 diabetes in individuals with symptoms of hyperglycemia. E
2.5 Screening for type 1 diabetes risk with a panel of autoantibodies is currently recommended only in the setting of a research trial or in first-degree family members of a proband with type 1 diabetes. B
2.6 Persistence of two or more autoantibodies predicts clinical diabetes and may serve as an indication for intervention in the setting of a clinical trial. B
xxxxx
3.2.0.0 Diagnosis
In a patient with classic symptoms, measurement of plasma glucose is sufficient to diagnose diabetes (symptoms of hyperglycemia or hyperglycemic crisis plus a random plasma glucose ≥200 mg/dL [11.1 mmol/L]). In these cases, knowing the plasma glucose level is critical because, in addition to confirming that symptoms are due to diabetes, it will inform management decisions. Some providers may also want to know the A1C to determine how long a patient has had hyperglycemia. The criteria to diagnose diabetes are listed in Table 2.2.
xxxxx
3.3.0.0 Immune-Mediated Diabetes
This form, previously called “insulin-dependent diabetes” or “juvenile-onset diabetes,”accounts for 5–10% of diabetes and is due to cellular-mediated autoimmune destruction of the pancreatic β-cells. Autoimmune markers include islet cell autoantibodies and autoantibodies to GAD (GAD65), insulin, the tyrosine phosphatases IA-2 and IA-2b, and ZnT8. Type 1 diabetes is defined by the presence of one or more of these autoimmune markers. The disease has strong HLA associations, with linkage to the DQA and DQB genes. These HLA-DR/DQ alleles can be either predisposing or protective.
The rate of β-cell destruction is quite variable, being rapid in some individuals (mainly infants and children) and slow in others (mainly adults). Children and adolescents may present with DKA as the first manifestation of the disease. Others have modest fasting hyperglycemia that can rapidly change to severe hyperglycemia and/or DKA with infection or other stress. Adults may retain sufficient β-cell function to prevent DKA for many years; such individuals eventually become dependent on insulin for survival and are at risk for DKA. At this latter stage of the disease, there is little or no insulin secretion, as manifested by low or undetectable levels of plasma C-peptide. Immune-mediated diabetes commonly occurs in childhood and adolescence, but it can occur at any age, even in the 8th and 9th decades of life.
Autoimmune destruction of β-cells has multiple genetic predispositions and is also related to environmental factors that are still poorly defined. Although patients are not typically obese when they present with type 1 diabetes, obesity should not preclude the diagnosis. People with type 1 diabetes are also prone to other autoimmune disorders such as Hashimoto thyroiditis, Graves disease, Addison disease, celiac disease, vitiligo, autoimmune hepatitis, myasthenia gravis, and pernicious anemia (see Section 4 “Comprehensive Medical Evaluation and Assessment of Comorbidities”).
xxxxx
3.4.0.0 Idiopathic Type 1 Diabetes
Some forms of type 1 diabetes have no known etiologies. These patients have permanent insulinopenia and are prone to DKA, but have no evidence of β-cell autoimmunity. Although only a minority of patients with type 1 diabetes fall into this category, of those who do, most are of African or Asian ancestry. Individuals with this form of diabetes suffer from episodic DKA and exhibit varying degrees of insulin deficiency between episodes. This form of diabetes is strongly inherited and is not HLA associated. An absolute requirement for insulin replacement therapy in affected patients may be intermittent.
xxxxx
3.5.0.0 Screening for Type 1 Diabetes Risk
The incidence and prevalence of type 1 diabetes is increasing (33). Patients with type 1 diabetes often present with acute symptoms of diabetes and markedly elevated blood glucose levels, and approximately one-third are diagnosed with life-threatening DKA (2). Several studies indicate that measuring islet autoantibodies in relatives of those with type 1 diabetes may identify individuals who are at risk for developing type 1 diabetes (5). Such testing, coupled with education about diabetes symptoms and close follow-up, may enable earlier identification of type 1 diabetes onset. A study reported the risk of progression to type 1 diabetes from the time of seroconversion to autoantibody positivity in three pediatric cohorts from Finland, Germany, and the U.S. Of the 585 children who developed more than two autoantibodies, nearly 70% developed type 1 diabetes within 10 years and 84% within 15 years (34). These findings are highly significant because while the German group was recruited from offspring of parents with type 1 diabetes, the Finnish and American groups were recruited from the general population. Remarkably, the findings in all three groups were the same, suggesting that the same sequence of events led to clinical disease in both “sporadic” and familial cases of type 1 diabetes. Indeed, the risk of type 1 diabetes increases as the number of relevant autoantibodies detected increases (35-37).
Although there is currently a lack of accepted screening programs, one should consider referring relatives of those with type 1 diabetes for antibody testing for risk assessment in the setting of a clinical research study (www.diabetestrialnet.org). Widespread clinical testing of asymptomatic low-risk individuals is not currently recommended due to lack of approved therapeutic interventions. Individuals who test positive should be counseled about the risk of developing diabetes, diabetes symptoms, and DKA prevention. Numerous clinical studies are being conducted to test various methods of preventing type 1 diabetes in those with evidence of autoimmunity (www.clinicaltrials.gov).
xxxxx
4.0.0.0 PREDIABETES AND TYPE 2 DIABETES
4.1.0.0 Recommendations
Recommendations
2.7 Screening for prediabetes and type 2 diabetes with an informal assessment of risk factors or validated tools should be considered in asymptomatic adults. B
2.8 Testing for prediabetes and/or type 2 diabetes in asymptomatic people should be considered in adults of any age who are overweight or obese(BMI ≥25 kg/m2 or ≥23 kg/m2 in Asian Americans) and who have one or more additional risk factors for diabetes (Table 2.3). B
2.9 For all people, testing should begin at age 45 years. B
2.10 If tests are normal, repeat testing carried out at a minimum of 3-year intervals is reasonable. C
2.11 To test for prediabetes and type 2 diabetes, fasting plasma glucose, 2-h plasma glucose during 75-g oral glucose tolerance test, and A1C are equally appropriate. B
2.12 In patients with prediabetes and type 2 diabetes, identify and, if appropriate, treat other cardiovascular disease risk factors. B
2.13 Risk-based screening for prediabetes and/or type 2 diabetes should be considered after the onset ofpuberty or after 10 years of age, whichever occurs earlier, in children and adolescents who are overweight(BMI ≥85th percentile) or obese (BMI ≥95th percentile) and who have additional risk factors for diabetes. (See Table 2.4 for evidence grading of risk factors.)
xxxxx
4.2.0.0 Prediabetes
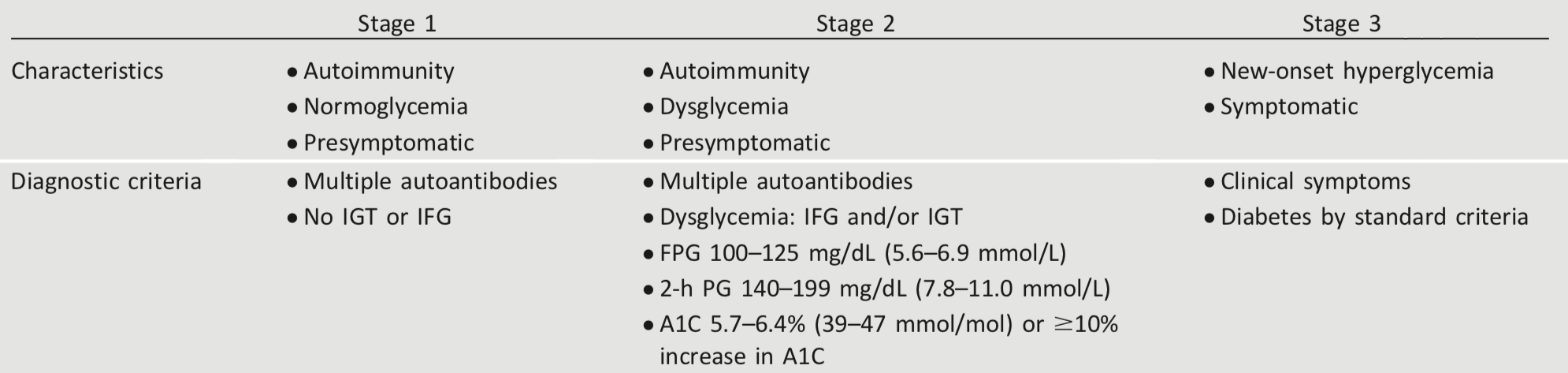
“Prediabetes” is the term used for individuals whose glucose levels do not meet the criteria for diabetes but are too high to be considered normal (23,24). Patients with prediabetes are defined by the presence of IFG and/or IGT and/or A1C 5.7–6.4% (39–47 mmol/mol) (Table 2.5). Prediabetes should not be viewed as a clinical entity in its own right but rather as an increased risk for diabetes and cardiovascular disease (CVD). Criteria for testing for diabetes or prediabetes in asymptomatic adults is outlined in Table 2.3. Prediabetes is associated with obesity (especially abdominal or visceral obesity), dyslipidemia with high triglycerides and/or low HDL cholesterol, and hypertension.
Diagnosis
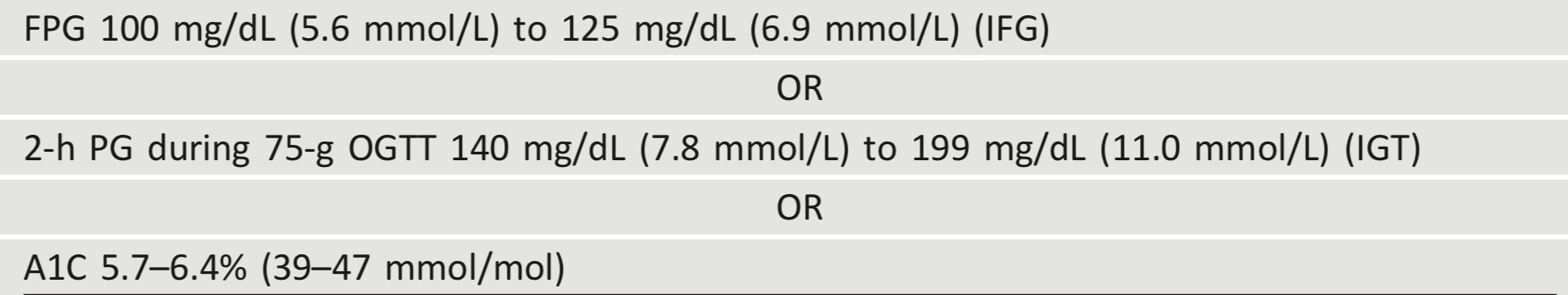
IFG is defined as FPG levels between 100 and 125 mg/dL (between 5.6 and 6.9 mmol/L) (38,39) and IGT as 2-h PG during 75-g OGTT levels between 140 and 199 mg/dL (between 7.8 and 11.0 mmol/L) (40). It should be noted that the World Health Organization (WHO) and numerous other diabetes organizations define the IFG cutoff at 110 mg/dL (6.1 mmol/L).
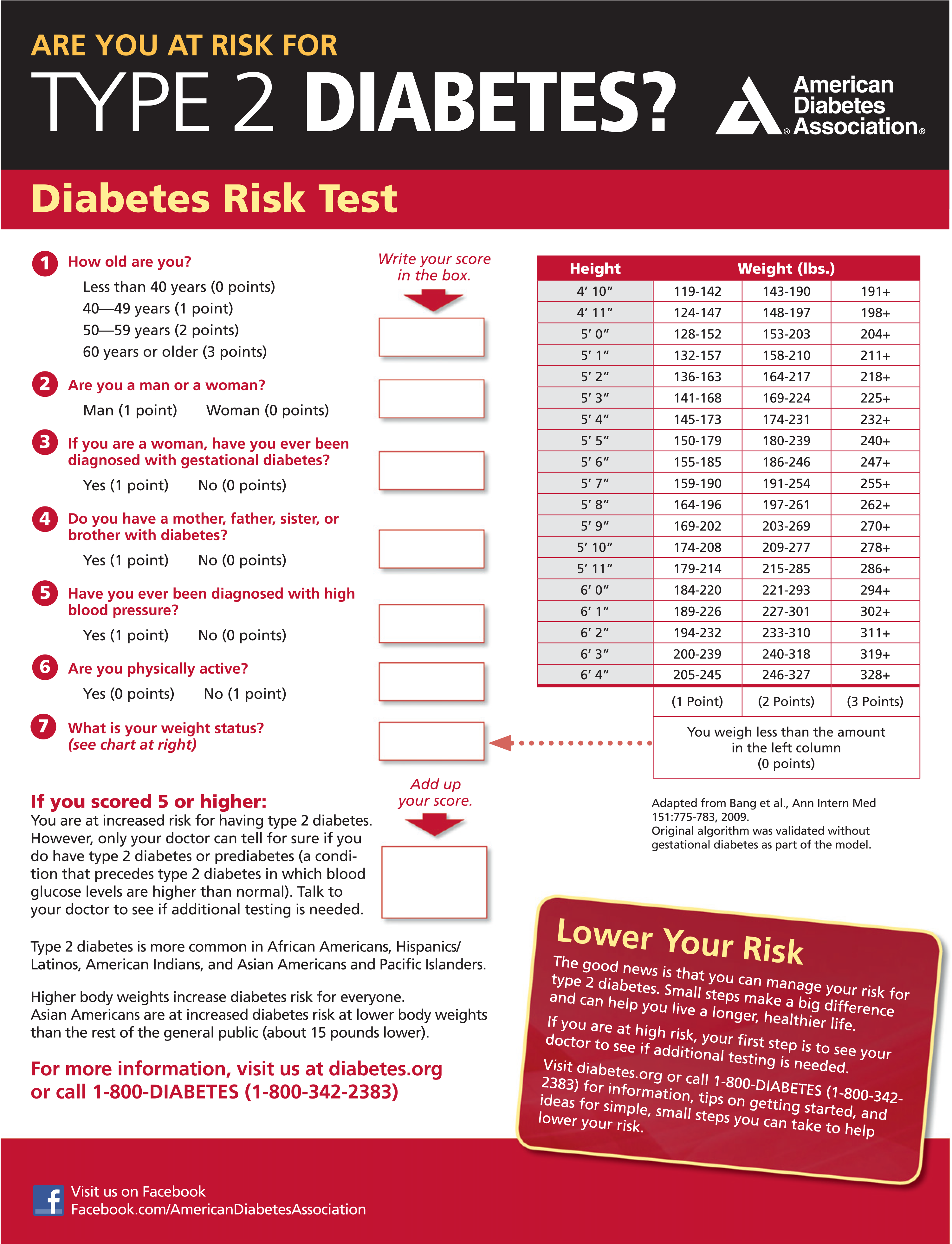
As with the glucose measures, several prospective studies that used A1C to predict the progression to diabetes as defined by A1C criteria demonstrated a strong, continuous association between A1C and subsequent diabetes. In a systematic review of 44,203 individuals from 16 cohort studies with a follow-up interval averaging 5.6 years (range 2.8– 12 years), those with A1C between 5.5 and 6.0% (between 37 and 42 mmol/mol) had a substantially increased risk of diabetes (5-year incidence from 9 to 25%). Those with an A1C range of 6.0–6.5% (42–48 mmol/mol) had a 5-year risk of developing diabetes between 25 and 50% and a relative risk 20 times higher compared with A1C of 5.0% (31 mmol/mol) (41). In a community-based study of African American and non-Hispanic white adults without diabetes, baseline A1C was a stronger predictor of subsequent diabetes and cardiovascular events than fasting glucose (42). Other analyses suggest that A1C of 5.7% (39 mmol/mol) or higher is associated with a diabetes risk similar to that of the high-risk participants in the Diabetes Prevention Program (DPP) (43), and A1C at baseline was a strong predictor of the development of glucose-defined diabetes during the DPP and its follow-up (44). Hence, it is reasonable to consider an A1C range of 5.7–6.4% (39–47 mmol/mol) as identifying individuals with prediabetes. Similar to those with IFG and/or IGT, individuals with A1C of 5.7– 6.4% (39–47 mmol/mol) should be informed of their increased risk for diabetes and CVD and counseled about effective strategies to lower their risks (see Section 3 “Prevention or Delay of Type 2 Diabetes”). Similar to glucose measurements, the continuum of risk is curvilinear, so as A1C rises, the diabetes risk rises disproportionately (41). Aggressive interventions and vigilant follow-up should be pursued for those considered at very high risk (e.g., those with A1C >6.0% [42 mmol/mol]). Table 2.5 summarizes the categories of prediabetes and Table 2.3 the criteria for prediabetes testing. The ADA diabetes risk test is an additional option for assessment to determine the appropriateness of testing for diabetes or prediabetes in asymptomatic adults. (Fig. 2.1) (diabetes.org/socrisktest). For additional background regarding risk factors and screening for prediabetes, see pp. S18–S20 (SCREENING AND TESTING FOR PREDIABETES AND TYPE 2 DIABETES IN ASYMPTOMATIC ADULTS and SCREENING AND TESTING FOR PREDIABETES AND TYPE 2 DIABETES IN CHILDREN AND ADOLESCENTS).
Table 2.3 - Criteria for testing for diabetes or prediabetes in asymptomatic adults

Table 2.4 - Risk-based screening for type 2 diabetes or prediabetes in asymptomatic children and adolescents in a clinical setting

Table 2.5 - Criteria defining prediabetes*
xxxxx
4.3.0.0 Type 2 Diabetes
Type 2 diabetes, previously referred to as “noninsulin-dependent diabetes” or “adult-onset diabetes,” accounts for 90– 95% of all diabetes. This form encompasses individuals who have relative (rather than absolute) insulin deficiency and have peripheral insulin resistance. At least initially, and often throughout their lifetime, these individuals may not need insulin treatment to survive.
There are various causes of type 2 diabetes. Although the specific etiologies are not known, autoimmune destruction of β-cells does not occur and patients do not have any of the other known causes of diabetes. Most but not all patients with type 2 diabetes are overweight or obese. Excess weight itself causes some degree of insulin resistance. Patients who are not obese or overweight by traditional weight criteria may have an increased percentage of body fat distributed predominantly in the abdominal region.
DKA seldom occurs spontaneously in type 2 diabetes; when seen, it usually arises in association with the stress of another illness such as infection or with the use of certain drugs (e.g., corticosteroids, atypical antipsychotics, and sodium–glucose cotransporter 2 inhibitors) (45,46). Type 2 diabetes frequently goes undiagnosed for many years because hyperglycemia develops gradually and, at earlier stages, is often not severe enough for the patient to notice the classic diabetes symptoms. Nevertheless, even undiagnosed patients are at increased risk of developing macrovascular and microvascular complications.
Whereas patients with type 2 diabetes may have insulin levels that appear normal or elevated, the higher blood glucose levels in these patients would be expected to result in even higher insulin values had their β-cell function been normal. Thus, insulin secretion is defective in these patients and insufficient to compensate for insulin resistance.
Insulin resistance may improve with weight reduction and/or pharmacologic treatment of hyperglycemia but is seldom restored to normal.
The risk of developing type 2 diabetes increases with age, obesity, and lack of physical activity. It occurs more frequently in women with prior GDM, in those with hypertension or dyslipidemia, and in certain racial/ethnic subgroups (African American, American Indian, Hispanic/Latino, and Asian American). It is often associated with a strong genetic predisposition or family history in first-degree relatives, more so than type 1 diabetes. However, the genetics of type 2 diabetes is poorly understood. In adults without traditional risk factors for type 2 diabetes and/or younger age, consider antibody testing to exclude the diagnosis of type 1 diabetes (i.e., GAD).
xxxxx
4.4.0.0 Screening and Testing
4.4.1.0 Screening and Testing for Prediabetes and Type 2 Diabetes in Asymptomatic Adults
Screening for prediabetes and type 2 diabetes risk through an informal assessment of risk factors (Table 2.3) or with an assessment tool, such as the ADA risk test (Fig. 2.1) (diabetes.org/socrisktest), is recommended to guide providers on whether performing a diagnostic test (Table 2.2) is appropriate. Prediabetes and type 2 diabetes meet criteria for conditions in which early detection is appropriate. Both conditions are common and impose significant clinical and public health burdens. There is often a long presymptomatic phase before the diagnosis of type 2 diabetes. Simple tests to detect preclinical disease are readily available. The duration of glycemic burden is a strong predictor of adverse outcomes. There are effective interventions that prevent progression from prediabetes to diabetes (see Section 3 “Prevention or Delay of Type 2 Diabetes”) and reduce the risk of diabetes complications (see Section 10 “Cardiovascular Disease and Risk Management” and Section 11 “Microvascular Complications and Foot Care”).
Approximately one-quarter of people with diabetes in the U.S. and nearly half of Asian and Hispanic Americans with diabetes are undiagnosed (38,39). Although screening of asymptomatic individuals to identify those with prediabetes or diabetes might seem reasonable, rigorous clinical trials to prove the effectiveness of such screening have not been conducted and are unlikely to occur.
A large European randomized controlled trial compared the impact of screening for diabetes and intensive multifactorial intervention with that of screening and routine care (47). General practice patients between the ages of 40 and 69 years were screened for diabetes and randomly assigned by practice to intensive treatment of multiple risk factors or routine diabetes care. After 5.3 years of follow-up, CVD risk factors were modestly but significantly improved with intensive treatment compared with routine care, but the incidence of first CVD events or mortality was not significantly different between the groups (40). The excellent care provided to patients in the routine care group and the lack of an unscreened control arm limited the authors’ ability to determine whether screening and early treatment improved outcomes compared with no screening and later treatment after clinical diagnoses. Computer simulation modeling studies suggest that major benefits are likely to accrue from the early diagnosis and treatment of hyperglycemia and cardiovascular risk factors in type 2 diabetes (48); moreover, screening, beginning at age 30 or 45 years and independent of risk factors, may be cost-effective (<$11,000 per quality-adjusted life-year gained) (49). Additional considerations regarding testing for type 2 diabetes and prediabetes in asymptomatic patients include the following.
Figure 2.1 - ADA risk test (diabetes.org/socrisktest).
Age
Age is a major risk factor for diabetes. Testing should begin at no later than age 45 years for all patients. Screening should be considered in overweight or obese adults of any age with one or more risk factors for diabetes.
BMI and Ethnicity
In general, BMI ≥25 kg/m2 is a risk factor for diabetes. However, data suggest that the BMI cut point should be lower for the Asian American population (50,51). The BMI cut points fall consistently between 23 and 24 kg/m2 (sensitivity of 80%) for nearly all Asian American subgroups (with levels slightly lower for Japanese Americans). This makes a rounded cut point of 23 kg/m2 practical. An argument can be made to push the BMI cut point to lower than 23 kg/m2 in favor of increased sensitivity; however, this would lead to an unacceptably low specificity (13.1%). Data from the WHO also suggest that a BMI of ≥23 kg/m2 should be used to define increased risk in Asian Americans (52). The finding that one-third to onehalf of diabetes in Asian Americans is undiagnosed suggests that testing is not occurring at lower BMI thresholds (53,54).
Evidence also suggests that other populations may benefit from lower BMI cut points. For example, in a large multiethnic cohort study, for an equivalent incidence rate of diabetes, a BMI of 30 kg/m2 in non-Hispanic whites was equivalent to a BMI of 26 kg/m2 in African Americans (55).
Medications
Certain medications, such as glucocorticoids, thiazide diuretics, some HIV medications, and atypical antipsychotics (56), are known to increase the risk of diabetes and should be considered when deciding whether to screen.
Testing Interval
The appropriate interval between screening tests is not known (57). The rationale for the 3-year interval is that with this interval, the number of false-positive tests that require confirmatory testing will be reduced and individuals with false-negative tests will be retested before substantial time elapses and complications develop (57).
Community Screening
Ideally, testing should be carried out within a health care setting because of the need for follow-up and treatment. Community screening outside a health care setting is generally not recommended because people with positive tests may not seek, or have access to, appropriate follow-up testing and care. However, in specific situations where an adequate referral system is established beforehand for positive tests, community screening may be considered. Community testing may also be poorly targeted; i.e., it may fail to reach the groups most at risk and inappropriately test those at very low risk or even those who have already been diagnosed (58).
Screening in Dental Practices
Because periodontal disease is associated with diabetes, the utility of screening in a dental setting and referral to primary care as a means to improve the diagnosis of prediabetes and diabetes has been explored (59-61), with one study estimating that 30% of patients ≥30 years of age seen in general dental practices had dysglycemia (61). Further research is needed to demonstrate the feasibility, effectiveness, and costeffectiveness of screening in this setting.
xxxxx
4.4.2.0 Screening and Testing for Prediabetes and Type 2 Diabetes in Children and Adolescents
In the last decade, the incidence and prevalence of type 2 diabetes in adolescents has increased dramatically, especially in racial and ethnic minority populations (33). See Table 2.4 for recommendations on risk-based screening for type 2 diabetes or prediabetes in asymptomatic children and adolescents in a clinical setting (13). See Tables 2.2 and 2.5 for the criteria for the diagnosis of diabetes and prediabetes, respectively, which apply to children, adolescents, and adults. See Section 13 “Children and Adolescents” for additional information on type 2 diabetes in children and adolescents.
Some studies question the validity of A1C in the pediatric population, especially among certain ethnicities, and suggest OGTT or FPG as more suitable diagnostic tests (62). However, many of these studies do not recognize that diabetes diagnostic criteria are based on long-term health outcomes, and validations are not currently available in the pediatric population (63). The ADA acknowledges the limited data supporting A1C for diagnosing type 2 diabetes in children and adolescents. Although A1C is not recommended for diagnosis of diabetes in children with cystic fibrosis or symptoms suggestive of acute onset of type 1 diabetes and only A1C assays without interference are appropriate for children with hemoglobinopathies, the ADA continues to recommend A1C for diagnosis of type 2 diabetes in this cohort (64,65).
xxxxx
5.0.0.0 GESTATIONAL DIABETES MELLITUS
5.1.0.0 Recommendations
Recommendations
2.14 Test for undiagnosed diabetes at the first prenatal visit in those with risk factors using standard diagnostic criteria. B
2.15 Test for gestational diabetes mellitus at 24–28 weeks of gestation in pregnant women not previously known to have diabetes. A
2.16 Test women with gestational diabetes mellitus for prediabetes or diabetes at 4–12 weeks postpartum, using the 75-g oral glucose tolerance test and clinically appropriate nonpregnancy diagnostic criteria. B
2.17 Women with a history of gestational diabetes mellitus should have lifelong screening for the development of diabetes or prediabetes at least every 3 years. B
2.18 Women with a history of gestational diabetes mellitus found to have prediabetes should receive intensive lifestyle interventions or metformin to prevent diabetes. A
xxxxx
5.2.0.0 Definition
For many years, GDM was defined as any degree of glucose intolerance that was first recognized during pregnancy (40), regardless of whether the condition may have predated the pregnancy or persisted after the pregnancy. This definition facilitated a uniform strategy for detection and classification of GDM, but it was limited by imprecision.
The ongoing epidemic of obesity and diabetes has led to more type 2 diabetes in women of childbearing age, with an increase in the number of pregnant women with undiagnosed type 2 diabetes (66). Because of the number of pregnant women with undiagnosed type 2 diabetes, it is reasonable to test women with risk factors for type 2 diabetes (67) (Table 2.3) at their initial prenatal visit, using standard diagnostic criteria (Table 2.2). Women diagnosed with diabetes by standard diagnostic criteria in the first trimester should be classified as having preexisting pregestational diabetes (type 2 diabetes or, very rarely, type 1 diabetes or monogenic diabetes). Women found to have prediabetes in the first trimester may be encouraged to make lifestyle changes to reduce their risk of developing type 2 diabetes, and perhaps GDM, though more study is needed (68). GDM is diabetes that is first diagnosed in the second or third trimester of pregnancy that is not clearly either preexisting type 1 or type 2 diabetes (see Section 14 “Management of Diabetes in Pregnancy”). The International Association of the Diabetes and Pregnancy Study Groups (IADPSG) GDM diagnostic criteria for the 75-g OGTT as well as the GDM screening and diagnostic criteria used in the two-step approach were not derived from data in the first half of pregnancy, so the diagnosis of GDM in early pregnancy by either FPG or OGTT values is not evidence based (69).
Because GDM confers increased risk for the development of type 2 diabetes after delivery (70,71) and because effective prevention interventions are available (72,73), women diagnosed with GDM should receive lifelong screening for prediabetes and type 2 diabetes.
xxxxx
5.3.0.0 Diagnosis
5.3.1.0 Overview
GDM carries risks for the mother, fetus, and neonate. Not all adverse outcomes are of equal clinical importance. The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) study (74), a large-scale multinational cohort study completed by more than 23,000 pregnant women, demonstrated that risk of adverse maternal, fetal, and neonatal outcomes continuously increased as a function of maternal glycemia at 24–28 weeks of gestation, even within ranges previously considered normal for pregnancy. For most complications, there was no threshold for risk. These results have led to careful reconsideration of the diagnostic criteria for GDM. GDM diagnosis (Table 2.6) can be accomplished with either of two strategies:
- “One-step” 75-g OGTT or
- “Two-step” approach with a 50-g (nonfasting) screen followed by a 100-g OGTT for those who screen positive
Different diagnostic criteria will identify different degrees of maternal hyperglycemia and maternal/fetal risk, leading some experts to debate, and disagree on, optimal strategies for the diagnosis of GDM.
Table 2.6 - Screening for and diagnosis of GDM

xxxxx
5.3.2.0 One-Step Strategy
The IADPSG defined diagnostic cut points for GDM as the average fasting, 1-h, and 2-h PG values during a 75-g OGTT in women at 24–28 weeks of gestation who participated in the HAPO study at which odds for adverse outcomes reached 1.75 times the estimated odds of these outcomes at the mean fasting, 1-h, and 2-h PG levels of the study population. This one-step strategy was anticipated to significantly increase the incidence of GDM (from 5–6% to 15–20%), primarily because only one abnormal value, not two, became sufficient to make the diagnosis (75). The anticipated increase in the incidence of GDM could have a substantial impact on costs and medical infrastructure needs and has the potential to “medicalize” pregnancies previously categorized as normal. A recent follow-up study of women participating in a blinded study of pregnancy OGTTs found that 11 years after their pregnancies, women who would have been diagnosed with GDM by the one-step approach, as compared with those without, were at 3.4-fold higher risk of developing prediabetes and type 2 diabetes and had children with a higher risk of obesity and increased body fat, suggesting that the larger group of women identified by the one-step approach would benefit from increased screening for diabetes and prediabetes that would accompany a history of GDM (76). Nevertheless, the ADA recommends these diagnostic criteria with the intent of optimizing gestational outcomes because these criteria were the only ones based on pregnancy outcomes rather than end points such as prediction of subsequent maternal diabetes.
The expected benefits to the offspring are inferred from intervention trials that focused on women with lower levels of hyperglycemia than identified using older GDM diagnostic criteria. Those trials found modest benefits including reduced rates of large-for-gestational-age births and preeclampsia (77,78). It is important to note that 80–90% of women being treated for mild GDM in these two randomized controlled trials could be managed with lifestyle therapy alone. The OGTT glucose cutoffs in these two trials overlapped with the thresholds recommended by the IADPSG, and in one trial (78), the 2-h PG threshold (140 mg/dL [7.8 mmol/L]) was lower than the cutoff recommended by the IADPSG (153 mg/dL [8.5 mmol/L]). No randomized controlled trials of identifying and treating GDM using the IADPSG criteria versus older criteria have been published to date. Data are also lacking on how the treatment of lower levels of hyperglycemia affects a mother’s future risk for the development of type 2 diabetes and her offspring’s risk for obesity, diabetes, and other metabolic disorders. Additional well-designed clinical studies are needed to determine the optimal intensity of monitoring and treatment of women with GDM diagnosed by the one-step strategy (79,80).
xxxxx
5.3.3.0 Two-Step Strategy
In 2013, the National Institutes of Health (NIH) convened a consensus development conference to consider diagnostic criteria for diagnosing GDM (81). The 15-member panel had representatives from obstetrics/gynecology, maternal-fetal medicine, pediatrics, diabetes research, biostatistics, and other related fields. The panel recommended a two-step approach to screening that used a 1-h 50-g glucose load test (GLT) followed by a 3-h 100-g OGTT for those who screened positive. The American College of Obstetricians and Gynecologists (ACOG) recommends any of the commonly used thresholds of 130, 135, or 140 mg/dL for the 1-h 50-g GLT (82). A systematic review for the U.S. Preventive Services Task Force compared GLT cut-offs of 130 mg/dL (7.2 mmol/L) and 140 mg/dL (7.8 mmol/L) (83). The higher cutoff yielded sensitivity of 70–88% and specificity of 69–89%, while the lower cutoff was 88–99% sensitive and 66– 77% specific. Data regarding a cutoff of 135 mg/dL are limited. As for other screening tests, choice of a cutoff is based upon the trade-off between sensitivity and specificity. The use of A1C at 24–28 weeks of gestation as a screening test for GDM does not function as well as the GLT (84).
Key factors cited by the NIH panel in their decision-making process were the lack of clinical trial data demonstrating the benefits of the one-step strategy and the potential negative consequences of identifying a large group of women with GDM, including medicalization of pregnancy with increased health care utilization and costs. Moreover, screening with a 50-g GLT does not require fasting and is therefore easier to accomplish for many women. Treatment of higher-threshold maternal hyperglycemia, as identified by the two-step approach, reduces rates of neonatal macrosomia, large-for-gestational-age births (85), and shoulder dystocia, without increasing small-for-gestational-age births. ACOG currently supports the two-step approach but notes that one elevated value, as opposed to two, may be used for the diagnosis of GDM (82). If this approach is implemented, the incidence of GDM by the two-step strategy will likely increase markedly. ACOG recommends either of two sets of diagnostic thresholds for the 3-h 100-g OGTT (86,87). Each is based on different mathematical conversions of the original recommended thresholds, which used whole blood and nonenzymatic methods for glucose determination. A secondary analysis of data from a randomized clinical trial of identification and treatment of mild GDM (88) demonstrated that treatment was similarly beneficial in patients meeting only the lower thresholds (86) and in those meeting only the higher thresholds (87). If the two-step approach is used, it would appear advantageous to use the lower diagnostic thresholds as shown in step 2 in Table 2.6.
xxxxx
5.3.4.0 Future Considerations
The conflicting recommendations from expert groups underscore the fact that there are data to support each strategy. A cost-benefit estimation comparing the two strategies concluded that the one-step approach is cost-effective only if patients with GDM receive postdelivery counseling and care to prevent type 2 diabetes (89). The decision of which strategy to implement must therefore be made based on the relative values placed on factors that have yet to be measured (e.g., willingness to change practice based on correlation studies rather than intervention trial results, available infrastructure, and importance of cost considerations).
As the IADPSG criteria (“one-step strategy”) have been adopted internationally, further evidence has emerged to support improved pregnancy outcomes with cost savings (90) and may be the preferred approach. Data comparing population-wide outcomes with one-step versus two-step approaches have been inconsistent to date (91,92). In addition, pregnancies complicated by GDM per the IADPSG criteria, but not recognized as such, have comparable outcomes to pregnancies diagnosed as GDM by the more stringent two-step criteria (93,94). There remains strong consensus that establishing a uniform approach to diagnosing GDM will benefit patients, caregivers, and policy makers. Longer-term outcome studies are currently underway.
xxxxx
6.0.0.0 CYSTIC FIBROSIS–RELATED DIABETES
Recommendations
2.19 Annual screening for cystic fibrosis–related diabetes with an oral glucose tolerance test should begin by age 10 years in all patients with cystic fibrosis not previously diagnosed with cystic fibrosis–related diabetes. B
2.20 A1C is not recommended as a screening test for cystic fibrosis– related diabetes. B
2.21 Patients with cystic fibrosis– related diabetes should be treated with insulin to attain individualized glycemic goals. A
2.22 Beginning 5 years after the diagnosis of cystic fibrosis–related diabetes, annual monitoring for complications of diabetes is recommended. E
Cystic fibrosis–related diabetes (CFRD) is the most common comorbidity in people with cystic fibrosis, occurring in about 20% of adolescents and 40–50% of adults (95). Diabetes in this population, compared with individuals with type 1 or type 2 diabetes, is associated with worse nutritional status, more severe inflammatory lung disease, and greater mortality. Insulin insufficiency is the primary defect in CFRD. Genetically determined β-cell function and insulin resistance associated with infection and inflammation may also contribute to the development of CFRD. Milder abnormalities of glucose tolerance are even more common and occur at earlier ages than CFRD. Whether individuals with IGT should be treated with insulin replacement has not currently been determined. Although screening for diabetes before the age of 10 years can identify risk for progression to CFRD in those with abnormal glucose tolerance, no benefit has been established with respect to weight, height, BMI, or lung function. Continuous glucose monitoring or HOMA of β-cell function (96) may be more sensitive than OGTT to detect risk for progression to CFRD; however, evidence linking these results to long-term outcomes is lacking, and these tests are not recommended for screening (97).
CFRD mortality has significantly decreased over time, and the gap in mortality between cystic fibrosis patients with and without diabetes has considerably narrowed (98). There are limited clinical trial dataon therapy for CFRD. The largest study compared three regimens: premeal insulin aspart, repaglinide, or oral placebo in cystic fibrosis patients with diabetes or abnormal glucose tolerance. Participants all had weight loss in the year preceding treatment; however, in the insulin-treated group, this pattern was reversed, and patients gained 0.39 (± 0.21) BMI units (P = 0.02). The repaglinide-treated group had initial weight gain, but this was not sustained by 6 months. The placebo group continued to lose weight (99). Insulin remains the most widely used therapy for CFRD (100).
Additional resources for the clinical management of CFRD can be found in the position statement “Clinical Care Guidelines for Cystic Fibrosis2Related Diabetes: A Position Statement of the American Diabetes Association and a Clinical Practice Guideline of the Cystic Fibrosis Foundation, Endorsed by the Pediatric Endocrine Society” (101) and in the International Society for Pediatric and Adolescent Diabetes’s 2014 clinical practice consensus guidelines (102).
xxxxx
7.0.0.0 POSTTRANSPLANTATION DIABETES MELLITUS
Recommendations
2.23 Patients should be screened after organ transplantation for hyperglycemia, with a formal diagnosis of posttransplantation diabetes mellitus being best made once a patient is stable on an immunosuppressive regimen and in the absence of an acute infection. E
2.24 The oral glucose tolerance test is the preferred test to make a diagnosis of posttransplantation diabetes mellitus. B
2.25 Immunosuppressive regimens shown to provide the best outcomes for patient and graft survival should be used, irrespective of posttransplantation diabetes mellitus risk. E
Several terms are used in the literature to describe the presence of diabetes following organ transplantation. “New-onset diabetes after transplantation” (NODAT) is one such designation that describes individuals who develop new-onset diabetes following transplant. NODAT excludes patients with pretransplant diabetes that was undiagnosed as well as posttransplant hyperglycemia that resolves by the time of discharge (103). Another term, “posttransplantation diabetes mellitus” (PTDM) (103,104), describes the presence of diabetes in the posttransplant setting irrespective of the timing of diabetes onset.
Hyperglycemia is very common during the early posttransplant period, with ˜90% of kidney allograft recipients exhibiting hyperglycemia in the first few weeks following transplant (103-106). In most cases, such stress- or steroidinduced hyperglycemia resolves by the time of discharge (106,107). Although the use of immunosuppressive therapies is a major contributor to the development of PTDM, the risks of transplant rejection outweigh the risks of PTDM and the role of the diabetes care provider is to treat hyperglycemia appropriately regardless of the type of immunosuppression (103). Risk factors for PTDM include both general diabetes risks (such as age, family history of diabetes, etc.) as well as transplant-specific factors, such as use of immunosuppressant agents (108). Whereas posttransplantation hyperglycemia is an important risk factor for subsequent PTDM, a formal diagnosis of PTDM is optimally made once the patient is stable on maintenance immunosuppression and in the absence of acute infection (106-108). The OGTT is considered the gold standard test for the diagnosis of PTDM (103,104,109, 110). However, screening patients using fasting glucose and/or A1C can identify high-risk patients requiring further assessment and may reduce the number of overall OGTTs required.
Few randomized controlled studies have reported on the short- and long-term use of antihyperglycemic agents in the setting of PTDM (108,111,112). Most studies have reported that transplant patients with hyperglycemia and PTDM after transplantation have higher rates of rejection, infection, and rehospitalization (106,108,113).
Insulin therapy is the agent of choice for the management of hyperglycemia and diabetes in the hospital setting. After discharge, patients with preexisting diabetes could go back on their pretransplant regimen if they were in good control before transplantation. Those with previously poor control or with persistent hyperglycemia should continue insulin with frequent home self-monitoring of blood glucose to determine when insulin dose reductions may be needed and when it may be appropriate to switch to noninsulin agents.
No studies to date have established which noninsulin agents are safest or most efficacious in PTDM. The choice of agent is usually made based on the side effect profile of the medication and possible interactions with the patient’s immunosuppression regimen (108). Drug dose adjustments may be required because of decreases in the glomerular filtration rate, a relatively common complication in transplant patients. A small short-term pilot study reported that metformin was safe to use in renal transplant recipients (114), but its safety has not been determined in other types of organ transplant. Thiazolidinediones have been used successfully in patients with liver and kidney transplants, but side effects include fluid retention, heart failure, and osteopenia (115,116). Dipeptidyl peptidase 4 inhibitors do not interact with immunosuppressant drugs and have demonstrated safety in small clinical trials (117,118). Well-designed intervention trials examining the efficacy and safety of these and other antihyperglycemic agents in patients with PTDM are needed.
xxxxx
8.0.0.0 MONOGENIC DIABETES SYNDROMES
8.1.0.0 Recommendations
Recommendations
2.26 All children diagnosed with diabetes in the first 6 months of life should have immediate genetic testing for neonatal diabetes. A
2.27 Children and adults, diagnosed in early adulthood, who have diabetes not characteristic of type 1 or type 2 diabetes that occurs in successive generations (suggestive of an autosomal dominant pattern of inheritance) should have genetic testing for maturity-onset diabetes of the young. A
2.28 In both instances, consultation with a center specializing in diabetes genetics is recommended to understand the significance of these mutations and how best to approach further evaluation, treatment, and genetic counseling. E
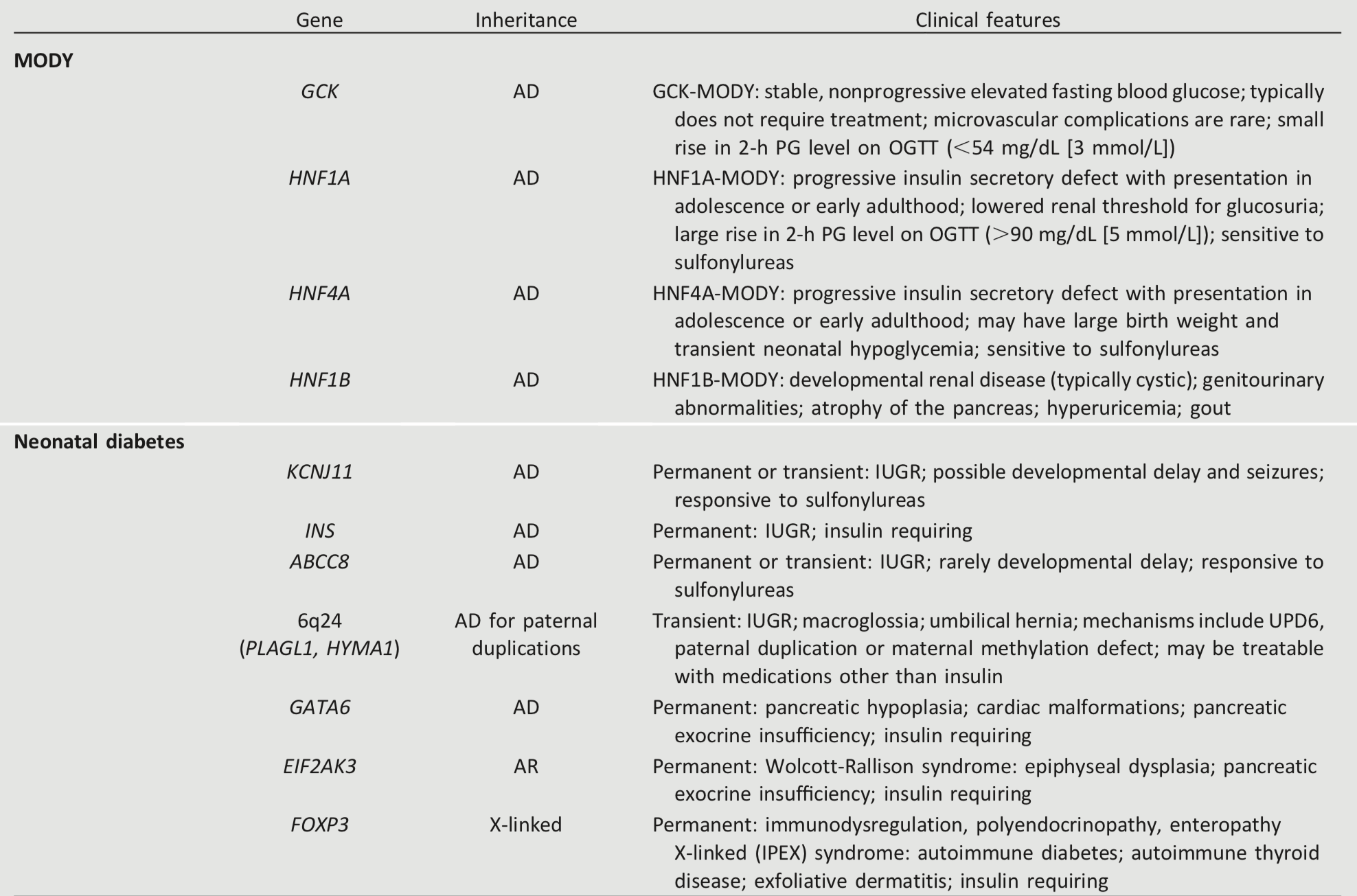
Monogenic defects that cause β-cell dysfunction, such as neonatal diabetes and MODY, represent a small fraction of patients with diabetes (<5%). Table 2.7 describes the most common causes of monogenic diabetes. For a comprehensive list of causes, see Genetic Diagnosis of Endocrine Disorders (119).
Table 2.7 - Most common causes of monogenic diabetes (119)
xxxxx
8.2.0.0 Neonatal Diabetes
Diabetes occurring under 6 months of age is termed “neonatal” or “congenital” diabetes, and about 80–85% of cases can be found to have an underlying monogenic cause (120). Neonatal diabetes occurs much less often after 6 months of age, whereas autoimmune type 1 diabetes rarely occurs before 6 months of age. Neonatal diabetes can either be transient or permanent. Transient diabetes is most often due to overexpression of genes on chromosome 6q24, is recurrent in about half of cases, and may be treatable with medications other than insulin. Permanent neonatal diabetes is most commonly due to autosomal dominant mutations in the genes encoding the Kir6.2 subunit (KCNJ11) and SUR1 subunit (ABCC8) of the β-cell KATP channel. Correct diagnosis has critical implications because most patients with KATP-related neonatal diabetes will exhibit improved glycemic control when treated with high-dose oral sulfonylureas instead of insulin. Insulin gene (INS) mutations are the second most common cause of permanent neonatal diabetes, and, while intensive insulin management is currently the preferred treatment strategy, there are important genetic considerations, as most of the mutations that cause diabetes are dominantly inherited.
xxxxx
8.3.0.0 Maturity-Onset Diabetes of the Young
MODY is frequently characterized by onset of hyperglycemia at an early age (classically before age 25 years, although diagnosis may occur at older ages). MODY is characterized by impaired insulin secretion with minimal or no defects in insulin action (in the absence of coexistent obesity). It is inherited in an autosomal dominant pattern with abnormalities in at least 13 genes on different chromosomes identified to date. The most commonly reported forms are GCK-MODY (MODY2), HNF1A-MODY (MODY3), and HNF4A-MODY (MODY1).
Clinically, patients with GCK-MODY exhibit mild, stable, fasting hyperglycemia and do not require antihyperglycemic therapy except sometimes during pregnancy. Patients with HNF1A- or HNF4A-MODY usually respond well to low doses of sulfonylureas, which are considered first-line therapy. Mutations or deletions in HNF1B are associated with renal cysts and uterine malformations (renal cysts and diabetes [RCAD] syndrome). Other extremely rare forms of MODY have been reported to involve other transcription factor genes including PDX1 (IPF1) and NEUROD1.
xxxxx
8.4.0.0 Diagnosis of Monogenic Diabetes
A diagnosis of one of the three most common forms of MODY, including GCK-MODY, HNF1A-MODY, and HNF4A-MODY, allows for more cost-effective therapy (no therapy for GCK-MODY; sulfonylureas as first-line therapy for HNF1A-MODY and HNF4A-MODY). Additionally, diagnosis can lead to identification of other affected family members.
A diagnosis of MODY should be considered in individuals who have atypical diabetes and multiple family members with diabetes not characteristic of type 1 or type 2 diabetes, although admittedly “atypical diabetes” is becoming increasingly difficult to precisely define in the absence of a definitive set of tests for either type of diabetes. In most cases, the presence of autoantibodies for type 1 diabetes precludes further testing for monogenic diabetes, but the presence of autoantibodies in patients with monogenic diabetes has been reported (121). Individuals in whom monogenic diabetes is suspected should be referred to a specialist for further evaluation if available, and consultation is available from several centers. Readily available commercial genetic testing following the criteria listed below now enables a cost-effective (122), often cost-saving, genetic diagnosis that is increasingly supported by health insurance. A biomarker screening pathway such as the combination of urinary C-peptide/ creatinine ratio and antibody screening may aid in determining who should get genetic testing for MODY (123). It is critical to correctly diagnose one of the monogenic forms of diabetes because these patients may be incorrectly diagnosed with type 1 or type 2 diabetes, leading to suboptimal, even potentially harmful, treatment regimens and delays in diagnosing other family members (124). The correct diagnosis is especially critical for those with GCK-MODY mutations where multiple studies have shown that no complications ensue in the absence of glucose-lowering therapy (125). Genetic counseling is recommended to ensure that affected individuals understand the patterns of inheritance and the importance of a correct diagnosis.
The diagnosis of monogenic diabetes should be considered in children and adults diagnosed with diabetes in early adulthood with the following findings:
Diabetes diagnosed within the first 6 months of life (with occasional cases presenting later, mostly INS and ABCC8 mutations) (120,126)
Diabetes without typical features of type 1 or type 2 diabetes (negative diabetes-associated autoantibodies, nonobese, lacking other metabolic features especially with strong family history of diabetes)
Stable, mild fasting hyperglycemia (100–150 mg/dL [5.5–8.5 mmol/L]), stable A1C between 5.6 and 7.6% (between 38 and 60 mmol/mol), especially if nonobese
xxxxx
9.0.0.0 References
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014;37(Suppl. 1):S81–S90
- Dabelea D, Rewers A, Stafford JM, et al.; SEARCH for Diabetes in Youth Study Group. Trends in the prevalence of ketoacidosis at diabetes diagnosis: the SEARCH for Diabetes in Youth Study. Pediatrics 2014;133:e938–e945
- Newton CA, Raskin P. Diabetic ketoacidosis in type 1 and type 2 diabetes mellitus: clinical and biochemical differences. Arch Intern Med 2004; 164:1925–1931
- Skyler JS, Bakris GL, Bonifacio E, et al. Differentiationof diabetes by pathophysiology, natural history, and prognosis. Diabetes 2017;66:241– 255
- Insel RA, Dunne JL, Atkinson MA, et al. Staging presymptomatic type 1 diabetes: a scientific statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care 2015;38:1964–1974
- International Expert Committee. International Expert Committee report on the role of the A1C assay in the diagnosis of diabetes. Diabetes Care 2009;32:1327–1334
- Knowler WC, Barrett-Connor E, Fowler SE, et al.; Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002;346:393–403
- Tuomilehto J, Lindstro¨m J, Eriksson JG, et al.; Finnish Diabetes Prevention Study Group. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 2001;344: 1343–1350
- Meijnikman AS, De Block CEM, Dirinck E, et al. Not performing an OGTT results in significant underdiagnosis of (pre)diabetes in a high risk adult Caucasian population. Int J Obes 2017;41: 1615–1620
- Cowie CC, Rust KF, Byrd-Holt DD, et al. Prevalence of diabetes and high risk for diabetes using A1C criteria in the U.S. population in 1988–2006. Diabetes Care 2010;33:562–568
- Eckhardt BJ, Holzman RS, Kwan CK, Baghdadi J, Aberg JA. Glycated hemoglobin A1c as screening for diabetes mellitus in HIV-infected individuals. AIDS Patient Care STDS 2012;26:197–201
- Kim PS, Woods C, Georgoff P, et al. A1C underestimates glycemia in HIV infection. Diabetes Care 2009;32:1591–1593
- Arslanian S, Bacha F, Grey M, Marcus MD, White NH, Zeitler P. Evaluation and management of youth-onset type 2 diabetes: a position statement by the American Diabetes Association. Diabetes Care 2018;41:2648–2668
- Lacy ME, Wellenius GA, Sumner AE, Correa A, Carnethon MR, Liem RI, et al. Association of sickle cell trait with hemoglobin A1c in African Americans. JAMA 2017;317:507–515
- Wheeler E, Leong A, Liu C-T, et al.; EPIC-CVD Consortium; EPIC-InterAct Consortium; Lifelines Cohort Study. Impact of common genetic determinants of hemoglobin A1c on type 2 diabetes risk and diagnosis in ancestrally diverse populations: a transethnic genome-wide metaanalysis. PLoS Med 2017;14:e1002383
- Ziemer DC, Kolm P, Weintraub WS, et al. Glucose-independent, black-white differences in hemoglobin A1c levels: a cross-sectional analysis of 2 studies. Ann Intern Med 2010;152:770–777
- Kumar PR, Bhansali A, Ravikiran M, et al. Utility of glycated hemoglobin in diagnosing type 2 diabetes mellitus: a community-based study. J Clin Endocrinol Metab 2010;95:2832– 2835
- Herman WH. Are there clinical implications of racial differences in HbA1c? Yes, to not consider can do great harm! Diabetes Care 2016;39: 1458–1461
- Herman WH, Ma Y, Uwaifo G, et al.; Diabetes Prevention Program Research Group. Differences in A1C by race and ethnicity among patients with impaired glucose tolerance in the Diabetes Prevention Program. Diabetes Care 2007;30: 2453–2457
- Bergenstal RM, Gal RL, Connor CG, et al.; T1D Exchange Racial Differences Study Group. Racial differences in the relationship of glucose concentrations and hemoglobin A1c levels. Ann Intern Med 2017;167:95–102
- Selvin E, Steffes MW, Ballantyne CM, Hoogeveen RC, Coresh J, Brancati FL. Racial differencesin glycemicmarkers: across-sectional analysis of community-based data. Ann Intern Med 2011;154:303–309
- Herman WH, Dungan KM, Wolffenbuttel BHR, et al. Racial and ethnic differences in mean plasma glucose, hemoglobin A1c, and 1,5-anhydroglucitol in over 2000 patients with type 2 diabetes. J Clin Endocrinol Metab 2009; 94:1689–1694
- Selvin E, Rawlings AM, Bergenstal RM, Coresh J, Brancati FL. No racial differences in the association of glycated hemoglobin with kidney disease and cardiovascular outcomes. Diabetes Care 2013;36:2995–3001
- Selvin E. Are there clinical implications of racial differences in HbA1c? A difference, to be a difference, must make a difference. Diabetes Care 2016;39:1462–1467
- Paterson AD. HbA1c for type 2 diabetes diagnosis in Africans and African Americans: personalized medicine NOW! PLoS Med 2017; 14:e1002384
- Cappellini MD, Fiorelli G. Glucose-6-phosphate dehydrogenase deficiency. Lancet 2008;371:64–74
- Pico´n MJ, Murri M, Mun~oz A, Ferna´ndez-Garc´ıa JC, Gomez-Huelgas R, Tinahones FJ. Hemoglobin A1c versus oral glucose tolerance test in postpartum diabetes screening. Diabetes Care 2012;35:1648–1653
- Go¨bl CS, Bozkurt L, Yarragudi R, Tura A, Pacini G, Kautzky-Willer A. Is early postpartum HbA1c an appropriate risk predictor after pregnancy with gestational diabetes mellitus? Acta Diabetol 2014;51:715–722
- Megia A, Na¨f S, Herranz L, et al. The usefulness of HbA1c in postpartum reclassification of gestational diabetes. BJOG 2012;119:891–894
- Welsh KJ, Kirkman MS, Sacks DB. Role of glycated proteins in the diagnosis and management of diabetes: research gaps and future directions. Diabetes Care 2016;39:1299–1306
- Kim C, Bullard KM, Herman WH, Beckles GL. Association between iron deficiency and A1C levels among adults without diabetes in the National Health and Nutrition Examination Survey, 1999-2006. Diabetes Care 2010;33:780–785
- Selvin E, Wang D, Matsushita K, Grams ME, Coresh J. Prognostic implications of singlesample confirmatory testing for undiagnosed diabetes: a prospective cohort study. Ann Intern Med 2018;169:156–164
- Dabelea D, Mayer-Davis EJ, Saydah S, et al.; SEARCH for Diabetes in Youth Study. Prevalence of type 1 and type 2 diabetes among children and adolescents from 2001 to 2009. JAMA 2014;311: 1778–1786
- Ziegler AG, Rewers M, Simell O, et al. Seroconversion to multiple islet autoantibodies and risk of progression to diabetes in children. JAMA 2013;309:2473–2479
- Sosenko JM, Skyler JS, Palmer JP, et al.; Type 1 Diabetes TrialNet Study Group; Diabetes Prevention Trial-Type 1 Study Group. The prediction of type 1 diabetes by multiple autoantibody levels and their incorporation into an autoantibody risk score in relatives of type 1 diabetic patients. Diabetes Care 2013;36:2615–2620
- Steck AK, Vehik K, Bonifacio E, et al.; TEDDY Study Group. Predictors of progression from the appearance of islet autoantibodies to early childhood diabetes: The Environmental Determinants of Diabetes in the Young (TEDDY). Diabetes Care 2015;38:808–813
- Orban T, Sosenko JM, Cuthbertson D, et al.; Diabetes Prevention Trial-Type 1 Study Group. Pancreatic islet autoantibodies as predictors of type 1 diabetes in the Diabetes Prevention Trial–Type 1. Diabetes Care 2009;32:2269– 2274
- Genuth S, Alberti KG, Bennett P, et al.; Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Follow-up report on the diagnosis of diabetes mellitus. Diabetes Care 2003; 26:3160–3167
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2011;34(Suppl. 1):S62–S69
- Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 1997;20:1183–1197
- Zhang X, Gregg EW, Williamson DF, et al. A1C level and future risk of diabetes: a systematic review. Diabetes Care 2010;33:1665–1673
- Selvin E, Steffes MW, Zhu H, et al. Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults. N Engl J Med 2010;362: 800–811
- Ackermann RT, Cheng YJ, Williamson DF, Gregg EW. Identifying adults at high risk for diabetes and cardiovascular disease using hemoglobin A1c National Health and Nutrition Examination Survey 2005-2006. Am J Prev Med 2011;40:11–17
- Diabetes Prevention Program Research Group. HbA1c as a predictor of diabetes and as an outcome in the Diabetes Prevention Program: a randomized clinical trial. Diabetes Care 2015;38:51–58
- Umpierrez G, Korytkowski M. Diabetic emergencies - ketoacidosis, hyperglycaemic hyperosmolar state and hypoglycaemia. Nat Rev Endocrinol 2016;12:222–232
- Fadini GP, Bonora BM, Avogaro A. SGLT2 inhibitors and diabetic ketoacidosis: data from the FDA Adverse Event Reporting System. Diabetologia 2017;60:1385–1389
- Griffin SJ, Borch-Johnsen K, Davies MJ, et al. Effect of early intensive multifactorial therapy on 5-year cardiovascular outcomes in individuals with type 2 diabetes detected by screening (ADDITION-Europe): a cluster-randomised trial. Lancet 2011;378:156–167
- Herman WH, Ye W, Griffin SJ, et al. Early detection and treatment of type 2 diabetes reduce cardiovascular morbidity and mortality: a simulation of the results of the Anglo-Danish-Dutch Study of Intensive Treatment in People with Screen-Detected Diabetes in Primary Care (ADDITION-Europe). Diabetes Care 2015;38: 1449–1455
- Kahn R, Alperin P, Eddy D, et al. Age at initiation and frequency of screening to detect type 2 diabetes: a cost-effectiveness analysis. Lancet 2010;375:1365–1374
- Araneta MRG, Kanaya A, Fujimoto W, et al. Optimum BMI cut-points to screen Asian Americans for type 2 diabetes: The UCSD Filipino Health Study and the North Kohala Study [Abstract]. Diabetes 2014;63(Suppl. 1):A20
- Hsu WC, Araneta MRG, Kanaya AM, Chiang JL, Fujimoto W. BMI cut points to identify at-risk Asian Americans for type 2 diabetes screening. Diabetes Care 2015;38:150–158
- WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 2004;363:157–163
- Menke A, Casagrande S, Geiss L, Cowie CC. Prevalence of and trends in diabetes among adults in the United States, 1988-2012. JAMA 2015;314:1021–1029
- Centers for Disease Control and Prevention. National diabetes statistics report: estimates of diabetes and its burden in the United States, 2017 [Internet]. Available from https://www.cdc.gov/diabetes/data/statistics/statistics-report.html. Accessed 20 September 2018
- Chiu M, Austin PC, Manuel DG, Shah BR, Tu JV. Deriving ethnic-specific BMI cutoff points for assessing diabetes risk. Diabetes Care 2011;34: 1741–1748
- Erickson SC, Le L, Zakharyan A, et al. New-onset treatment-dependent diabetes mellitus and hyperlipidemia associated with atypical antipsychotic use in older adults without schizophrenia or bipolar disorder. J Am Geriatr Soc 2012;60:474–479
- Johnson SL, Tabaei BP, Herman WH. The efficacy and cost of alternative strategies for systematic screening for type 2 diabetes in the U.S. population 45–74 years of age. Diabetes Care 2005;28:307–311
- Tabaei BP, Burke R, Constance A, et al. Community-based screening for diabetes in Michigan. Diabetes Care 2003;26:668–670
- Lalla E, Kunzel C, Burkett S, Cheng B, Lamster IB. Identification of unrecognized diabetes and pre-diabetes in a dental setting. J Dent Res 2011; 90:855–860
- Lalla E, Cheng B, Kunzel C, Burkett S, Lamster IB. Dental findings and identification of undiagnosed hyperglycemia. J Dent Res 2013;92:888– 892
- Herman WH, Taylor GW, Jacobson JJ, Burke R, Brown MB. Screening for prediabetes and type 2 diabetes in dental offices. J Public Health Dent 2015;75:175–182
- Buse JB, Kaufman FR, Linder B, Hirst K, El Ghormli L, Willi S; HEALTHY Study Group. Diabetes screening with hemoglobin A1c versus fasting plasma glucose in a multiethnic middle-school cohort. Diabetes Care 2013;36:429–435
- Kapadia C, Zeitler P; Drugs and Therapeutics Committee of the Pediatric Endocrine Society. Hemoglobin A1c measurement for the diagnosis of type 2 diabetes in children. Int J Pediatr Endocrinol 2012;2012:31
- Kester LM, Hey H, Hannon TS. Using hemoglobin A1c for prediabetes and diabetes diagnosis in adolescents: can adult recommendations be upheld for pediatric use? J Adolesc Health 2012;50:321–323
- Wu E-L, Kazzi NG, Lee JM. Cost-effectiveness of screening strategies for identifying pediatric diabetes mellitus and dysglycemia. JAMA Pediatr 2013;167:32–39
- Lawrence JM, Contreras R, Chen W, Sacks DA. Trends in the prevalence of preexisting diabetes and gestational diabetes mellitus among a racially/ethnically diverse population of pregnant women, 1999–2005. Diabetes Care 2008;31:899–904
- Poltavskiy E, Kim DJ, Bang H. Comparison of screening scores for diabetes and prediabetes. Diabetes Res Clin Pract 2016;118:146–153
- Hughes RCE, Rowan J, Williman J. Prediabetes in pregnancy, can early intervention improve outcomes? A feasibility study for a parallel randomised clinical trial. BMJ Open 2018;8: e018493
- McIntyre HD, Sacks DA, Barbour LA, et al. Issues with the diagnosis and classification of hyperglycemia in early pregnancy. Diabetes Care 2016;39:53–54
- Noctor E, Crowe C, Carmody LA, et al.; ATLANTIC-DIP investigators. Abnormal glucose tolerance post-gestational diabetes mellitus as defined by the International Association of Diabetes and Pregnancy Study Groups criteria. Eur J Endocrinol 2016;175:287–297
- Kim C, Newton KM, Knopp RH. Gestational diabetes and the incidence of type 2 diabetes: a systematic review. Diabetes Care 2002;25: 1862–1868
- Ratner RE, Christophi CA, Metzger BE, et al.; Diabetes Prevention Program Research Group. Prevention ofdiabetesinwomenwithahistory of gestational diabetes: effects of metformin and lifestyle interventions. J Clin Endocrinol Metab 2008;93:4774–4779
- Aroda VR, Christophi CA, Edelstein SL, et al.; Diabetes Prevention Program Research Group. The effect of lifestyle intervention and metformin on preventing or delaying diabetes among women with and without gestational diabetes: the Diabetes Prevention Program Outcomes Study 10-year follow-up. J Clin Endocrinol Metab 2015;100:1646–1653
- Metzger BE, Lowe LP, Dyer AR, et al.; HAPO Study Cooperative Research Group. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 2008;358:1991–2002
- Sacks DA, Hadden DR, Maresh M, et al.; HAPO Study Cooperative Research Group. Frequency of gestational diabetes mellitus at collaborating centers based on IADPSG consensus panel-recommended criteria: the Hyperglycemia and Adverse Pregnancy Outcome (HAPO) Study. Diabetes Care 2012;35:526–528
- Lowe WL Jr, Scholtens DM, Lowe LP, et al.; HAPO Follow-up Study Cooperative Research Group. Association of gestational diabetes with maternal disorders of glucose metabolism and childhood adiposity. JAMA 2018;320:1005–016
- Landon MB, Spong CY, Thom E, et al.; Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. A multicenter, randomized trial of treatment for mild gestational diabetes. N Engl J Med 2009;361:1339–1348
- Crowther CA, Hiller JE, Moss JR, McPhee AJ, Jeffries WS, Robinson JS; Australian Carbohydrate Intolerance Study in Pregnant Women (ACHOIS) Trial Group. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N Engl J Med 2005; 352:2477–2486
- Tam WH, Ma RCW, Ozaki R, et al. In utero exposure to maternal hyperglycemia increases childhood cardiometabolic risk in offspring. Diabetes Care 2017;40:679–686
- Landon MB, Rice MM, Varner MW, et al.; Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units (MFMU) Network. Mild gestational diabetes mellitus and long-term child health. Diabetes Care 2015;38:445–452
- Vandorsten JP, Dodson WC, Espeland MA, et al. NIH consensus development conference: diagnosing gestational diabetes mellitus. NIH Consens State Sci Statements 2013;29:1–31
- Committee on Practice BulletinsdObstetrics. Practice Bulletin No. 190: gestational diabetes mellitus. Obstet Gynecol 2018;131:e49–e64
- Donovan L, Hartling L, Muise M, Guthrie A, Vandermeer B, Dryden DM. Screening tests for gestational diabetes: a systematic review for the U.S. Preventive Services Task Force. Ann Intern Med 2013;159:115–122
- Khalafallah A, Phuah E, Al-Barazan AM, et al. Glycosylated haemoglobin for screening and diagnosis of gestational diabetes mellitus. BMJ Open 2016;6:e011059
- Horvath K, Koch K, Jeitler K, et al. Effects of treatment in women with gestational diabetes mellitus: systematic review and meta-analysis. BMJ 2010;340:c1395
- Carpenter MW, Coustan DR. Criteria for screening tests for gestational diabetes. Am J Obstet Gynecol 1982;144:768–773
- National Diabetes Data Group. Classification and diagnosis of diabetes mellitus and other categories of glucose intolerance. Diabetes 1979; 28:1039–1057
- Harper LM, Mele L, Landon MB, et al.; Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Maternal-Fetal Medicine Units (MFMU) Network. Carpenter-Coustan compared with National Diabetes Data Group criteria for diagnosing gestational diabetes. Obstet Gynecol 2016;127:893–898
- Werner EF, Pettker CM, Zuckerwise L, et al. Screening for gestational diabetes mellitus: are the criteria proposed by the International Association of the Diabetes and Pregnancy Study Groups cost-effective? Diabetes Care 2012;35: 529–535
- Duran A, Sa´enz S, Torrejo´n MJ, et al. Introduction of IADPSG criteria for the screening and diagnosis of gestational diabetes mellitus results in improved pregnancy outcomes at a lower cost in a large cohort of pregnant women: the St. Carlos Gestational Diabetes Study. Diabetes Care 2014;37:2442–2450
- Wei Y, Yang H, Zhu W, et al. International Association of Diabetes and Pregnancy Study Group criteria is suitable for gestational diabetes mellitus diagnosis: further evidence from China. Chin Med J (Engl) 2014;127:3553–3556
- Feldman RK, Tieu RS, Yasumura L. Gestational diabetes screening: the International Association of the Diabetes and Pregnancy Study Groups compared with Carpenter-Coustan screening. Obstet Gynecol 2016;127:10–17
- Ethridge JK Jr, Catalano PM, Waters TP. Perinatal outcomes associated with the diagnosis of gestational diabetes made by the International Association of the Diabetes and Pregnancy Study Groups criteria. Obstet Gynecol 2014;124: 571–578
- Mayo K, Melamed N, Vandenberghe H, Berger H. The impact of adoption of the international association of diabetes in pregnancy study group criteria for the screening and diagnosis of gestational diabetes. Am J Obstet Gynecol 2015;212:224.e1–224.e9
- Moran A, Pillay K, Becker D, Granados A, Hameed S, Acerini CL. ISPAD Clinical Practice Consensus Guidelines 2018: management of cystic fibrosis-related diabetes in children and adolescents. Pediatr Diabetes 2018;19(Suppl. 27):64–74
- Mainguy C, Bellon G, Delaup V, et al. Sensitivity and specificity of different methods for cystic fibrosis-related diabetes screening: is the oral glucose tolerance test still the standard? J Pediatr Endocrinol Metab 2017; 30:27–35
- Ode KL, Moran A. New insights into cystic fibrosis-related diabetes in children. Lancet Diabetes Endocrinol 2013;1:52–58
- Moran A, Dunitz J, Nathan B, Saeed A, Holme B, Thomas W. Cystic fibrosis–related diabetes: current trends in prevalence, incidence, and mortality. Diabetes Care 2009;32:1626–1631
- Moran A, Pekow P, Grover P, et al.; Cystic Fibrosis Related Diabetes Therapy Study Group. Insulin therapy to improve BMI in cystic fibrosis– related diabetes without fasting hyperglycemia: results of the Cystic Fibrosis Related Diabetes Therapy Trial. Diabetes Care 2009;32: 1783–1788
- Onady GM, Stolfi A. Insulin and oral agents for managing cystic fibrosis–related diabetes. Cochrane Database Syst Rev 2016;4:CD004730
- Moran A, Brunzell C, Cohen RC, et al.; CFRD Guidelines Committee. Clinical care guidelines for cystic fibrosis–related diabetes: a position statement of the American Diabetes Association and a clinical practice guideline of the Cystic Fibrosis Foundation, endorsed by the Pediatric Endocrine Society. Diabetes Care 2010;33:2697–2708
- Moran A, Pillay K, Becker DJ, Acerini CL; International Society for Pediatric and Adolescent Diabetes. ISPAD Clinical Practice Consensus Guidelines 2014. Management of cystic fibrosi-related diabetes in children and adolescents. Pediatr Diabetes 2014;15(Suppl. 20):65–76
- Sharif A, Hecking M, de Vries APJ, et al. Proceedings from an international consensus meeting on posttransplantation diabetes mellitus: recommendations and future directions. Am J Transplant 2014;14:1992–2000
- Hecking M, Werzowa J, Haidinger M, et al. Novel views on new-onset diabetes after transplantation: development, prevention and treatment. Nephrol Dial Transplant 2013;28:550–566
- Ramirez SC, Maaske J, Kim Y, et al. The association between glycemic control and clin-ical outcomes after kidney transplantation. En-docr Pract 2014;20:894–900
- Thomas MC, Moran J, Mathew TH, Russ GR, Rao MM. Early peri-operative hyperglycaemia and renal allograft rejection in patients without diabetes. BMC Nephrol 2000;1:1
- Chakkera HA, Weil EJ, Castro J, et al. Hyperglycemia during the immediate period after kidney transplantation. Clin J Am Soc Nephrol 2009;4:853–859
- Wallia A, Illuri V, Molitch ME. Diabetes care after transplant: definitions, risk factors, and clinical management. Med Clin North Am 2016;100: 535–550
- Sharif A, Moore RH, Baboolal K. The use of oral glucose tolerance tests to risk stratify for new-onset diabetes after transplantation: an underdiagnosed phenomenon. Transplantation 2006;82:1667–1672
- Hecking M, Kainz A, Werzowa J, et al. Glucose metabolism after renal transplantation. Diabetes Care 2013;36:2763–2771
- Galindo RJ, Fried M, Breen T, Tamler R. Hyperglycemia management in patients with posttransplantation diabetes. Endocr Pract 2016;22:454–465
- Jenssen T, Hartmann A. Emerging treatments for post-transplantation diabetes mellitus. Nat Rev Nephrol 2015;11:465–477
- Thomas MC, Mathew TH, Russ GR, Rao MM, Moran J. Early peri-operative glycaemic control and allograft rejection in patients with diabetes mellitus: a pilot study. Transplantation 2001;72:1321–1324
- Kurian B, Joshi R, Helmuth A. Effectiveness and long-term safety of thiazolidinediones and metformin in renal transplant recipients. Endocr Pract 2008;14:979–984
- Budde K, Neumayer H-H, Fritsche L, Sulowicz W, Stompor T, Eckland D. The pharmacokinetics of pioglitazone in patients with impaired renal function. Br J Clin Pharmacol 2003; 55:368–374
- Luther P, Baldwin D Jr. Pioglitazone in the management of diabetes mellitus after transplantation. Am J Transplant 2004;4:2135– 2138
- Strøm Halden TA, ° Asberg A, Vik K, Hartmann A, Jenssen T. Short-term efficacy and safety of sitagliptin treatment in longterm stable renal recipients with new-onset diabetes after transplantation. Nephrol Dial Transplant 2014;29:926–933
- Lane JT, Odegaard DE, Haire CE, Collier DS, Wrenshall LE, Stevens RB. Sitagliptin therapy in kidney transplant recipients with new-onset diabetes after transplantation. Transplantation 2011;92:e56–e57
- Carmody D, Støy J, Greeley SA, Bell GI, Philipson LH. A clinical guide to monogenic diabetes. In Genetic Diagnosis of Endocrine Disorders. 2nd ed. Weiss RE, Refetoff S, Eds. Philadelphia, PA, Elsevier, 2016
- De Franco E, Flanagan SE, Houghton JAL, et al. The effect of early, comprehensive genomic testing on clinical care in neonatal diabetes: an international cohort study. Lancet 2015;386: 957–963
- Urbanova´ J, Rypa´ckova´ B, Procha´zkova´ Z, et al. Positivity for islet cell autoantibodies in patients with monogenic diabetes is associated with later diabetes onset and higher HbA1c level. Diabet Med 2014;31:466–471
- Naylor RN, John PM, Winn AN, et al. Cost-effectiveness of MODY genetic testing: translating genomic advances into practical health applications. Diabetes Care 2014;37: 202–209
- Shields BM, Shepherd M, Hudson M, et al.; UNITED study team. Population-based assessment of a biomarker-based screening pathway to aid diagnosis of monogenic diabetes in young-onset patients. Diabetes Care 2017;40:1017– 1025
- Hattersley A, Bruining J, Shield J, Njolstad P, Donaghue KC. The diagnosis and management of monogenic diabetes in children and adolescents. Pediatr Diabetes 2009;10(Suppl. 12):33– 42
- Rubio-Cabezas O, Hattersley AT, Njølstad PR, et al.; International Society for Pediatric and Adolescent Diabetes. ISPAD Clinical Practice Consensus Guidelines 2014. The diagnosis and management of monogenic diabetes in children and adolescents. Pediatr Diabetes 2014;15 (Suppl. 20):47–64
- Greeley SAW, Naylor RN, Philipson LH, Bell GI. Neonatal diabetes: an expanding list of genes allows for improved diagnosis and treatment. Curr Diab Rep 2011;11:519–532